
Biophysical chemistry explores how physical principles such as thermodynamics, kinetics, and molecular structure influence chemical behavior in biological systems. One of its central focuses is molecular interaction—how molecules recognize, bind, and influence each other.
These interactions are essential for life. Processes such as enzyme activity, DNA replication, protein folding, and cellular signaling all rely on highly specific molecular interactions. By studying these interactions, scientists gain insight into biological mechanisms and develop technologies used in medicine, biotechnology, and molecular engineering.
What Is Biophysical Chemistry?
Biophysical chemistry is an interdisciplinary field that combines chemistry, biology, and physics to study biological molecules and their behavior.
It investigates how molecules such as:
- Proteins
- DNA and RNA
- Lipids
- Small metabolites
interact and function in biological systems.
The field often focuses on:
- Molecular structure
- Interaction forces
- Energy changes during reactions
- Reaction rates and dynamics
Understanding these factors allows scientists to describe how biological molecules behave at the atomic and molecular level.
The Importance of Molecular Interactions
Molecular interactions determine how biological molecules communicate and perform their functions.
These interactions allow molecules to:
- Recognize specific binding partners
- Form temporary or permanent complexes
- Catalyze chemical reactions
- Transmit biological signals
For example:
- Enzymes bind substrates to catalyze reactions
- Antibodies bind antigens to trigger immune responses
- Hormones bind receptors to regulate physiological processes
Without these interactions, biological systems would not function properly.
Types of Molecular Forces
Molecular interactions are primarily driven by non-covalent forces. While these forces are weaker than covalent bonds, their combined effects can create strong and highly specific interactions.
Hydrogen Bonding
Hydrogen bonds occur when a hydrogen atom bonded to an electronegative atom interacts with another electronegative atom.
These bonds play a key role in:
- DNA base pairing
- Protein structure
- Molecular recognition
Electrostatic Interactions
Electrostatic interactions occur between charged molecules or molecular groups.
These forces influence:
- Protein folding
- Enzyme–substrate interactions
- Molecular stability
Van der Waals Forces
Van der Waals interactions arise from temporary fluctuations in electron distribution between molecules.
Although individually weak, these forces become significant when many atoms interact simultaneously.
Hydrophobic Interactions
Hydrophobic interactions occur when nonpolar molecules cluster together in aqueous environments.
These interactions are essential for:
- Protein folding
- Cell membrane formation
- Molecular self-assembly
Protein–Ligand Interactions
One of the most studied topics in biophysical chemistry is protein–ligand binding.
A ligand is a molecule that binds to a protein at a specific site, often influencing the protein’s activity.
Examples include:
- Drug molecules binding to receptors
- Substrates binding to enzymes
- Signaling molecules binding to cellular receptors
Protein–ligand interactions are governed by factors such as:
- Molecular shape compatibility
- Charge distribution
- Hydrophobic surfaces
- Binding site flexibility
Understanding these interactions is essential for drug discovery and biochemical research.
Thermodynamics of Molecular Binding
Thermodynamics helps explain why molecules interact and form complexes.
When molecules bind, several thermodynamic factors determine whether the interaction is favorable.
Important parameters include:
- Free energy change (ΔG)
- Enthalpy change (ΔH)
- Entropy change (ΔS)
A negative free energy change indicates that a molecular interaction occurs spontaneously.
Thermodynamic analysis helps scientists understand the stability and strength of molecular complexes.
Kinetics of Molecular Interaction
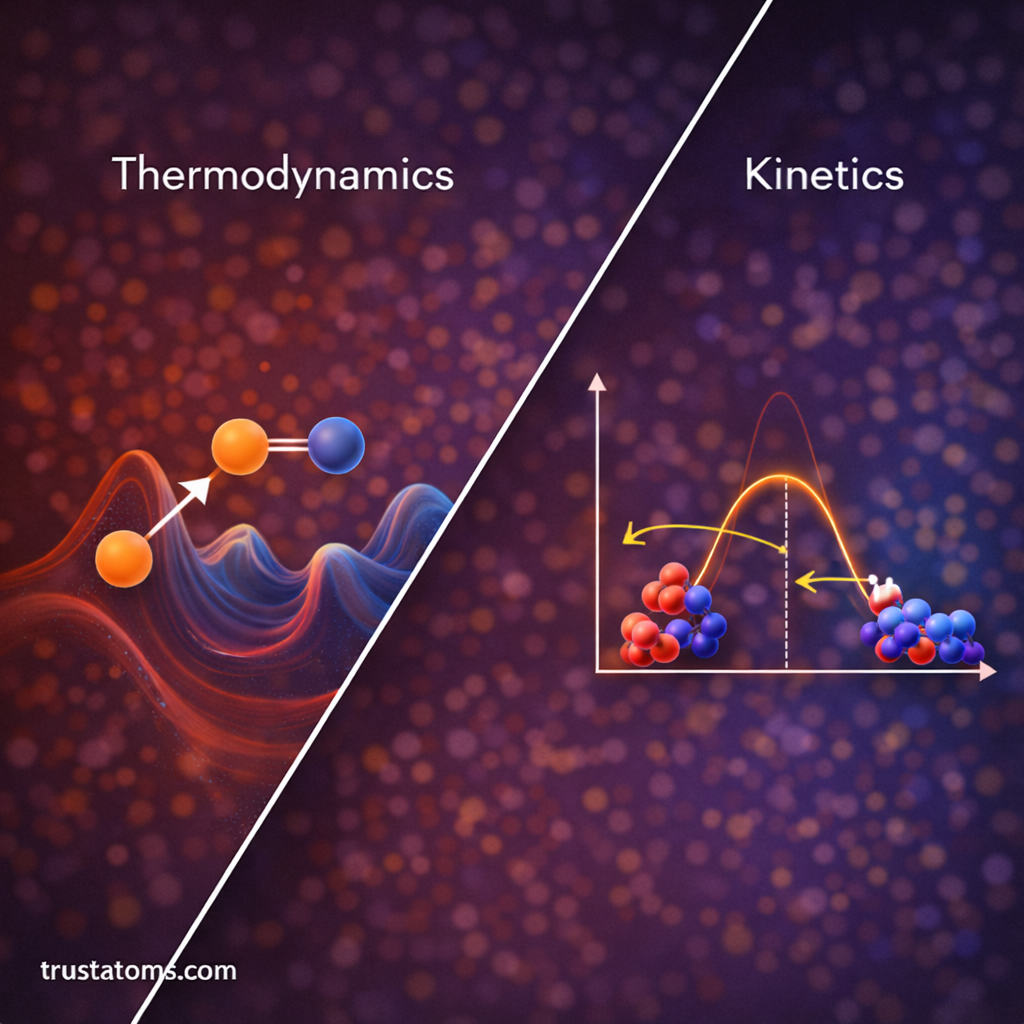
While thermodynamics determines whether a reaction can occur, kinetics determines how quickly it happens.
Binding kinetics describes how rapidly molecules associate and dissociate.
Two important rate constants are:
- Association rate constant – how quickly molecules bind
- Dissociation rate constant – how quickly molecules separate
These rates influence biological processes such as:
- Signal transmission in cells
- Enzyme catalysis
- Drug binding to targets
Studying these rates helps scientists design molecules with improved biological activity.
Techniques Used to Study Molecular Interactions
Biophysical chemistry relies on several advanced experimental techniques to study molecular interactions.
Spectroscopy
Spectroscopic methods measure how molecules interact with light.
Common techniques include:
- Fluorescence spectroscopy
- Infrared spectroscopy
- Nuclear magnetic resonance (NMR)
These methods reveal structural and dynamic information about molecular systems.
X-Ray Crystallography
X-ray crystallography allows scientists to determine the three-dimensional structure of molecules at atomic resolution.
This technique is widely used to study:
- Protein structures
- Protein–ligand complexes
- Molecular binding sites
Cryo-Electron Microscopy
Cryo-electron microscopy provides high-resolution images of large molecular complexes without requiring crystallization.
It has become an important tool for studying complex biological structures.
Surface Plasmon Resonance
Surface plasmon resonance measures binding interactions in real time and provides information about binding strength and kinetics.
Molecular Interaction Networks
In biological systems, molecules rarely act alone. Instead, they form complex networks of interactions that regulate cellular functions.
Examples include:
- Protein–protein interaction networks
- Metabolic pathways
- Gene regulation systems
- Signal transduction pathways
Biophysical chemistry helps researchers understand how these networks operate and respond to environmental changes.
Applications in Medicine and Biotechnology
Understanding molecular interactions has major practical applications.
Drug Discovery
Many drugs work by binding to specific proteins or receptors. Studying these interactions helps scientists design safer and more effective medications.
Protein Engineering
Biophysical chemistry enables the design of proteins with new or improved functions.
Diagnostic Technologies
Many diagnostic tools rely on molecular recognition, such as antibody-based detection methods.
Biotechnology
Biophysical principles are used to develop enzymes, biosensors, and molecular devices used in industry and research.
Challenges in Studying Molecular Interactions
Despite major advances, studying molecular interactions remains complex.
Some challenges include:
- Highly dynamic molecular systems
- Large biological structures
- Environmental influences on interactions
- Computational complexity in modeling interactions
Advances in computational chemistry and imaging technologies are helping overcome these challenges.
The Future of Biophysical Chemistry
Biophysical chemistry continues to expand as new technologies allow scientists to explore molecular systems in greater detail.
Future developments may include:
- Artificial molecular machines
- Advanced computational simulations
- Precision drug design
- Synthetic biological systems
These innovations will deepen our understanding of molecular interactions and enable new applications in medicine and biotechnology.
Conclusion
Biophysical chemistry provides a powerful framework for understanding how molecules interact within biological systems. Through the study of non-covalent forces, thermodynamics, kinetics, and molecular structure, scientists can explain how biological processes operate at the molecular level.
From protein–ligand binding to cellular signaling networks, molecular interactions are essential for life. By exploring these interactions, biophysical chemistry continues to drive advances in medicine, biotechnology, and molecular science.




