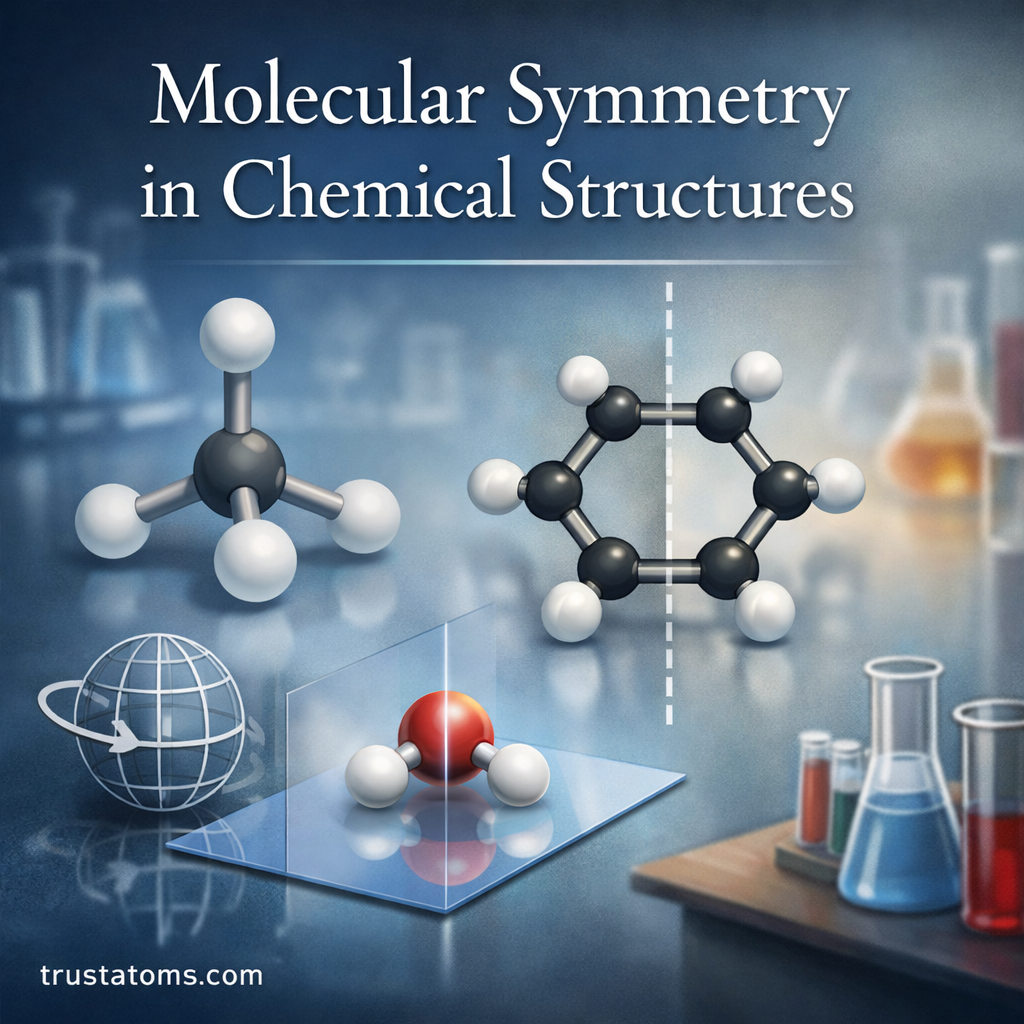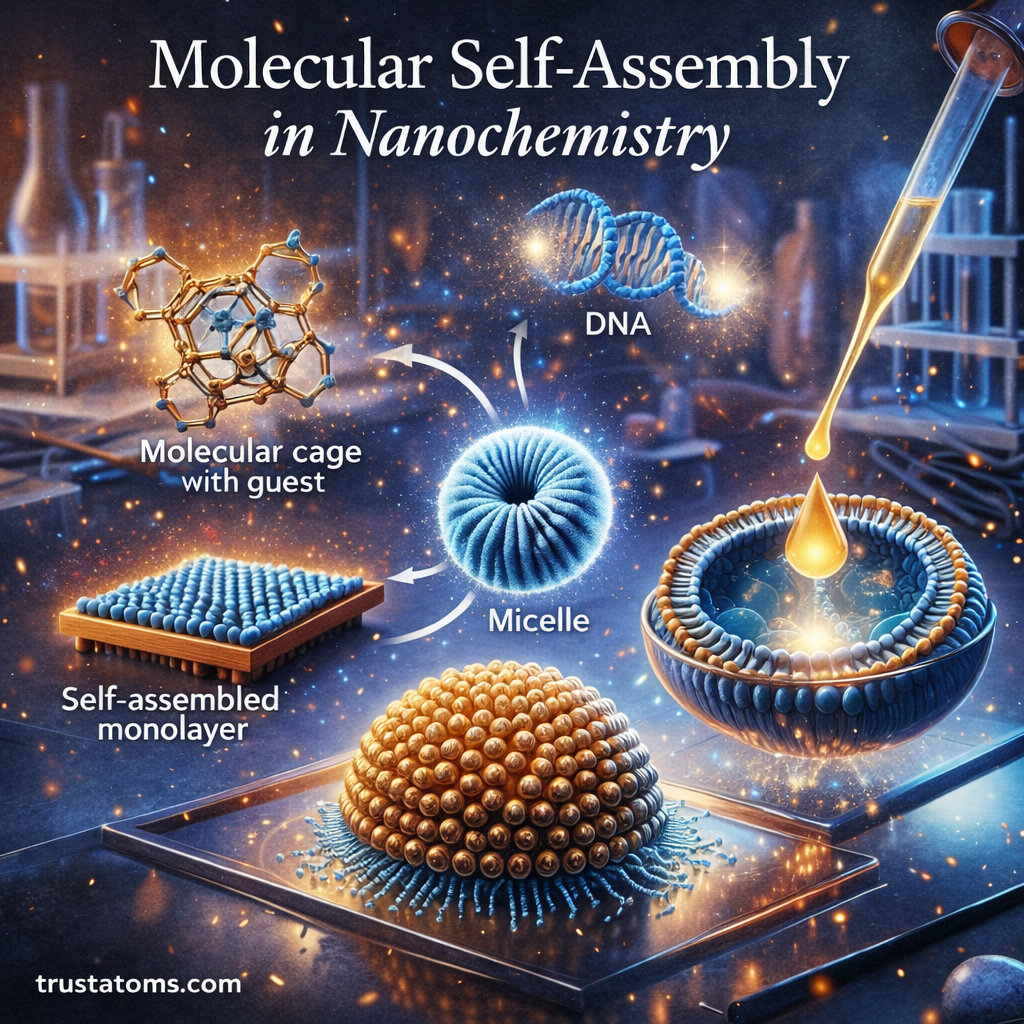
Molecular self-assembly is a fundamental concept in nanochemistry where molecules organize themselves into structured arrangements without external direction. Through natural chemical interactions, individual molecules can spontaneously form ordered patterns, layers, and complex nanostructures.
This phenomenon plays a critical role in the creation of advanced materials, nanotechnology devices, drug delivery systems, and biological structures. By understanding and controlling self-assembly processes, scientists can design materials at the molecular scale with precise functions and properties.
In many ways, molecular self-assembly allows chemistry to mimic nature’s ability to build complex structures efficiently and reliably.
Continue reading “Molecular Self-Assembly in Nanochemistry”