Enzyme catalysis is one of the most important processes in biochemistry. Enzymes allow chemical reactions inside living cells to occur quickly and efficiently, often accelerating reactions that would otherwise take years or even centuries under normal conditions.
These biological catalysts are responsible for countless processes necessary for life, including metabolism, DNA replication, digestion, and cellular energy production. Without enzymes, most biochemical reactions would occur far too slowly to sustain living organisms.
Understanding enzyme catalysis helps scientists explain how biological systems function at the molecular level and has important applications in medicine, biotechnology, and industrial chemistry.
What Are Enzymes?
Enzymes are specialized proteins that act as catalysts, meaning they speed up chemical reactions without being consumed in the process.
A catalyst works by lowering the activation energy required for a reaction to occur. Activation energy is the minimum energy needed for molecules to react and form new products.
Enzymes increase reaction rates by:
- Stabilizing reaction intermediates
- Properly orienting reactant molecules
- Creating favorable chemical environments
- Reducing the energy barrier of reactions
Because of this, enzymes can increase reaction speeds by millions or even billions of times compared to uncatalyzed reactions.
Key Components of Enzyme Catalysis
Several important components define how enzymes function in biochemical reactions.
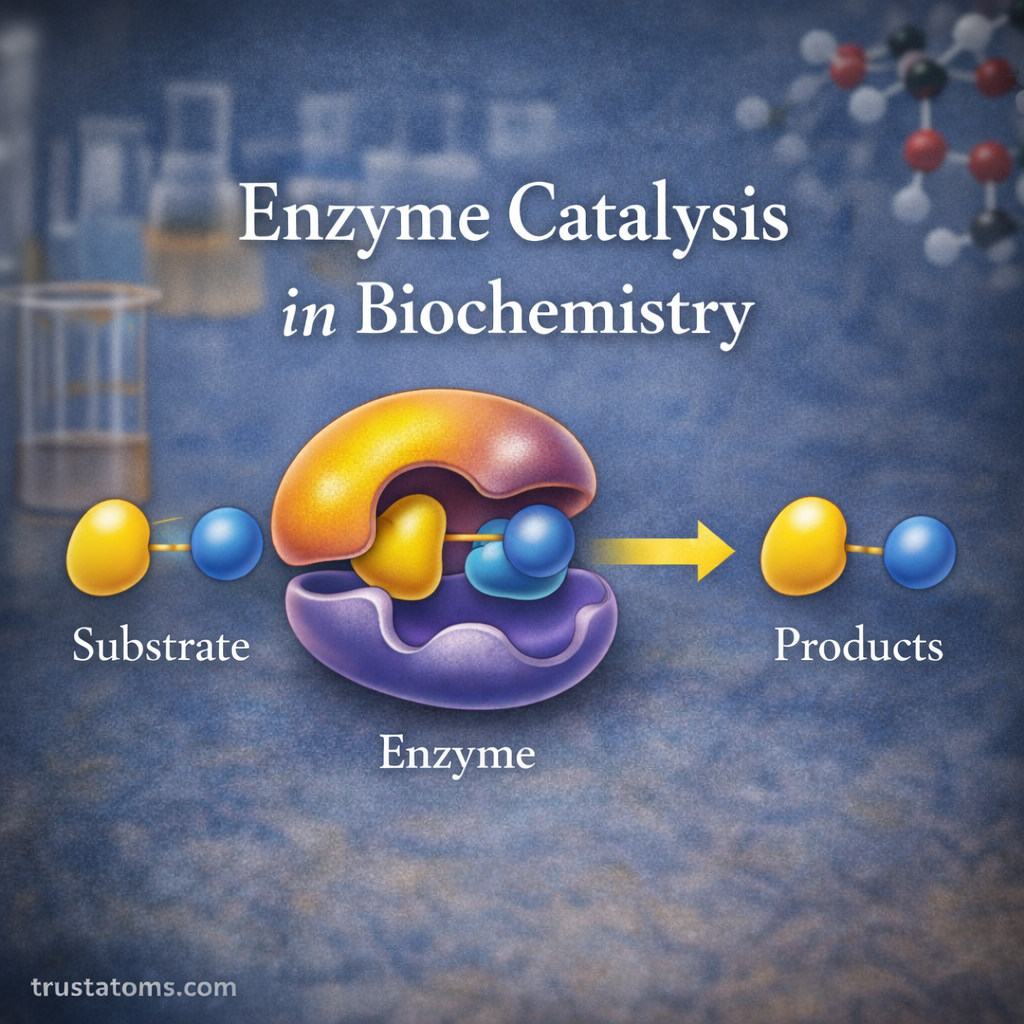
Substrate
The substrate is the molecule upon which an enzyme acts.
During enzyme catalysis:
- The substrate binds to the enzyme.
- The enzyme converts the substrate into products.
- The products are released from the enzyme.
The enzyme itself remains unchanged and can participate in additional reactions.
Active Site
The active site is a specialized region of the enzyme where the substrate binds.
Characteristics of the active site include:
- A specific three-dimensional shape
- Chemical groups that interact with the substrate
- A microenvironment optimized for catalysis
The shape and chemical properties of the active site determine the enzyme’s specificity, meaning each enzyme typically catalyzes only one type of reaction.
The Enzyme–Substrate Complex
When a substrate binds to an enzyme, they form a temporary structure called the enzyme–substrate complex.
This complex allows the enzyme to stabilize the transition state of the reaction, making it easier for chemical bonds to break and new ones to form.
The catalytic process generally follows these steps:
- Substrate molecules bind to the enzyme’s active site
- An enzyme–substrate complex forms
- Chemical transformation occurs
- Products are released
- The enzyme returns to its original state
This process enables enzymes to catalyze reactions repeatedly.
Models of Enzyme Binding
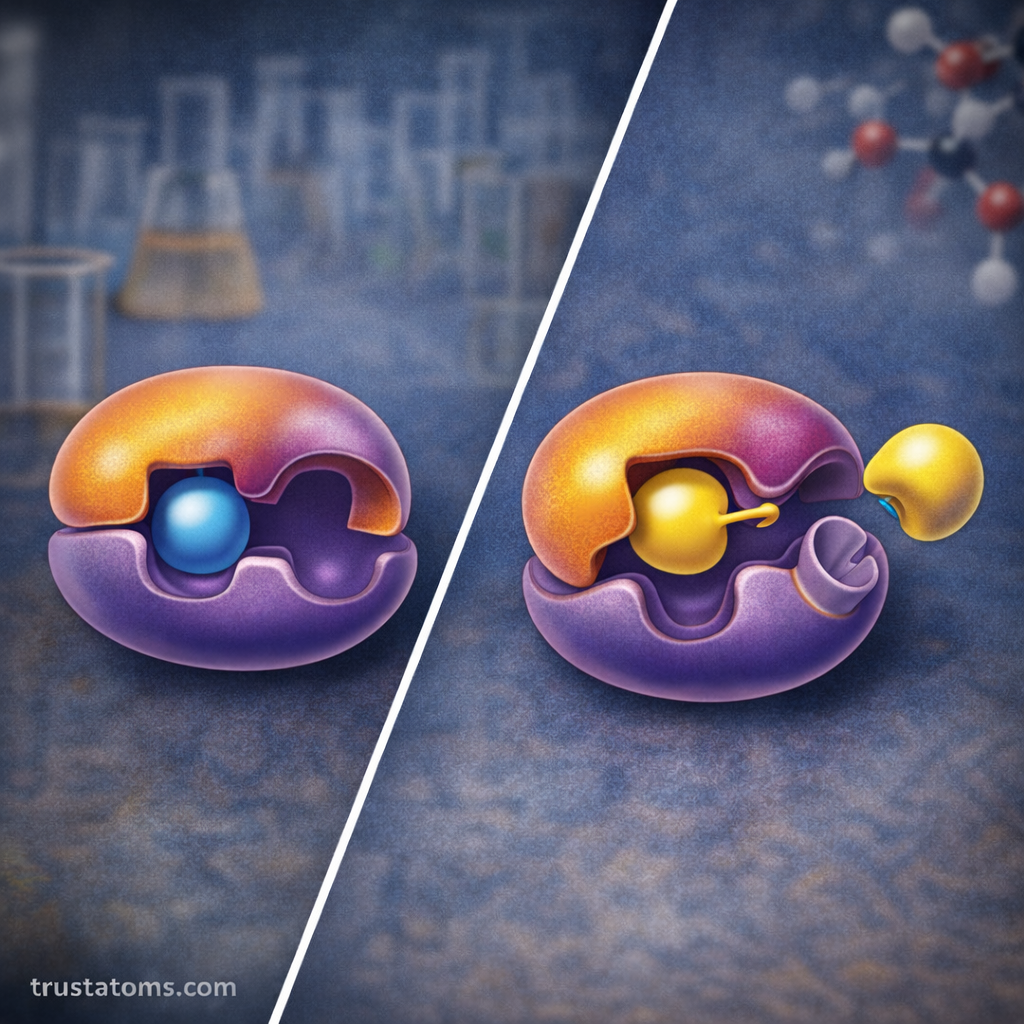
Scientists use two primary models to explain how enzymes interact with substrates.
Lock-and-Key Model
The lock-and-key model suggests that the enzyme’s active site has a fixed shape that perfectly fits the substrate.
Key characteristics:
- Highly specific binding
- Substrate fits precisely into the active site
- Little structural change occurs during binding
This model explains enzyme specificity but does not fully describe the flexibility observed in many enzymes.
Induced Fit Model
The induced fit model proposes that the enzyme changes shape slightly when the substrate binds.
Important aspects include:
- Flexible enzyme structure
- Active site adjusts to accommodate the substrate
- Improved catalytic efficiency
This model better reflects how many enzymes behave in biological systems.
Types of Enzyme Catalysis Mechanisms
Enzymes use several catalytic strategies to accelerate biochemical reactions.
Acid–Base Catalysis
Some enzymes donate or accept protons during reactions.
This process helps stabilize reaction intermediates and promotes bond rearrangements.
Covalent Catalysis
In covalent catalysis, the enzyme temporarily forms a covalent bond with the substrate.
This intermediate step lowers the activation energy and allows the reaction to proceed more efficiently.
Metal Ion Catalysis
Certain enzymes use metal ions to assist in catalysis.
Metal ions may:
- Stabilize charged molecules
- Facilitate electron transfer
- Help bind substrates
Examples include enzymes that rely on zinc, magnesium, or iron ions.
Factors Affecting Enzyme Activity
Several environmental conditions influence enzyme function and catalytic efficiency.
Temperature
Temperature affects the rate of molecular movement and enzyme stability.
General trends include:
- Reaction rates increase as temperature rises
- Excessive heat can denature enzymes
- Each enzyme has an optimal temperature range
pH Levels
Enzymes function best within specific pH ranges.
Changes in pH can alter the charge of amino acids in the active site, affecting substrate binding and catalytic activity.
For example:
- Pepsin works best in acidic conditions
- Trypsin functions optimally in slightly alkaline environments
Substrate Concentration
The rate of enzyme-catalyzed reactions increases with higher substrate concentration until the enzyme becomes saturated.
Once all active sites are occupied, the reaction rate reaches its maximum velocity.
Enzyme Concentration
Increasing the amount of enzyme in a system generally increases the reaction rate, provided enough substrate is available.
More enzyme molecules mean more active sites available for catalysis.
Enzyme Inhibition
Enzyme activity can also be regulated or reduced by molecules known as inhibitors.
Inhibitors control metabolic pathways and help maintain cellular balance.
Competitive Inhibition
In competitive inhibition:
- The inhibitor resembles the substrate
- It competes for binding at the active site
- Increasing substrate concentration can overcome inhibition
Noncompetitive Inhibition
In noncompetitive inhibition:
- The inhibitor binds at a different site on the enzyme
- The enzyme’s shape or function is altered
- Increasing substrate concentration does not restore full activity
Enzyme Catalysis in Biological Systems
Enzyme catalysis supports nearly every metabolic process in living organisms.
Examples include:
Cellular Respiration
Enzymes drive the series of reactions that convert nutrients into usable energy in the form of ATP.
DNA Replication
Specialized enzymes help copy genetic material accurately during cell division.
Digestion
Digestive enzymes break down food molecules into smaller components that the body can absorb.
Metabolic Regulation
Enzymes control pathways that produce or break down essential biological molecules.
Applications of Enzyme Catalysis
Enzyme catalysis is widely used outside of natural biological systems as well.
Important applications include:
- Pharmaceutical drug development
- Food processing and fermentation
- Biofuel production
- Biotechnology and genetic engineering
- Industrial chemical manufacturing
Because enzymes are highly efficient and environmentally friendly catalysts, they are valuable tools in sustainable technologies.
Final Thoughts
Enzyme catalysis is central to the chemistry of life. By lowering activation energy and stabilizing reaction intermediates, enzymes allow biochemical reactions to occur rapidly and efficiently within living systems.
The interaction between enzymes and substrates, the mechanisms of catalysis, and the factors affecting enzyme activity all contribute to the complex network of reactions that sustain life.
As research continues to expand our understanding of enzymes, scientists are discovering new ways to harness enzyme catalysis in medicine, biotechnology, and industrial chemistry.




