
Understanding how energy changes during chemical reactions is a central part of thermochemistry. One of the most important principles used to analyze these energy changes is Hess’s Law. This law allows scientists to calculate the heat change of a reaction even when it cannot be measured directly.
Hess’s Law plays a critical role in thermochemical calculations, helping researchers determine enthalpy changes, analyze reaction pathways, and understand energy conservation in chemical systems.
What Is Hess’s Law?
Hess’s Law states that the total enthalpy change of a chemical reaction is the same regardless of the pathway the reaction takes.
In other words, whether a reaction occurs in one step or several intermediate steps, the overall energy change remains constant.
This happens because energy is conserved in chemical processes. The heat released or absorbed depends only on the initial reactants and final products, not on how the reaction proceeds.
This principle makes it possible to determine unknown enthalpy values by combining known reactions.
Why Hess’s Law Is Important in Thermochemistry
Many chemical reactions cannot be easily measured in a laboratory due to safety concerns, reaction speed, or unstable intermediates. Hess’s Law allows scientists to calculate the heat change for these reactions indirectly.
It is widely used to:
- Determine enthalpy changes for complex reactions
- Predict whether reactions release or absorb heat
- Analyze reaction efficiency in industrial chemistry
- Calculate energy changes in biological processes
- Build thermodynamic data tables for chemical substances
Because of its reliability, Hess’s Law is a foundational tool in chemistry education and research.
The Principle of Energy Conservation
Hess’s Law is based on the law of conservation of energy, which states that energy cannot be created or destroyed, only transferred or transformed.
In chemical reactions, energy is often transferred as heat. If a reaction occurs through multiple steps, each step may release or absorb energy, but the total change will always equal the overall reaction energy.
For example, if a reaction proceeds through two intermediate steps:
- Step 1 releases a certain amount of heat
- Step 2 absorbs some heat
The total heat change equals the combined energy change of both steps.
This predictable relationship makes thermochemical calculations possible.
How Hess’s Law Works in Calculations
To apply Hess’s Law, chemists combine multiple known reactions to produce the target reaction.
This process usually involves three steps:
1. Identify Known Reactions
Chemists start with reactions that have known enthalpy changes from experimental data.
2. Manipulate the Reactions
Reactions may be:
- Reversed
- Multiplied by a coefficient
- Added together
These adjustments help match the target chemical equation.
3. Combine Enthalpy Changes
Once the equations are combined to produce the desired reaction, the enthalpy values are added or adjusted accordingly.
Because energy changes follow the same mathematical operations as the reactions themselves, the final result gives the enthalpy change of the target reaction.
Example of Hess’s Law Concept
Consider a situation where a chemical reaction occurs in multiple steps.
Instead of measuring the direct reaction, scientists may measure two related reactions that are easier to study. By combining those reactions, they can determine the heat change for the original reaction.
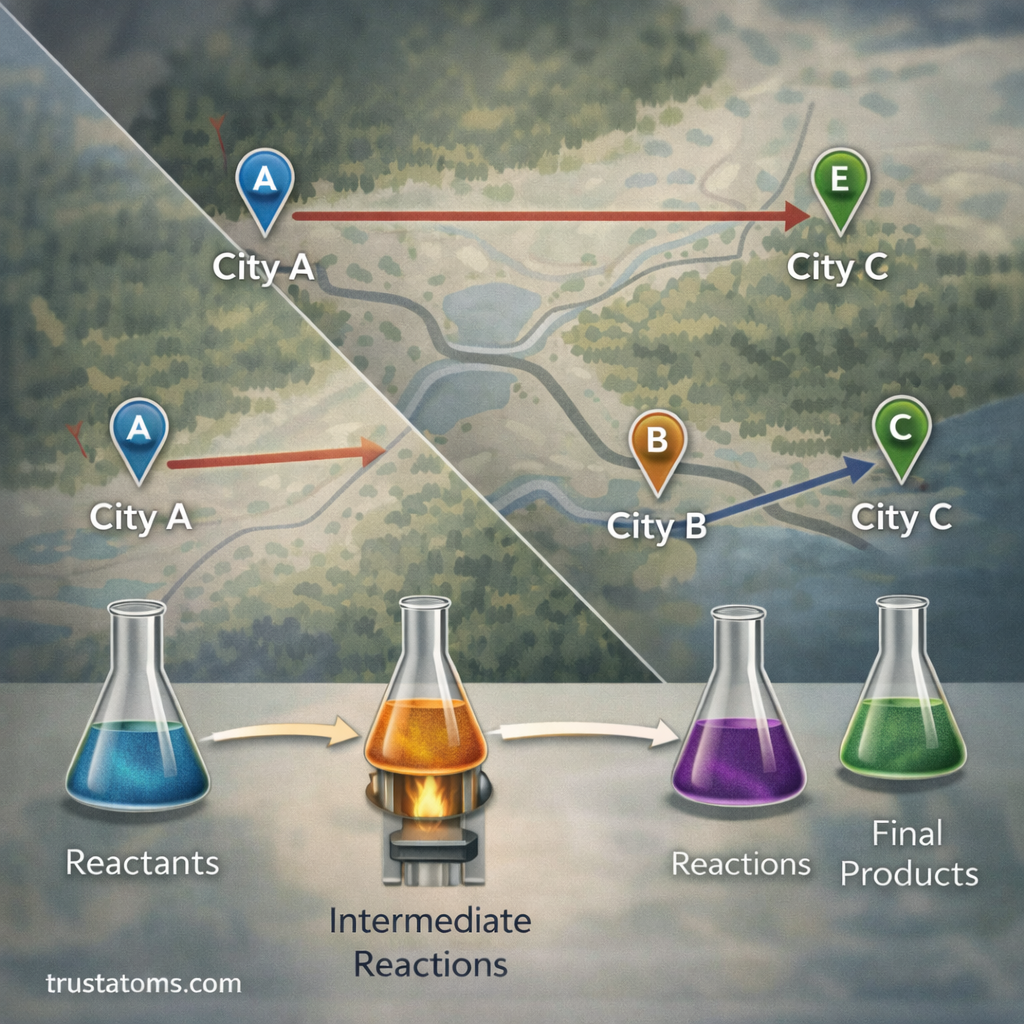
Conceptually, this works like a path-independent journey:
- Traveling from City A to City C directly requires a certain distance.
- Traveling from City A to City B and then B to C results in the same total distance.
Similarly, energy changes depend only on the starting and ending chemical states.
Standard Enthalpy of Formation
Hess’s Law is frequently used with standard enthalpy of formation values, which describe the heat change when a compound forms from its elements under standard conditions.
These values allow chemists to calculate the enthalpy change of reactions using reference data.
The process involves:
- Summing the enthalpies of formation of the products
- Summing the enthalpies of formation of the reactants
- Subtracting the reactant total from the product total
This approach simplifies thermochemical calculations for complex reactions.
Applications of Hess’s Law
Hess’s Law is used in many scientific and industrial fields.
Industrial Chemistry
Chemical manufacturers rely on thermochemical calculations to optimize energy efficiency and reaction conditions.
Fuel Energy Analysis
Scientists determine how much energy is released when fuels burn, which helps evaluate energy resources and combustion systems.
Environmental Chemistry
Researchers use thermochemical data to study atmospheric reactions and pollution-related processes.
Biochemistry
Energy changes in metabolic reactions can be analyzed using principles similar to Hess’s Law.
These applications highlight how important thermochemical calculations are for understanding real-world chemical systems.
Advantages of Using Hess’s Law
Hess’s Law provides several important benefits in chemical analysis.
- Allows indirect calculation of reaction heat changes
- Works even when reactions cannot be measured directly
- Simplifies complex thermodynamic problems
- Supports development of reliable thermochemical databases
Because of these advantages, Hess’s Law remains a key concept in chemistry education and research.
Common Challenges in Thermochemical Calculations
While Hess’s Law is straightforward conceptually, calculations can sometimes become complex.
Common challenges include:
- Correctly balancing chemical equations
- Adjusting enthalpy values when reactions are multiplied or reversed
- Managing large thermochemical datasets
Careful organization and step-by-step calculations help ensure accurate results.
Conclusion
Hess’s Law is a powerful principle that allows chemists to determine the heat changes of reactions through indirect calculations. By relying on energy conservation and known thermochemical data, scientists can analyze reactions that would otherwise be difficult or impossible to measure directly.
From laboratory experiments to large-scale industrial processes, Hess’s Law provides essential insight into how energy flows through chemical reactions. Its role in thermochemistry makes it one of the most important tools for understanding the energetic behavior of chemical systems.




