Acids and bases are fundamental concepts in chemistry that help explain how substances interact, react, and transform in chemical systems. Over time, scientists have developed several frameworks to describe acid–base behavior more accurately across different environments and reactions.
These frameworks—known as acid–base theories—allow chemists to understand chemical reactions ranging from simple neutralization in water to complex reactions in organic chemistry and industrial processes.
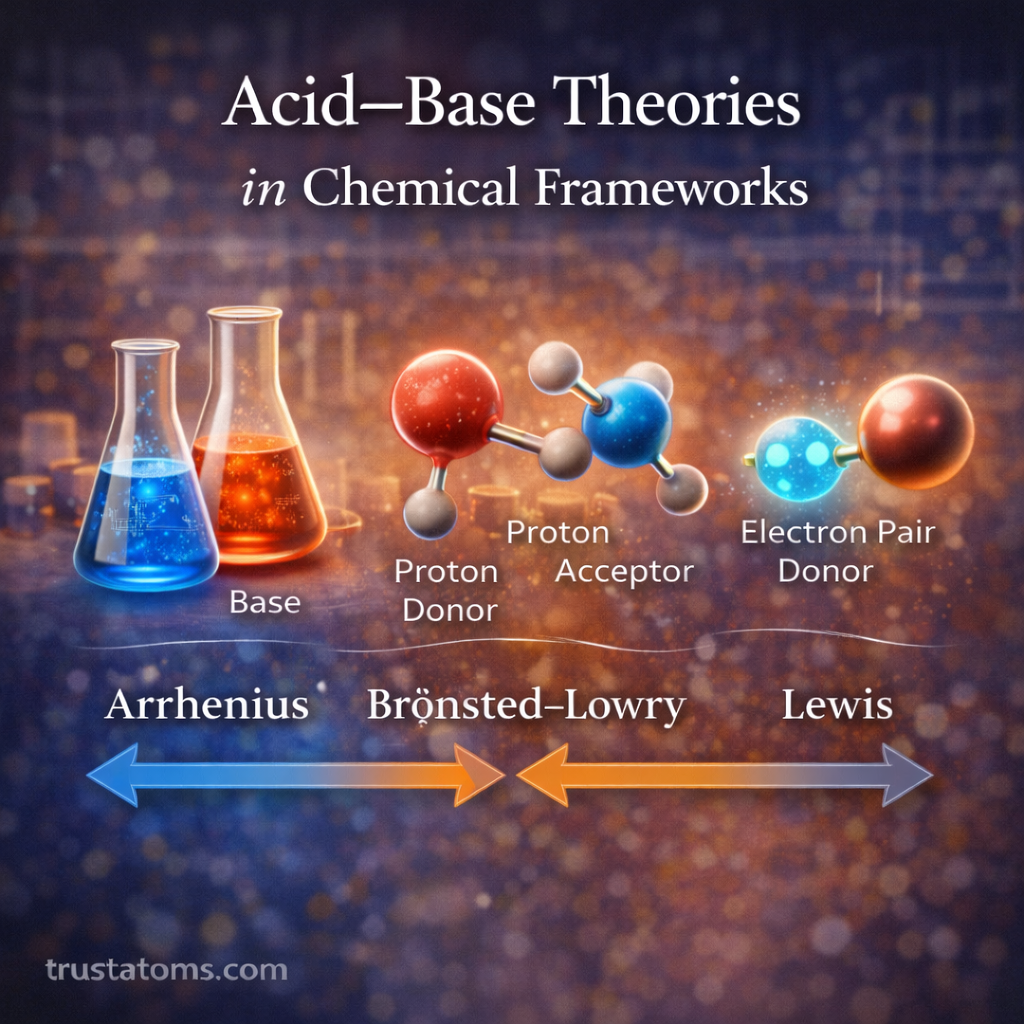
Three major theories are commonly used to explain acid–base interactions:
- Arrhenius theory
- Brønsted–Lowry theory
- Lewis theory
Each theory expands on the previous one, providing a broader explanation of chemical behavior.
What Are Acids and Bases?
In general terms, acids and bases are substances that participate in reactions involving protons, ions, or electron pairs.
Acids often display properties such as:
- Sour taste
- Ability to react with metals
- Capability to donate hydrogen ions
- Conductivity in aqueous solutions
Bases often show characteristics such as:
- Bitter taste
- Slippery feel
- Ability to neutralize acids
- Production of hydroxide ions in solution
However, the scientific definitions of acids and bases depend on the theoretical framework being used.
The Arrhenius Acid–Base Theory
The Arrhenius theory, developed in the late 19th century by Swedish chemist Svante Arrhenius, provides the earliest scientific explanation of acids and bases.
According to Arrhenius:
- Acids produce hydrogen ions (H⁺) in water.
- Bases produce hydroxide ions (OH⁻) in water.
Examples of Arrhenius Acids
Common Arrhenius acids include:
- Hydrochloric acid
- Nitric acid
- Sulfuric acid
These substances release hydrogen ions when dissolved in water.
Examples of Arrhenius Bases
Typical Arrhenius bases include:
- Sodium hydroxide
- Potassium hydroxide
- Calcium hydroxide
These compounds release hydroxide ions in aqueous solutions.
Limitations of the Arrhenius Theory
Although useful, the Arrhenius model has several limitations:
- It applies only to reactions in aqueous solutions
- It cannot explain reactions that occur without hydroxide ions
- It does not account for many acid–base reactions in organic chemistry
Because of these limitations, broader theories were developed.
The Brønsted–Lowry Acid–Base Theory
In 1923, chemists Johannes Brønsted and Thomas Lowry independently proposed a more flexible model for acid–base behavior.
According to the Brønsted–Lowry theory:
- An acid is a proton donor.
- A base is a proton acceptor.
This theory focuses on the transfer of hydrogen ions (protons) between substances.
Proton Transfer Reactions
In a Brønsted–Lowry reaction:
- The acid donates a proton.
- The base accepts the proton.
For example, when hydrogen chloride reacts with water:
- Hydrogen chloride donates a proton.
- Water accepts the proton.
This process forms hydronium ions and chloride ions.
Conjugate Acid–Base Pairs
One important concept in this theory is the conjugate acid–base pair.
When an acid donates a proton, it forms a conjugate base.
When a base accepts a proton, it forms a conjugate acid.
Example pair relationships:
- Acid → conjugate base
- Base → conjugate acid
These pairs help chemists understand reversible reactions in acid–base chemistry.
The Lewis Acid–Base Theory

The Lewis theory, developed by Gilbert N. Lewis in 1923, provides the broadest definition of acids and bases.
According to Lewis:
- A Lewis acid accepts an electron pair.
- A Lewis base donates an electron pair.
Unlike earlier theories, the Lewis model does not require protons or aqueous solutions.
Lewis Acid–Base Reactions
Lewis acid–base reactions involve the formation of coordinate covalent bonds, where both electrons in a bond come from the same atom.
Examples include:
- Metal ion interactions with ligands
- Reactions involving electron-deficient molecules
- Complex formation in coordination chemistry
Because it focuses on electron pair interactions, the Lewis theory applies to many reactions beyond traditional acid–base chemistry.
Comparing the Three Acid–Base Theories
Each theory describes acid–base behavior from a different perspective.
Arrhenius Theory
- Focuses on hydrogen and hydroxide ions
- Applies mainly to aqueous solutions
- Useful for simple acid–base reactions
Brønsted–Lowry Theory
- Describes proton transfer reactions
- Applies to a wider range of chemical systems
- Introduces conjugate acid–base pairs
Lewis Theory
- Focuses on electron pair exchange
- Applies to many types of chemical reactions
- Widely used in modern chemistry
These theories are often used together to analyze chemical behavior.
Acid–Base Reactions in Chemical Systems
Acid–base reactions play a central role in many chemical processes.
Common types of reactions include:
Neutralization Reactions
Acids and bases react to form water and salts.
Buffer Systems
Solutions that resist changes in pH by balancing acids and bases.
Coordination Chemistry
Metal ions interact with electron-donating molecules.
Organic Reactions
Many organic mechanisms involve proton transfer or electron pair donation.
Understanding these reactions helps chemists predict chemical outcomes.
Applications of Acid–Base Chemistry
Acid–base chemistry influences many scientific and industrial processes.
Examples include:
Biological Systems
- Enzyme activity depends on pH levels
- Blood chemistry relies on buffering systems
Environmental Chemistry
- Ocean acidification
- Soil chemistry and nutrient availability
Industrial Chemistry
- Fertilizer production
- Chemical manufacturing processes
Medicine and Pharmaceuticals
- Drug stability and formulation
- Antacid medications
These applications highlight the importance of acid–base frameworks in real-world systems.
Why Acid–Base Theories Matter
Understanding acid–base theories helps chemists interpret chemical behavior in a wide range of contexts.
Key benefits include:
- Predicting reaction behavior
- Understanding molecular interactions
- Designing chemical processes
- Explaining biological chemical systems
These frameworks provide essential tools for analyzing chemical reactions and molecular structures.
Final Thoughts
Acid–base theories have evolved over time to provide increasingly comprehensive explanations of chemical behavior. From the early Arrhenius model to the broader Brønsted–Lowry and Lewis frameworks, each theory expands our ability to understand chemical interactions.
Together, these theories form the foundation of modern acid–base chemistry, helping scientists analyze reactions in laboratory research, industrial processes, and biological systems.
By applying these frameworks, chemists can better understand how substances interact, react, and transform within complex chemical environments.