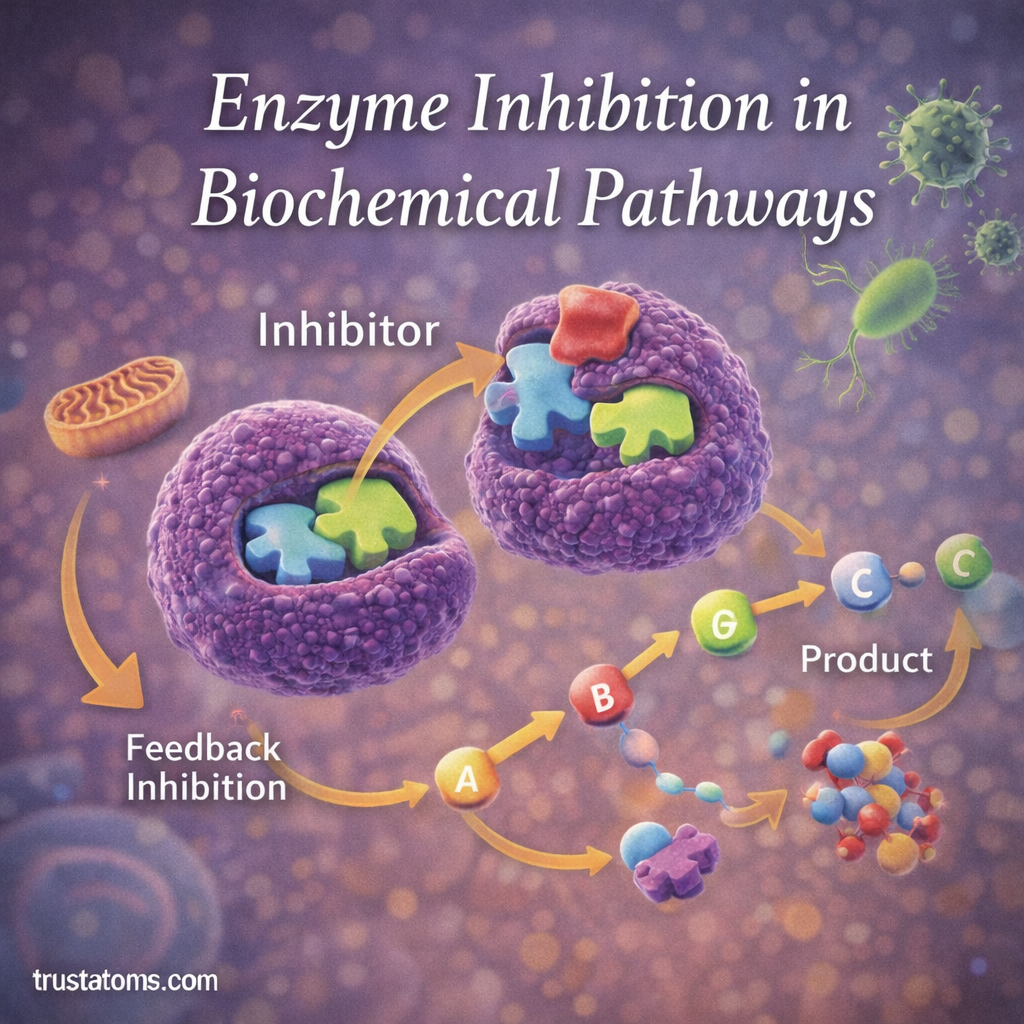
Enzyme inhibition is a biological process in which molecules reduce or stop the activity of enzymes. Because enzymes control many chemical reactions in living organisms, inhibiting their activity can significantly affect metabolic pathways and cellular processes.
In biochemical pathways, enzyme inhibition helps regulate metabolism, maintain cellular balance, and control the speed of chemical reactions. It also plays an important role in medicine, where many drugs work by inhibiting specific enzymes linked to disease.
Understanding enzyme inhibition helps scientists explain how metabolic pathways are controlled and how biological systems maintain stability.
What Is Enzyme Inhibition?
Enzyme inhibition occurs when a molecule called an inhibitor interferes with the normal activity of an enzyme.
This interference may:
- Prevent the substrate from binding to the enzyme
- Alter the shape of the enzyme’s active site
- Reduce the enzyme’s catalytic efficiency
- Completely block the reaction
Inhibitors can occur naturally in cells or be introduced from outside sources such as medications or toxins.
The Role of Enzyme Inhibition in Metabolic Pathways
Biochemical pathways consist of a series of enzyme-controlled reactions that convert molecules into different products.
Enzyme inhibition helps regulate these pathways by:
- Controlling reaction speed
- Preventing overproduction of molecules
- Balancing metabolic processes
- Responding to environmental changes
Without enzyme inhibition, many metabolic reactions would continue unchecked, potentially disrupting cellular function.
Types of Enzyme Inhibition
Scientists classify enzyme inhibition into several categories depending on how the inhibitor interacts with the enzyme.
Competitive Inhibition
In competitive inhibition, the inhibitor competes with the substrate for access to the enzyme’s active site.
Key characteristics include:
- The inhibitor resembles the substrate’s structure
- Both molecules compete for the same binding location
- Increasing substrate concentration can overcome inhibition
Because the inhibitor blocks the active site, the enzyme cannot bind the substrate until the inhibitor leaves.
Common examples of competitive inhibitors include drugs that mimic natural substrates.
Noncompetitive Inhibition
In noncompetitive inhibition, the inhibitor binds to a location other than the active site.
This site is often called an allosteric site.
Important features include:
- The inhibitor changes the enzyme’s shape
- The active site becomes less effective
- Increasing substrate concentration does not restore enzyme activity
Noncompetitive inhibition reduces enzyme efficiency regardless of how much substrate is available.
Uncompetitive Inhibition
Uncompetitive inhibition occurs when the inhibitor binds only to the enzyme–substrate complex.
In this case:
- The inhibitor attaches after the substrate binds
- The enzyme–substrate complex becomes inactive
- Both reaction rate and enzyme efficiency decrease
Although less common than other forms, uncompetitive inhibition plays an important role in certain biochemical pathways.
Reversible vs Irreversible Inhibition
Enzyme inhibition can also be categorized based on whether the inhibitor can detach from the enzyme.
Reversible Inhibition
Reversible inhibitors bind temporarily to enzymes.
Characteristics include:
- The inhibitor can detach from the enzyme
- Normal enzyme function may resume
- Many metabolic regulators act through reversible inhibition
Reversible inhibition allows cells to control metabolic pathways dynamically.
Irreversible Inhibition
Irreversible inhibitors permanently disable enzyme function.
They often form strong chemical bonds with the enzyme.
Examples include:
- Certain toxins
- Heavy metals
- Some pharmaceutical drugs
Once the enzyme is permanently altered, the cell must produce new enzymes to restore activity.
Feedback Inhibition in Metabolic Pathways
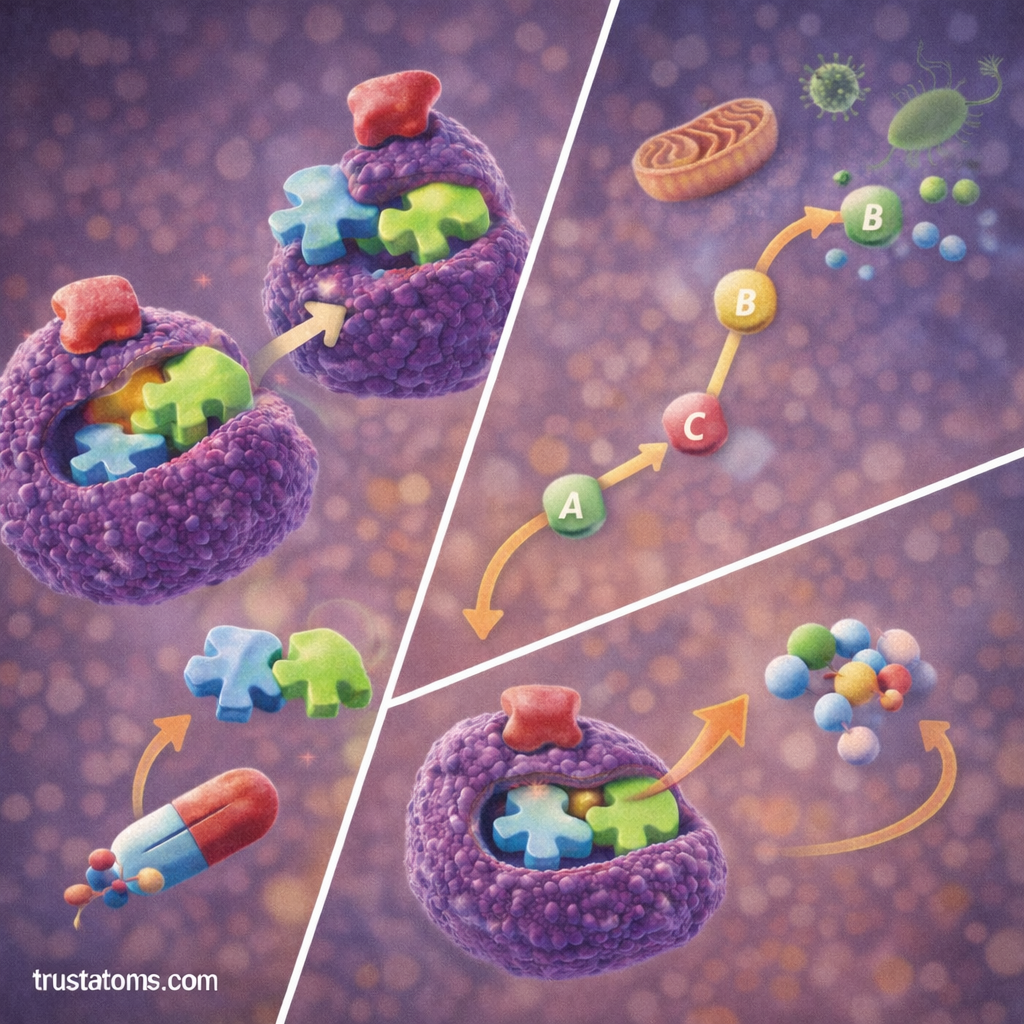
One of the most important regulatory mechanisms in biology is feedback inhibition.
Feedback inhibition occurs when the final product of a metabolic pathway inhibits an earlier enzyme in the pathway.
This mechanism allows cells to control product levels efficiently.
How Feedback Inhibition Works
- A metabolic pathway produces a final product.
- When enough product accumulates, it binds to an enzyme earlier in the pathway.
- The enzyme’s activity decreases.
- The pathway slows down or stops.
When product levels drop, the inhibition is removed and the pathway resumes.
This process prevents wasteful energy use and maintains metabolic balance.
Enzyme Inhibition in Medicine
Many medications work by inhibiting enzymes associated with disease.
Examples include:
Antibiotics
Certain antibiotics inhibit bacterial enzymes required for cell wall synthesis or protein production.
By blocking these enzymes, antibiotics prevent bacteria from growing and reproducing.
Antiviral Drugs
Antiviral medications often inhibit viral enzymes needed for replication.
Examples include drugs that target enzymes involved in:
- Viral genome replication
- Viral protein processing
- Virus assembly
Cancer Treatments
Some cancer therapies inhibit enzymes involved in uncontrolled cell growth.
These drugs target enzymes that regulate:
- DNA replication
- Cell division
- Signal transduction pathways
By blocking these enzymes, cancer drugs can slow tumor growth.
Enzyme Inhibition in Toxicology
Enzyme inhibition also explains how certain toxins affect the body.
For example:
- Some pesticides inhibit enzymes involved in nerve signaling
- Heavy metals may bind to enzyme active sites and block activity
- Certain poisons disrupt metabolic enzymes
These effects can interfere with essential biological processes and lead to harmful physiological outcomes.
Why Enzyme Inhibition Is Important in Biology
Enzyme inhibition is essential for maintaining proper metabolic regulation.
Key roles include:
- Controlling metabolic pathway activity
- Preventing excessive production of molecules
- Regulating energy use
- Protecting cells from harmful chemical reactions
- Supporting cellular homeostasis
Through inhibition, biological systems can adjust reaction rates to meet changing physiological needs.
Final Thoughts
Enzyme inhibition plays a critical role in regulating biochemical pathways and maintaining balance within living organisms. By slowing or stopping enzyme activity, cells can control metabolic reactions, conserve energy, and respond to environmental changes.
Beyond its biological importance, enzyme inhibition is also a powerful tool in medicine, pharmacology, and biotechnology. Studying how inhibitors interact with enzymes helps scientists develop new drugs, understand metabolic diseases, and uncover the complex mechanisms that govern life at the molecular level.