
Many chemical reactions occur very slowly under normal conditions. In both natural systems and industrial processes, reactions often need to happen faster or more efficiently. This is where catalysis becomes important.
Catalysis refers to the process in which a substance called a catalyst speeds up a chemical reaction without being permanently consumed. By lowering the energy barrier of a reaction, catalysts help reactions occur more quickly and efficiently while maintaining the same overall chemical outcome.
Understanding catalysis is a key part of reaction dynamics, which studies how chemical reactions proceed and what factors influence their speed and pathways.
What Is Catalysis?
Catalysis is the acceleration of a chemical reaction through the presence of a catalyst. The catalyst participates in the reaction process but is regenerated at the end, meaning it can be used repeatedly.
Key characteristics of catalysts include:
- They increase the rate of reaction
- They are not permanently consumed
- They provide an alternative reaction pathway
- They typically lower the activation energy
Importantly, catalysts do not change the final products of a reaction. They only affect how quickly the reaction occurs.
How Catalysts Influence Reaction Dynamics
In reaction dynamics, catalysts modify the pathway that reactants follow as they transform into products.
Without a catalyst:
- Reactants must overcome a higher energy barrier
- The reaction may proceed slowly or require high temperatures
With a catalyst:
- The reaction follows a lower-energy pathway
- Molecular interactions become more efficient
- Reaction rates increase significantly
By lowering the energy barrier, catalysts make it easier for molecules to reach the transition state needed for the reaction to occur.
Types of Catalysis
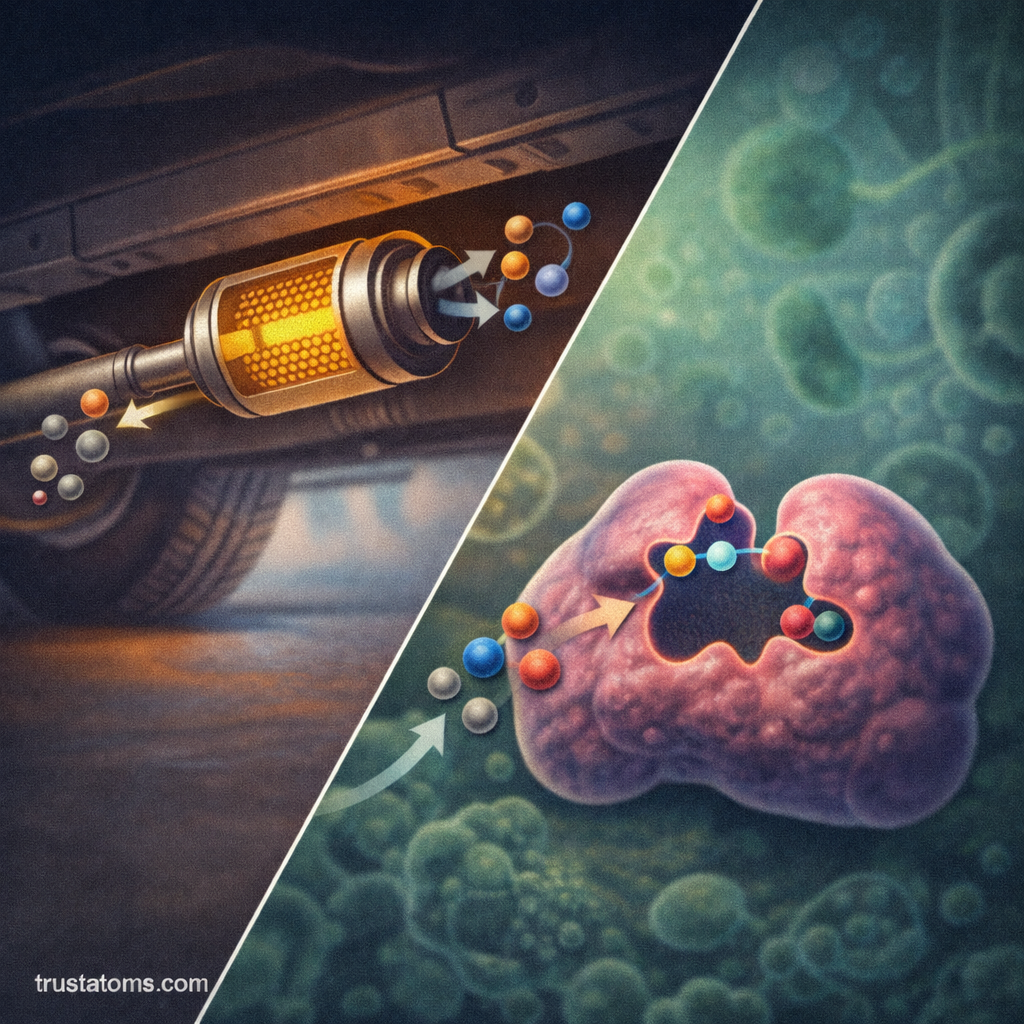
Catalysis can occur in several different forms depending on the physical state of the catalyst and reactants.
Homogeneous Catalysis
In homogeneous catalysis, the catalyst and reactants exist in the same phase, typically a liquid or gas.
Examples include:
- Acid-catalyzed reactions in solution
- Certain reactions in atmospheric chemistry
Advantages of homogeneous catalysis:
- Uniform mixing of reactants and catalyst
- High selectivity in many reactions
However, separating the catalyst from the products can sometimes be challenging.
Heterogeneous Catalysis
In heterogeneous catalysis, the catalyst and reactants exist in different phases.
Most commonly:
- The catalyst is a solid
- Reactants are liids or gases
Reactions occur on the surface of the catalyst, where molecules attach temporarily and react.
Examples include:
- Catalytic converters in automobiles
- Industrial ammonia production
- Petroleum refining
Heterogeneous catalysis is widely used in industrial chemistry because catalysts can be easily separated and reused.
Enzymatic Catalysis
In biological systems, catalysis is carried out by enzymes, which are specialized protein catalysts.
Enzymes:
- Accelerate reactions in living cells
- Work under mild conditions such as body temperature
- Exhibit high specificity for particular reactions
Examples include enzymes involved in:
- Digestion
- Cellular respiration
- DNA replication
Without enzymes, many biochemical reactions would occur far too slowly to sustain life.
The Role of Activation Energy in Catalysis
One of the main reasons catalysts increase reaction speed is because they lower the activation energy.
Activation energy is the minimum energy required for reactants to reach the transition state.
Catalysts lower this barrier by:
- Stabilizing intermediate structures
- Altering the reaction pathway
- Facilitating bond breaking and bond formation
As a result, more molecular collisions lead to successful reactions.
Surface Interactions in Catalytic Reactions
In heterogeneous catalysis, the surface of the catalyst plays a critical role in reaction dynamics.
Typical steps in surface catalysis include:
- Reactant molecules attach to the catalyst surface
- Bonds weaken or rearrange
- New bonds form between atoms
- Product molecules detach from the surface
This process allows reactions to occur more easily than they would in free solution.
The surface area of the catalyst is especially important because more available surface provides more reaction sites.
Factors That Influence Catalytic Efficiency
The effectiveness of a catalyst depends on several factors.
Surface Area
Greater surface area increases the number of active sites where reactions can occur.
This is why many industrial catalysts are designed as:
- Porous materials
- Finely divided solids
- Nanostructured surfaces
Temperature
Higher temperatures can increase reaction rates but may also damage certain catalysts, especially enzymes.
Catalyst Structure
The arrangement of atoms in a catalyst affects how reactants interact with the surface and how easily intermediates form.
Reaction Environment
Pressure, solvents, and chemical conditions can all influence how effectively a catalyst functions.
Real-World Applications of Catalysis
Catalysis is essential across many areas of science, technology, and industry.
Industrial Chemistry
Catalysts are used to improve efficiency in large-scale chemical production, including:
- Fuel processing
- Fertilizer manufacturing
- Plastics production
These processes often rely on catalysts to reduce energy consumption and increase output.
Environmental Protection
Catalysts help reduce harmful emissions and support cleaner technologies.
Examples include:
- Vehicle catalytic converters
- Pollution control systems
- Waste treatment processes
These systems convert harmful chemicals into less harmful substances.
Biological Systems
Enzymes enable living organisms to perform complex chemical reactions rapidly.
Biological catalysis supports processes such as:
- Energy production
- Nutrient breakdown
- Cellular repair
These reactions are essential for maintaining life.
Why Catalysis Is Important in Chemistry
Catalysis provides chemists with powerful tools to control reaction speed and efficiency.
By studying catalytic processes, scientists can:
- Design faster chemical reactions
- Reduce energy requirements
- Improve selectivity toward desired products
- Develop environmentally friendly chemical technologies
Catalysis allows complex chemical transformations to occur under conditions that would otherwise be impractical.
Final Thoughts
Catalysis is a fundamental concept in reaction dynamics that explains how certain substances accelerate chemical reactions without being consumed. By lowering activation energy and providing alternative pathways, catalysts allow reactions to proceed more efficiently.
From industrial manufacturing to biological metabolism, catalytic processes play a central role in shaping chemical behavior across natural and technological systems. Understanding catalysis helps scientists improve chemical processes, develop new materials, and advance sustainable technologies.




