
Understanding how atoms arrange themselves in molecules is a key part of chemistry. One of the most widely used models for predicting molecular shape is VSEPR theory, which stands for Valence Shell Electron Pair Repulsion.
VSEPR theory helps chemists predict how atoms bond and how molecules take shape in three-dimensional space. These shapes influence physical properties, chemical reactivity, polarity, and even biological interactions.
In this article, we’ll explore what VSEPR theory is, how molecular geometry is determined, and why molecular shapes matter in chemistry.
What Is VSEPR Theory?
VSEPR theory explains molecular geometry based on a simple principle:
Electron pairs repel each other and arrange themselves as far apart as possible around a central atom.
These electron pairs can include:
- Bonding pairs (shared between atoms)
- Lone pairs (nonbonding electrons)
Because electrons carry negative charge, they repel one another. To minimize this repulsion, electron groups spread out in space, creating predictable molecular shapes.
Chemists use this model to estimate the three-dimensional arrangement of atoms around a central atom in a molecule.
Electron Groups and Molecular Shape
In VSEPR theory, the number of electron groups around a central atom determines the overall geometry.
An electron group can be:
- A single bond
- A double bond
- A triple bond
- A lone pair
Important note:
Even though double and triple bonds contain multiple electrons, they count as one electron group in VSEPR calculations.
Basic Molecular Geometries in VSEPR Theory
Below are the most common molecular shapes predicted by VSEPR theory.
Linear Geometry (2 Electron Groups)
When a central atom has two electron groups, they arrange themselves opposite each other.
Characteristics:
- Bond angle: 180°
- Shape: straight line
- Example molecules: carbon dioxide (CO₂)
This arrangement minimizes repulsion because the electron groups are positioned as far apart as possible.
Trigonal Planar Geometry (3 Electron Groups)
With three electron groups, the structure forms a flat triangular arrangement.
Characteristics:
- Bond angle: 120°
- Shape: triangular and planar
- Example molecules: boron trifluoride (BF₃)
All atoms lie in the same plane, creating a symmetrical structure.
Tetrahedral Geometry (4 Electron Groups)
When there are four electron groups, they form a three-dimensional structure called a tetrahedron.
Characteristics:
- Bond angle: 109.5°
- Shape: four bonds directed toward the corners of a tetrahedron
- Example molecules: methane (CH₄)
This geometry is one of the most common shapes in chemistry.
Trigonal Bipyramidal Geometry (5 Electron Groups)
Five electron groups arrange themselves in a shape known as trigonal bipyramidal.
Characteristics:
- Two axial positions (top and bottom)
- Three equatorial positions (around the center)
- Bond angles: 90° and 120°
- Example molecules: phosphorus pentachloride (PCl₅)
Equatorial positions experience less electron repulsion than axial ones.
Octahedral Geometry (6 Electron Groups)
When six electron groups surround a central atom, the molecule adopts an octahedral shape.
Characteristics:
- Bond angles: 90°
- Symmetrical arrangement
- Example molecules: sulfur hexafluoride (SF₆)
Atoms are positioned at the corners of an octahedron around the central atom.
The Role of Lone Pairs in Molecular Geometry
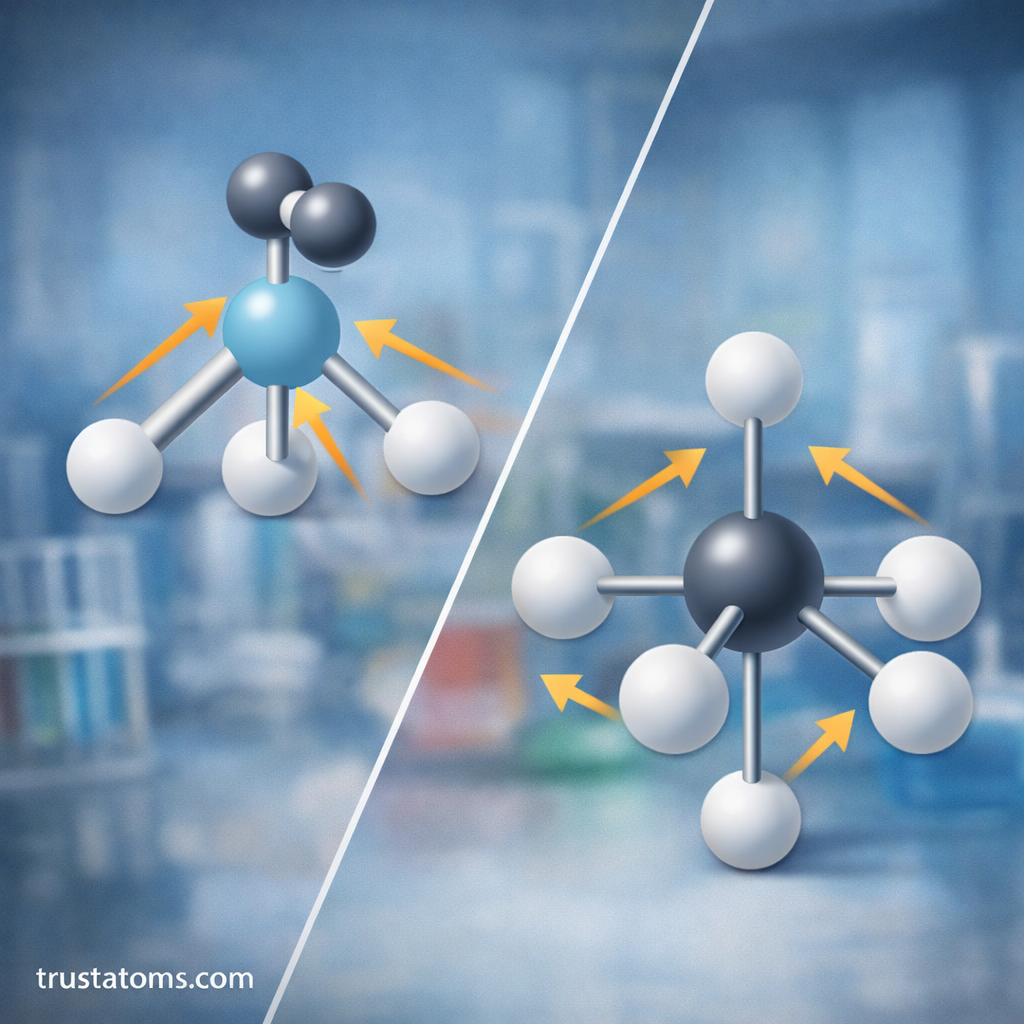
Lone pairs significantly affect molecular shape because they occupy space and repel bonding pairs.
However, lone pairs are not visible in molecular geometry diagrams, since geometry describes the positions of atoms rather than electron pairs.
Lone pairs exert stronger repulsive forces than bonding pairs, which can distort ideal bond angles.
Examples:
- Ammonia (NH₃): trigonal pyramidal shape
- Water (H₂O): bent shape
In both cases, lone pairs compress the bond angles between atoms.
Steps for Predicting Molecular Geometry
Chemists typically follow a systematic process when applying VSEPR theory.
Step 1: Draw the Lewis Structure
Start by identifying:
- The central atom
- Bonding pairs
- Lone pairs
This structure shows how electrons are arranged in the molecule.
Step 2: Count Electron Groups
Determine how many electron groups surround the central atom.
Remember:
- Multiple bonds count as one group
- Lone pairs count as one group
Step 3: Determine Electron Geometry
Electron geometry describes the arrangement of all electron groups around the central atom.
Common examples:
- 2 groups → Linear
- 3 groups → Trigonal planar
- 4 groups → Tetrahedral
- 5 groups → Trigonal bipyramidal
- 6 groups → Octahedral
Step 4: Determine Molecular Geometry
Molecular geometry focuses on the positions of atoms only, ignoring lone pairs.
For example:
- Tetrahedral electron geometry + 1 lone pair → trigonal pyramidal
- Tetrahedral electron geometry + 2 lone pairs → bent
Why Molecular Geometry Matters
The shape of a molecule influences many chemical properties.
Polarity
Molecular geometry determines whether a molecule is polar or nonpolar, which affects:
- Solubility
- Intermolecular forces
- Biological activity
Chemical Reactivity
The spatial arrangement of atoms affects how molecules interact during chemical reactions.
For example:
- Enzyme-substrate binding
- Reaction mechanisms
- Catalytic activity
Physical Properties
Molecular shape can influence:
- Boiling point
- Melting point
- Density
- Phase behavior
Even small changes in geometry can significantly alter how substances behave.
Limitations of VSEPR Theory
Although VSEPR theory is extremely useful, it has limitations.
Some of these include:
- It is a simplified model
- It does not fully account for electron delocalization
- It may not accurately predict shapes for complex molecules
More advanced methods, such as molecular orbital theory and quantum chemistry calculations, provide deeper insights into molecular structure.
However, VSEPR remains an essential starting point for understanding molecular geometry.
Final Thoughts
VSEPR theory provides a straightforward way to predict the shapes of molecules based on electron pair repulsion. By examining how electron groups arrange themselves around a central atom, chemists can determine molecular geometry and better understand chemical behavior.
From simple linear molecules to complex three-dimensional structures, molecular geometry plays a crucial role in chemistry, biology, and materials science. Learning how to apply VSEPR theory gives students and scientists a powerful tool for visualizing the invisible world of atoms and molecules.
SEO Meta Description




