Chemical reactions do not occur randomly. Some reactions happen naturally without any outside influence, while others require constant energy input. One of the key principles that helps scientists understand why reactions occur is entropy.
Entropy is a fundamental concept in thermodynamics and chemistry that describes the level of disorder or randomness in a system. When combined with energy considerations, entropy helps determine whether a chemical reaction will occur spontaneously.
Understanding entropy and spontaneity allows chemists to predict reaction behavior, explain natural processes, and design chemical systems used in industry, biology, and environmental science.
What Is Entropy?
Entropy is a thermodynamic property that measures the degree of disorder, randomness, or energy distribution in a system.
In simple terms:
- Low entropy = more ordered system
- High entropy = more disordered system
A system naturally tends to move toward greater disorder over time. This tendency is a key idea in thermodynamics and is related to the Second Law of Thermodynamics.
Everyday Examples of Entropy
Entropy is not just a chemistry concept—it appears in everyday situations.
Examples include:
- Ice melting into liquid water
- Perfume spreading through a room
- A tidy desk gradually becoming messy
- Gas expanding to fill a container
In each case, the system moves from a more ordered state to a more disordered state, increasing entropy.
The Second Law of Thermodynamics
The Second Law of Thermodynamics states:
The total entropy of an isolated system always increases over time.
This law explains why certain processes occur naturally while others do not.
For example:
- Heat naturally flows from hot objects to cold ones.
- Gases expand to occupy available space.
- Chemical reactions often proceed in directions that increase overall disorder.
The second law does not mean entropy always increases in a single part of a system. Instead, the total entropy of the system and its surroundings must increase.
What Is a Spontaneous Reaction?
A spontaneous reaction is a chemical reaction that occurs naturally under a given set of conditions without continuous external energy input.
Important clarification:
- Spontaneous does not mean fast.
- Some spontaneous reactions occur very slowly.
Examples:
- Rust forming on iron
- Ice melting at room temperature
- Hydrogen and oxygen reacting to form water
A reaction may be spontaneous even if it requires a small initial energy input to begin.
Relationship Between Entropy and Spontaneity
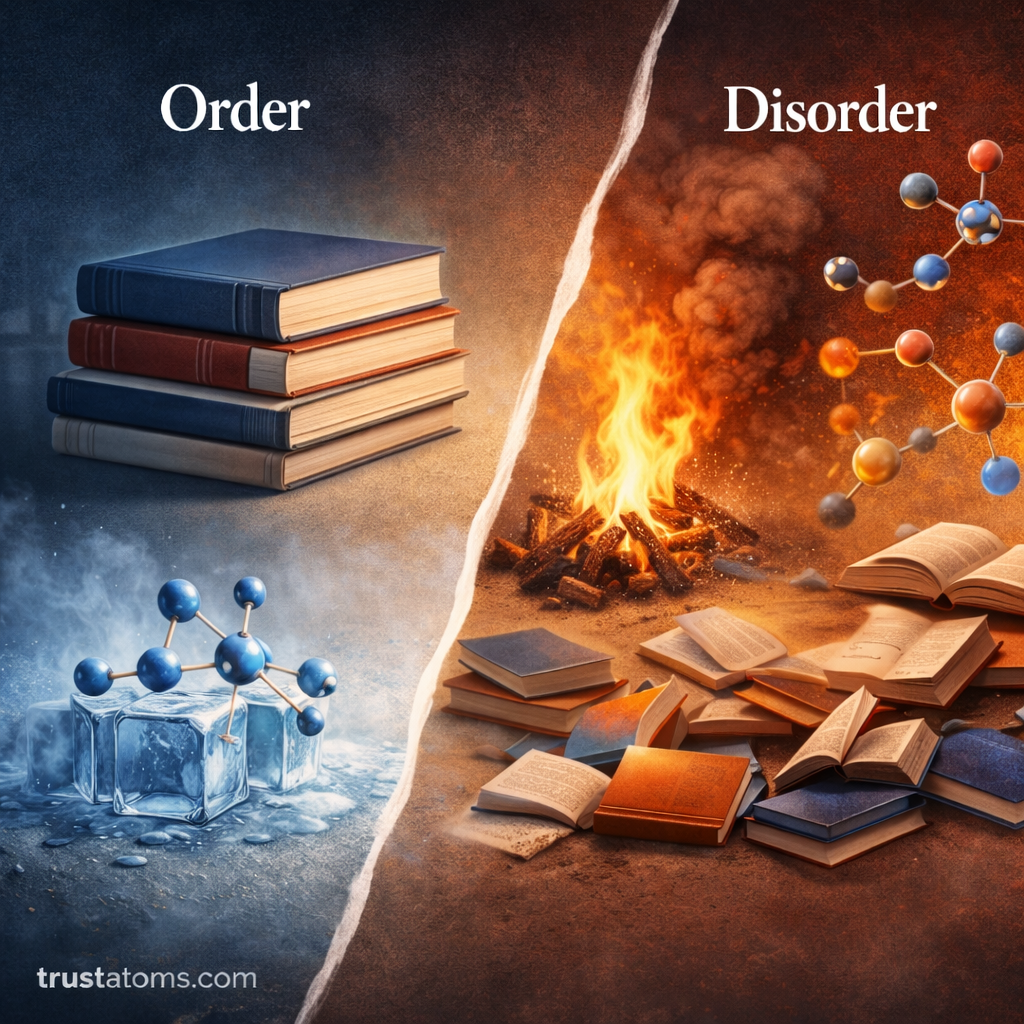
Entropy plays a major role in determining whether a reaction will occur spontaneously.
A reaction tends to be spontaneous when:
- Entropy increases
- Energy becomes more evenly distributed
- The system moves toward greater randomness
However, entropy alone does not determine spontaneity. Chemists must also consider enthalpy, which represents the heat absorbed or released during a reaction.
The combined effect of enthalpy and entropy determines reaction spontaneity.
Entropy Changes in Chemical Reactions
During a chemical reaction, entropy can either increase or decrease depending on the changes occurring in the system.
Situations That Increase Entropy
Entropy usually increases when:
- A solid becomes a liquid
- A liquid becomes a gas
- The number of gas molecules increases
- Substances dissolve in water
- Particles become more dispersed
Example:
A solid dissolving in water increases entropy because particles spread out and become more randomly arranged.
Situations That Decrease Entropy
Entropy decreases when:
- Gas becomes liquid
- Liquid becomes solid
- Molecules combine into fewer particles
- Systems become more structured
Example:
When water freezes into ice, molecules become more ordered, resulting in lower entropy.
Predicting Entropy Changes
Chemists often predict entropy changes by observing the physical states and number of particles in a reaction.
General guidelines include:
- Reactions producing gases tend to increase entropy.
- Reactions forming solids often decrease entropy.
- Reactions producing more particles usually increase entropy.
- Reactions producing fewer particles usually decrease entropy.
These rules provide useful predictions, but exact entropy values are determined experimentally.
The Role of Gibbs Free Energy
To determine whether a reaction is spontaneous, chemists use a thermodynamic quantity called Gibbs free energy.
Gibbs free energy combines enthalpy and entropy effects into a single value that predicts reaction spontaneity.
A reaction is considered:
- Spontaneous if Gibbs free energy is negative
- Non-spontaneous if Gibbs free energy is positive
- At equilibrium if Gibbs free energy equals zero
This relationship allows scientists to evaluate how temperature, energy changes, and entropy influence reaction direction.
Temperature and Spontaneity
Temperature can strongly influence whether a reaction is spontaneous.
Some reactions become spontaneous only at certain temperatures.
Examples include:
- Ice melting only above 0°C
- Certain chemical reactions occurring only when heated
- Some processes becoming unfavorable at very high temperatures
Temperature affects both energy distribution and entropy, which together determine spontaneity.
Entropy in Biological and Environmental Systems
Entropy plays a crucial role in many natural processes beyond laboratory chemistry.
Biological Systems
Living organisms constantly manage entropy by:
- Consuming energy
- Breaking down nutrients
- Maintaining organized structures
Although organisms maintain internal order, they increase entropy in their surroundings through energy release.
Environmental Processes
Entropy helps explain processes such as:
- Heat flow in Earth’s atmosphere
- Mixing of gases in the air
- Ocean circulation
- Energy transfer in ecosystems
These processes contribute to the natural balance of energy and matter on Earth.
Common Misconceptions About Entropy
Entropy is often misunderstood, especially when described simply as “disorder.”
Some clarifications include:
- Entropy refers to energy distribution, not just visible disorder.
- Spontaneous reactions can still require activation energy.
- Not all spontaneous reactions occur quickly.
- Entropy applies to the combined system and surroundings, not just the reaction itself.
Understanding these points helps avoid common mistakes when analyzing chemical processes.
Why Entropy Matters in Chemistry
Entropy is essential for predicting and understanding chemical behavior.
It helps scientists:
- Determine whether reactions occur naturally
- Understand energy flow in chemical systems
- Design industrial chemical processes
- Study biological metabolism
- Model environmental and atmospheric reactions
Together with enthalpy and Gibbs free energy, entropy forms the foundation of chemical thermodynamics.
Final Thoughts
Entropy provides a powerful framework for understanding why chemical reactions occur. By describing how energy and particles distribute within a system, entropy reveals the natural tendency of systems to move toward greater randomness and energy dispersal.
When combined with energy changes and temperature effects, entropy helps chemists predict whether reactions will occur spontaneously. This concept is fundamental to fields ranging from chemical engineering and biology to environmental science and physics.
Understanding entropy and spontaneity allows scientists to better explain the behavior of matter and energy throughout the natural world.