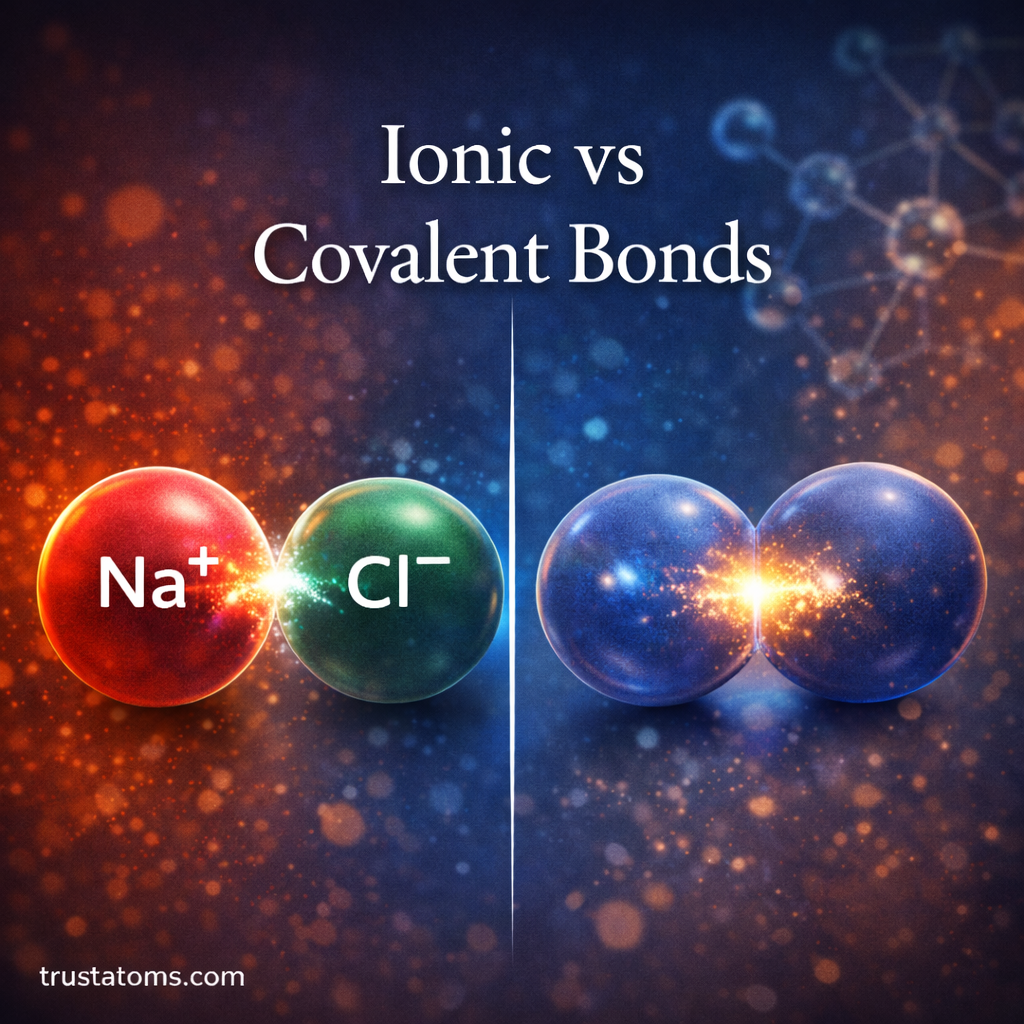
Ionic and covalent bonds are the two primary ways atoms join together to form compounds. While both involve electrons and the pursuit of stability, they differ in how those electrons behave.
Understanding ionic vs covalent bonds helps explain why salt dissolves in water, why metals conduct electricity, and why gases like oxygen exist as molecules. Let’s break down the key differences in a clear, simple way.
What Is an Ionic Bond?
An ionic bond forms when one atom transfers one or more electrons to another atom.
- One atom loses electrons and becomes positively charged (cation).
- The other atom gains electrons and becomes negatively charged (anion).
- Opposite charges attract, forming a strong electrostatic bond.
This type of bonding usually occurs between a metal and a nonmetal.
Example of an Ionic Bond
Sodium (a metal) loses one electron.
Chlorine (a nonmetal) gains that electron.
The result is sodium chloride (table salt).
What Is a Covalent Bond?
A covalent bond forms when two atoms share electrons instead of transferring them.
Rather than becoming full positive or negative ions, both atoms share pairs of electrons to fill their outer shells.
Covalent bonds typically form between two nonmetals.
Example of a Covalent Bond
In water (H₂O):
- Hydrogen and oxygen share electrons.
- The shared electrons help both atoms achieve stability.
Covalent bonds can be:
- Single (one shared pair)
- Double (two shared pairs)
- Triple (three shared pairs)
Key Differences Between Ionic and Covalent Bonds

Here’s a direct comparison:
1. Electron Behavior
Ionic bond:
- Electrons are transferred.
- Ions form (positive and negative charges).
Covalent bond:
- Electrons are shared.
- No full charges form (though partial charges may).
2. Types of Elements Involved
Ionic bond:
- Metal + nonmetal
Covalent bond:
- Nonmetal + nonmetal
3. Melting and Boiling Points
Ionic compounds:
- Usually high melting and boiling points.
- Strong electrostatic attraction between ions.
Covalent compounds:
- Usually lower melting and boiling points.
- Weaker intermolecular forces (in many cases).
4. Electrical Conductivity
Ionic compounds:
- Conduct electricity when dissolved in water or melted.
- Do not conduct well as solid crystals.
Covalent compounds:
- Generally poor conductors of electricity.
- Exceptions exist (like graphite).
Why Ionic Bonds Are Strong
Ionic bonds are strong because they involve full positive and negative charges attracting each other.
This electrostatic attraction can extend in a repeating crystal lattice, creating rigid, structured solids.
That’s why ionic compounds like salt are often:
- Hard
- Brittle
- Crystalline
Why Covalent Bonds Create Molecules
Covalent bonds form discrete molecules or extended networks.
For example:
- Oxygen gas (O₂) is a small molecule.
- Carbon dioxide (CO₂) is a linear molecule.
- Diamond is a giant covalent network.
Covalent bonding allows for enormous diversity in chemical structures — especially in organic chemistry, where carbon forms long chains and rings.
Polar vs Nonpolar Covalent Bonds
Not all covalent bonds share electrons equally.
Nonpolar Covalent Bond
- Electrons shared evenly.
- No partial charges.
- Example: O₂
Polar Covalent Bond
- Electrons shared unevenly.
- Slight positive and negative regions form.
- Example: Water (H₂O)
Polarity plays a major role in properties like solubility and intermolecular attraction.
How Electronegativity Explains the Difference
Electronegativity is an atom’s ability to attract electrons.
The greater the difference in electronegativity between two atoms:
- Large difference → Ionic bond
- Small difference → Covalent bond
- Moderate difference → Polar covalent bond
This concept helps predict the type of bond that will form.
Real-World Examples
Ionic compounds:
- Sodium chloride (NaCl)
- Calcium fluoride (CaF₂)
- Potassium bromide (KBr)
Covalent compounds:
- Water (H₂O)
- Methane (CH₄)
- Carbon dioxide (CO₂)
Summary: Ionic vs Covalent Bonds
Ionic and covalent bonds both allow atoms to achieve stability, but they do so differently.
Ionic bonds:
- Transfer electrons
- Form charged ions
- Create strong crystal lattices
Covalent bonds:
- Share electrons
- Form molecules
- Allow structural diversity
Understanding these two bonding types provides the foundation for chemistry. Nearly every substance around you — from table salt to the air you breathe — exists because of ionic or covalent bonding.