Chemical reactions often move toward a state where reactants and products exist together in balanced proportions. This state is known as chemical equilibrium. At equilibrium, reactions do not stop, but the forward and reverse reactions occur at equal rates.
However, equilibrium conditions can change when external factors influence the system. When this happens, the reaction adjusts to restore balance. These adjustments are called equilibrium shifts.
Understanding equilibrium shifts is essential in chemistry because it allows scientists and engineers to predict how reactions respond to changes in temperature, pressure, and concentration. This knowledge is widely used in industrial chemistry, environmental science, and biological systems.
What Is Chemical Equilibrium?
Chemical equilibrium occurs when a reversible reaction reaches a state where the rate of the forward reaction equals the rate of the reverse reaction.
At this point:
- Reactants and products are present in constant concentrations
- The reaction continues in both directions
- No net change in composition occurs over time
Equilibrium does not mean the reaction has stopped. Instead, it means the reaction has reached a dynamic balance.
Many important chemical processes operate under equilibrium conditions, including atmospheric reactions and biological metabolic pathways.
Understanding Equilibrium Shifts
An equilibrium shift occurs when a system at equilibrium responds to a disturbance by adjusting the concentrations of reactants and products.
The system shifts in a direction that helps reduce the effect of the disturbance.
This behavior is described by Le Châtelier’s Principle, which states:
When a system at equilibrium is disturbed, it shifts in the direction that counteracts the disturbance.
This principle helps predict how chemical systems respond to external changes.
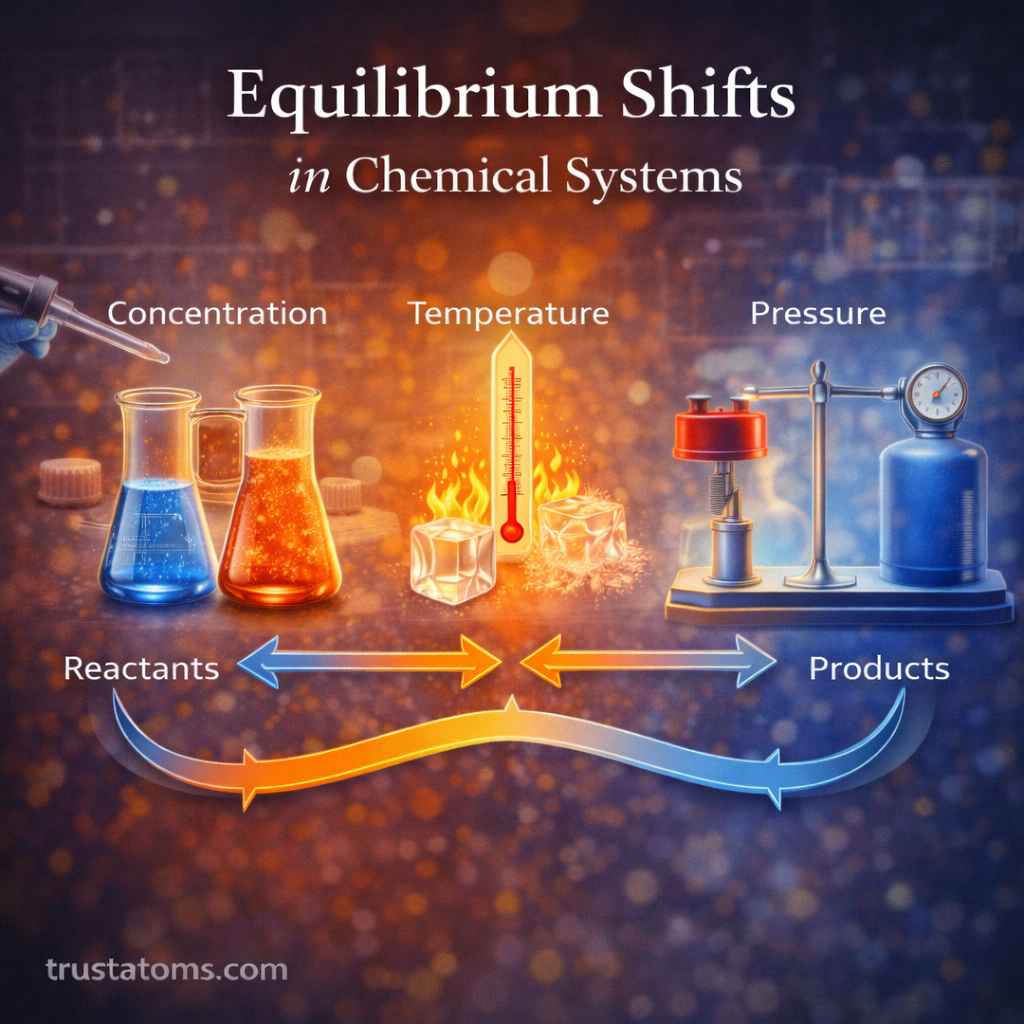
Factors That Cause Equilibrium Shifts
Several external factors can disturb equilibrium and cause a shift in reaction balance.
Changes in Concentration
When the concentration of reactants or products changes, the system adjusts to restore equilibrium.
Examples:
- Adding more reactants typically shifts equilibrium toward product formation.
- Removing products can also push the reaction toward producing more products.
- Increasing product concentration may shift the reaction back toward reactants.
These adjustments help restore balance within the system.
Changes in Temperature
Temperature changes influence reaction equilibrium because they affect the energy of the system.
Reactions can be categorized as:
- Exothermic reactions – release heat
- Endothermic reactions – absorb heat
Temperature changes affect these reactions differently:
- Increasing temperature favors the endothermic direction
- Decreasing temperature favors the exothermic direction
This occurs because the system shifts to absorb or release energy in response to temperature changes.
Changes in Pressure (for Gases)
Pressure changes mainly affect reactions involving gases.
When pressure increases, the equilibrium shifts toward the side with fewer gas molecules.
When pressure decreases, equilibrium shifts toward the side with more gas molecules.
This adjustment helps reduce the pressure change in the system.
Pressure effects are especially important in industrial reactions involving gases.
Changes in Volume
Changing the volume of a reaction container affects pressure in gas-phase reactions.
For example:
- Decreasing volume increases pressure
- Increasing volume decreases pressure
As a result, volume changes indirectly influence equilibrium by altering pressure conditions.
The Role of Catalysts
Catalysts are substances that speed up chemical reactions without being permanently consumed.
However, catalysts do not shift the equilibrium position.
Instead, catalysts:
- Increase the rate of both forward and reverse reactions
- Help the system reach equilibrium faster
- Do not change the equilibrium concentrations of reactants and products
This distinction is important when designing industrial chemical processes.
Equilibrium Constants and Reaction Balance
Chemical equilibrium can be described quantitatively using the equilibrium constant.
The equilibrium constant expresses the ratio of product concentrations to reactant concentrations at equilibrium.
Important features of equilibrium constants include:
- They are specific to a particular reaction
- They depend on temperature
- They indicate the position of equilibrium
Interpretation of equilibrium constant values:
- Large values indicate product-favored reactions
- Small values indicate reactant-favored reactions
Equilibrium constants help chemists predict the direction and extent of chemical reactions.
Industrial Applications of Equilibrium Shifts
Understanding equilibrium shifts is critical in industrial chemistry, where maximizing product yield is often a primary goal.
Many large-scale chemical processes rely on careful control of reaction conditions.
Examples include:
Ammonia Production
The Haber process produces ammonia from nitrogen and hydrogen.
Engineers adjust:
- Pressure
- Temperature
- Catalyst use
to shift equilibrium toward ammonia production.
Sulfuric Acid Production
The Contact process relies on equilibrium control to efficiently produce sulfur trioxide.
Careful temperature management improves reaction yield.
Environmental Chemistry
Equilibrium shifts help explain processes such as:
- Ocean acidification
- Atmospheric chemical balance
- Carbon dioxide absorption in water
These examples highlight how equilibrium principles influence both industrial and environmental systems.
Why Understanding Equilibrium Shifts Matters
Studying equilibrium shifts allows chemists to predict how chemical systems respond to changing conditions.
Important benefits include:
- Improving industrial chemical production
- Optimizing reaction efficiency
- Understanding biological chemical processes
- Predicting environmental chemical behavior
By applying equilibrium principles, scientists can control reactions and design systems that operate more effectively.
Final Thoughts
Equilibrium shifts play a central role in chemical systems. Although equilibrium represents a balanced state, changes in concentration, temperature, pressure, or volume can disturb that balance.
Through Le Châtelier’s Principle, chemical systems respond by shifting in a direction that counteracts these disturbances. This adaptive behavior allows chemists to predict reaction outcomes and control reaction conditions in both laboratory and industrial environments.
Understanding equilibrium shifts provides deeper insight into how chemical reactions behave and how they can be manipulated to achieve desired results.




