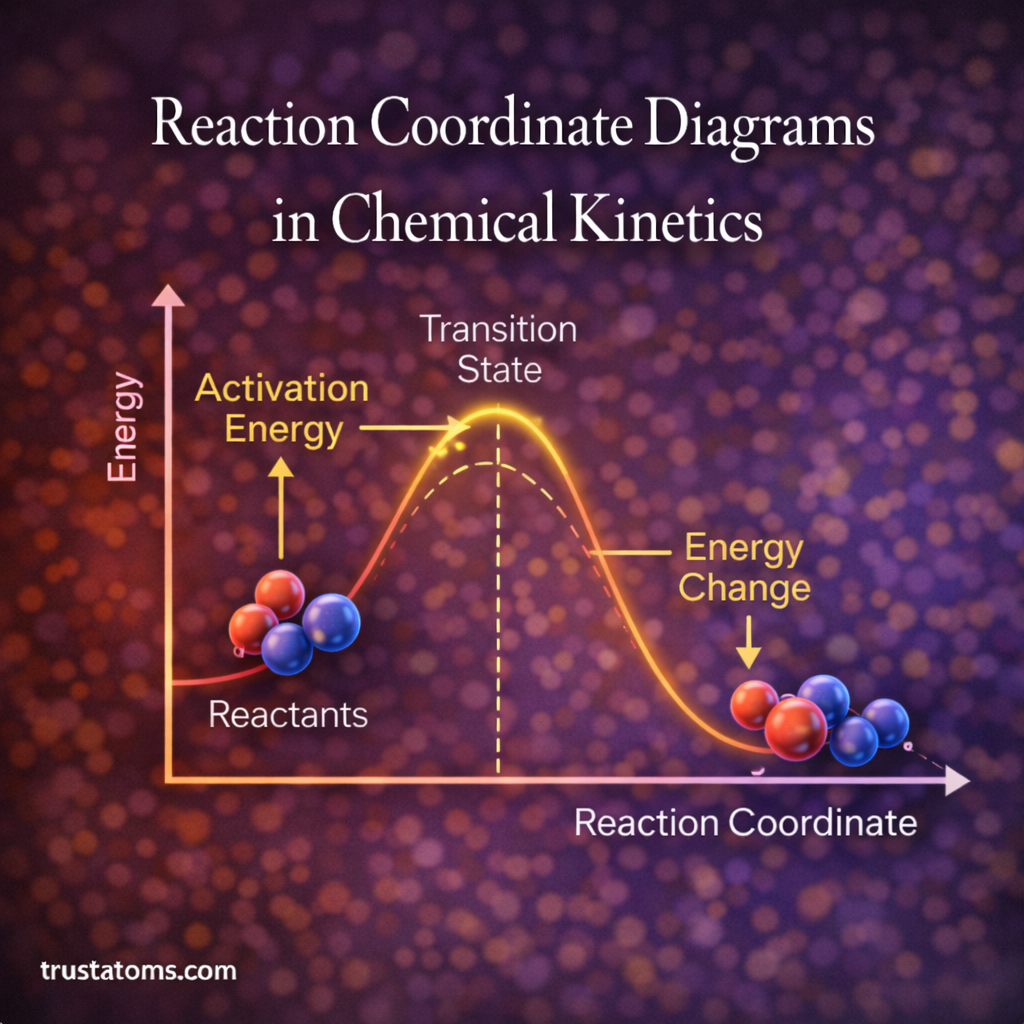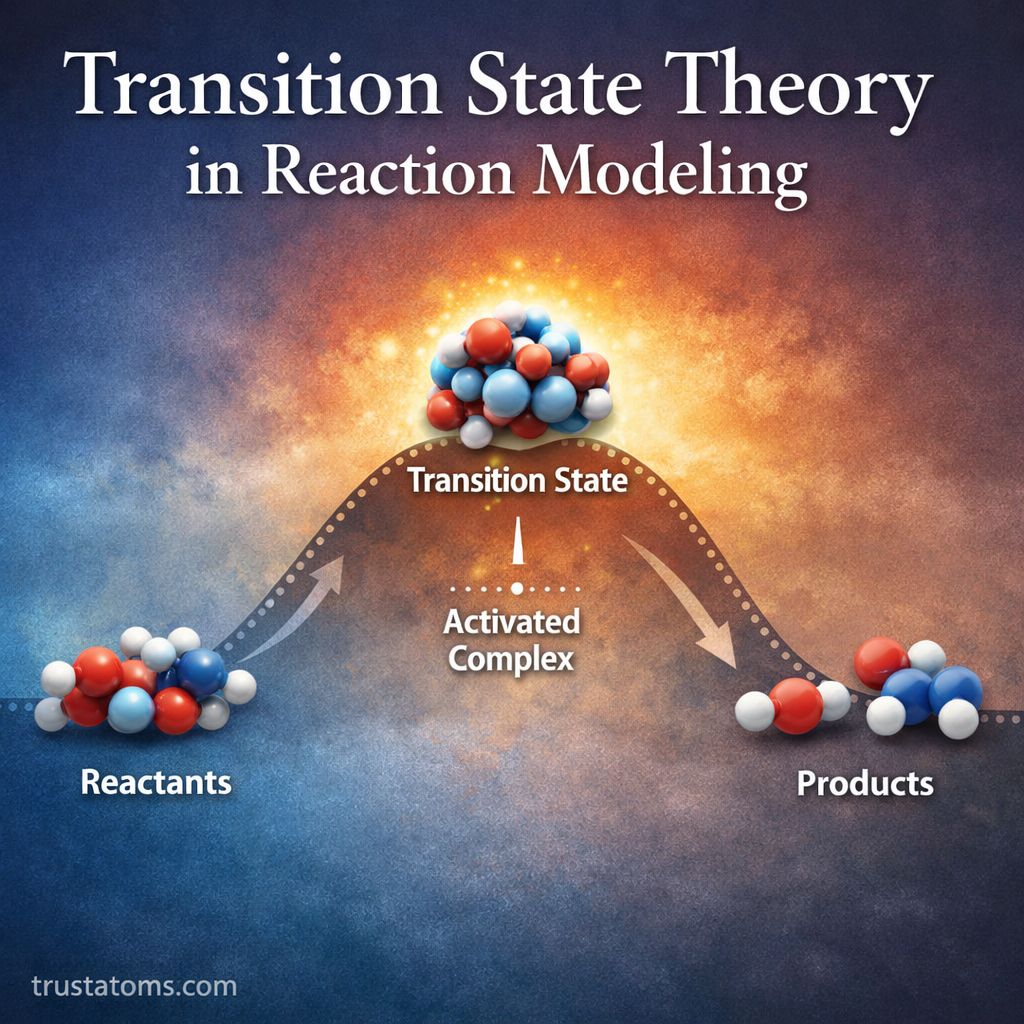
Chemical reactions are rarely instantaneous. Instead, they pass through a series of energetic changes as reactants transform into products. Understanding this process is essential for predicting how fast reactions occur and why certain reactions proceed more easily than others.
Transition State Theory (TST) provides a framework for modeling these processes. Developed in the 1930s, it helps chemists estimate reaction rates by examining the short-lived, high-energy configuration known as the transition state. This theory plays a key role in fields such as chemical kinetics, catalysis, atmospheric chemistry, and enzyme reactions.
Continue reading “Transition State Theory in Reaction Modeling”