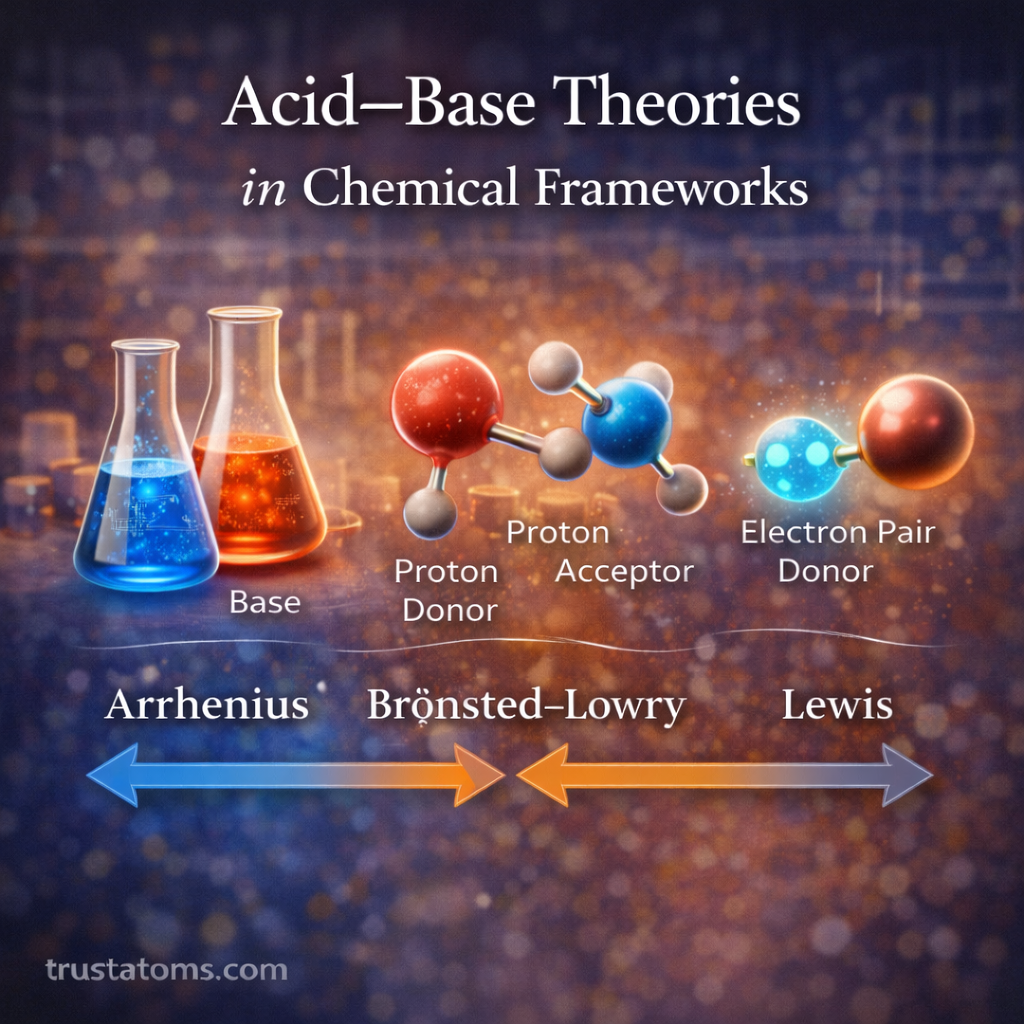
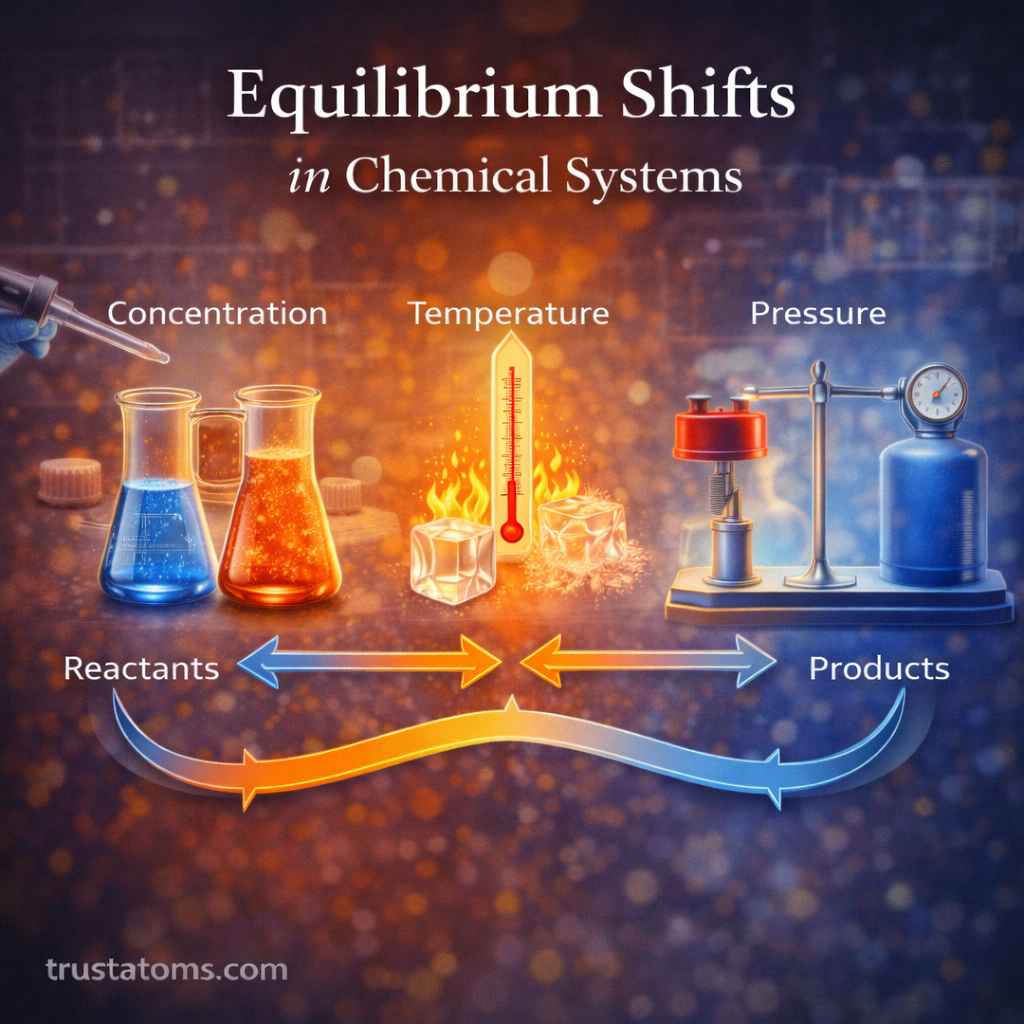
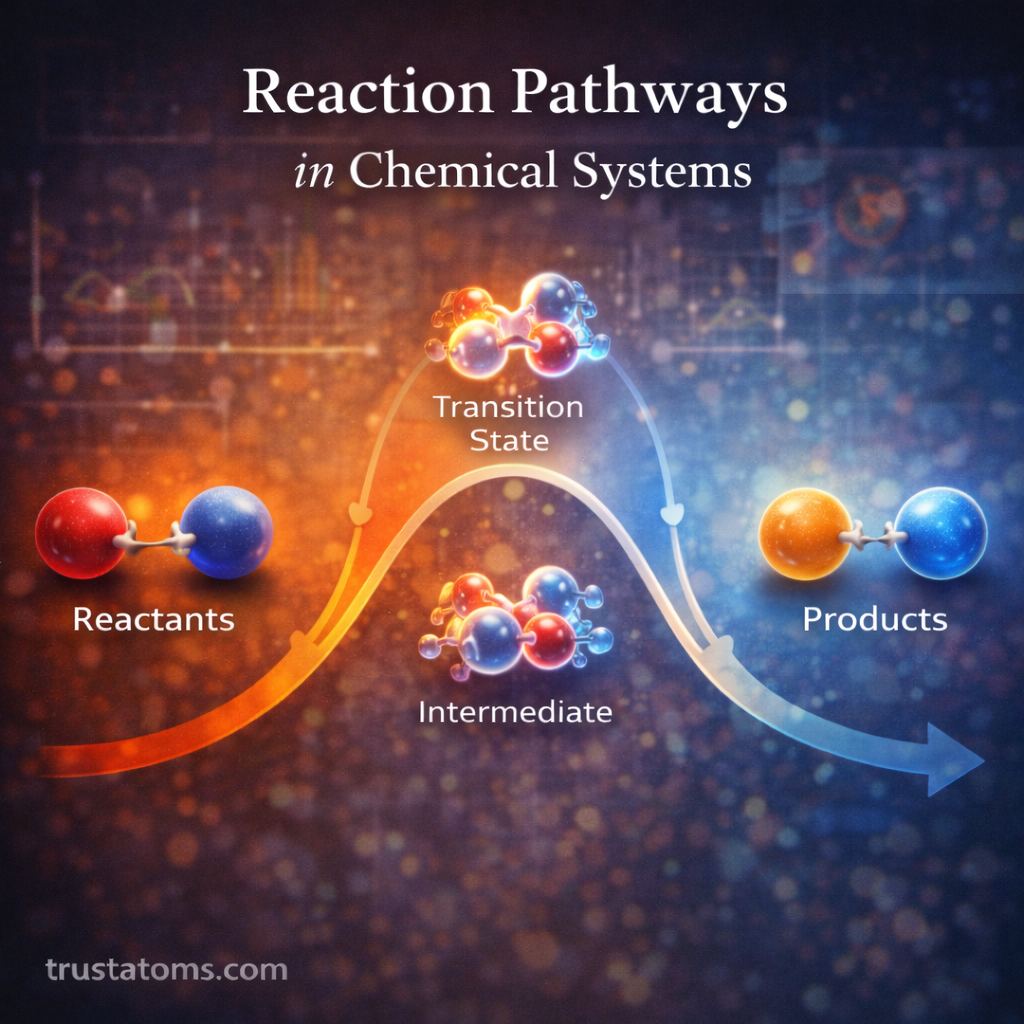
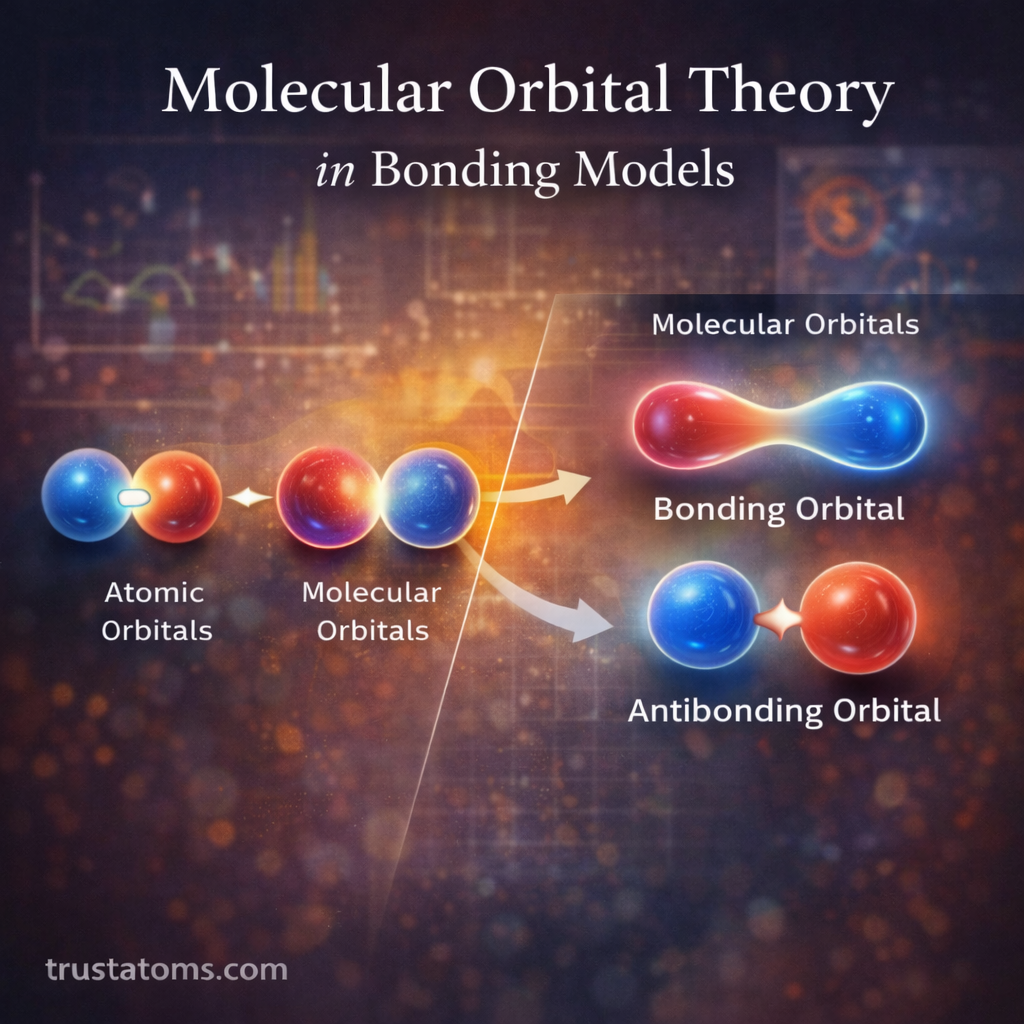
Surface chemistry plays a central role in catalytic reactions. Catalysts accelerate chemical reactions without being consumed, and most catalytic processes occur at the surface of a solid material. Understanding how molecules interact with these surfaces is essential in fields ranging from industrial manufacturing to environmental technology and energy production.
In catalytic systems, the surface of a material provides the active sites where reactant molecules attach, react, and form products. Because reactions occur at the interface between phases—typically solid catalysts and gaseous or liquid reactants—surface chemistry determines how efficient a catalyst can be.
Continue reading “Surface Chemistry in Catalytic Systems”