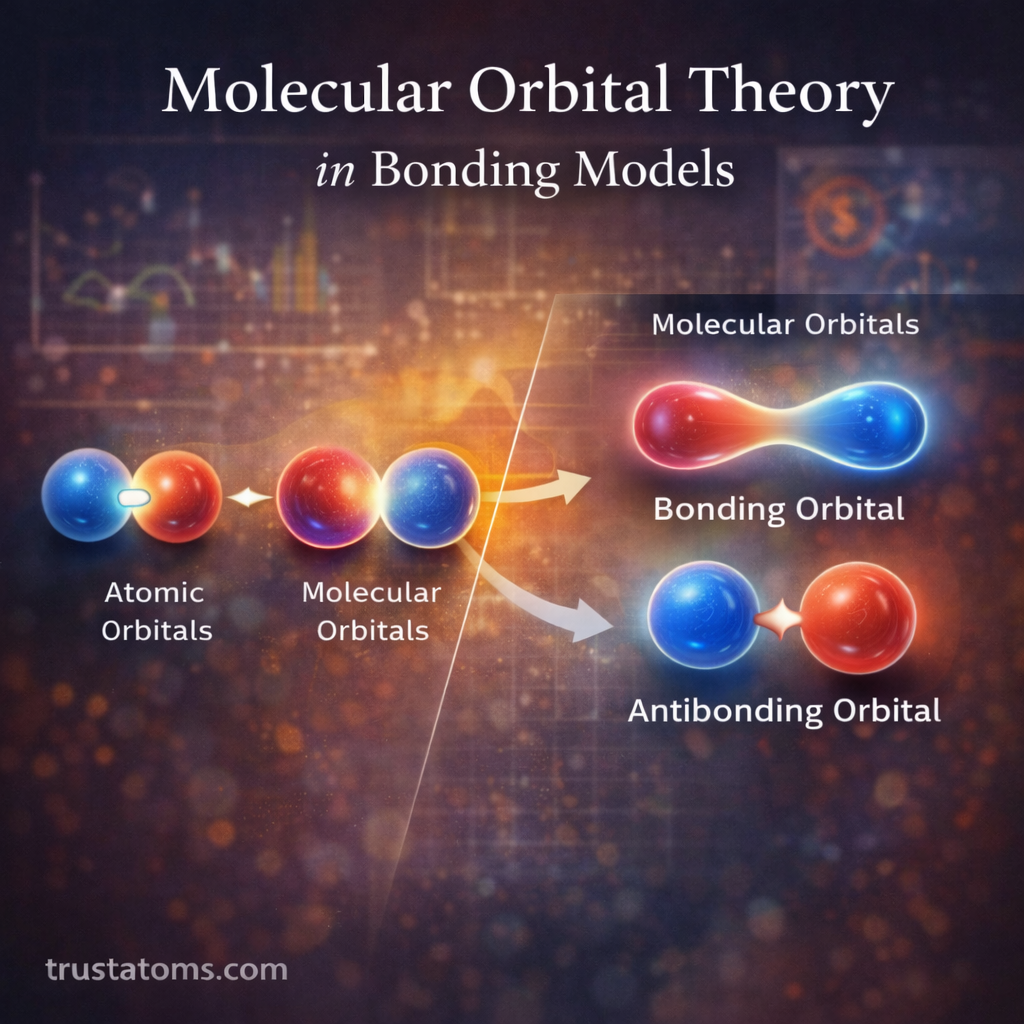
Molecular Orbital (MO) Theory is one of the most powerful models used in modern chemistry to explain how atoms bond together to form molecules. Unlike earlier bonding theories that treat electrons as localized between specific atoms, molecular orbital theory describes electrons as occupying orbitals that extend across an entire molecule.
This perspective allows chemists to understand bonding, molecular stability, magnetism, and even the color of substances at a deeper level. Today, molecular orbital theory plays a central role in quantum chemistry, materials science, and spectroscopy.
What Is Molecular Orbital Theory?
Molecular Orbital Theory describes chemical bonding by combining atomic orbitals from individual atoms to form molecular orbitals that belong to the entire molecule rather than a single atom.
In this model:
- Atomic orbitals merge when atoms approach each other
- New orbitals form that can extend across multiple atoms
- Electrons occupy these orbitals according to energy rules
Instead of electrons being confined between two atoms, molecular orbital theory allows electrons to be delocalized across the molecule.
This approach provides a more accurate description of many molecules, especially those that cannot be explained well using simple bonding models.
How Molecular Orbitals Form
Molecular orbitals form when atomic orbitals overlap and combine mathematically. When two atomic orbitals interact, they create two new molecular orbitals:
- A bonding orbital
- An antibonding orbital
Bonding Orbitals
Bonding orbitals occur when atomic orbitals combine constructively.
Characteristics include:
- Increased electron density between nuclei
- Lower energy than the original atomic orbitals
- Stabilization of the molecule
Electrons placed in bonding orbitals help hold atoms together.
Antibonding Orbitals
Antibonding orbitals form when atomic orbitals combine destructively.
Characteristics include:
- Reduced electron density between nuclei
- Higher energy than atomic orbitals
- Destabilization of the molecule
Electrons occupying antibonding orbitals weaken chemical bonds.
Types of Molecular Orbitals
Molecular orbitals are categorized based on how atomic orbitals overlap.
Sigma (σ) Orbitals
Sigma orbitals form when atomic orbitals overlap head-on along the axis connecting two nuclei.
Key features:
- Strong orbital overlap
- Symmetrical electron density around the bond axis
- Typically form single bonds
Examples include overlap between s–s, s–p, or p–p orbitals along the bonding axis.
Pi (π) Orbitals
Pi orbitals form when p orbitals overlap side-by-side.
Characteristics include:
- Electron density above and below the bond axis
- Weaker overlap than sigma bonds
- Common in double and triple bonds
Pi bonds often work alongside sigma bonds to strengthen molecular connections.
Bond Order and Molecular Stability
One important concept in molecular orbital theory is bond order, which helps determine the strength and stability of a bond.
Bond order can be calculated using the number of electrons in bonding and antibonding orbitals.
Bond order = (bonding electrons − antibonding electrons) ÷ 2
The bond order indicates how strong the bond is.
Interpretations of bond order:
- Bond order = 0 → no stable bond forms
- Bond order = 1 → single bond
- Bond order = 2 → double bond
- Bond order = 3 → triple bond
Higher bond orders generally mean stronger and shorter bonds.
Molecular Orbital Diagrams
Chemists often visualize molecular orbitals using molecular orbital diagrams.
These diagrams display:
- Atomic orbitals from each atom
- The resulting molecular orbitals
- Electron filling according to energy levels
Electrons fill orbitals according to three important rules:
- Aufbau Principle – Electrons occupy the lowest energy orbitals first.
- Pauli Exclusion Principle – Each orbital holds a maximum of two electrons with opposite spins.
- Hund’s Rule – Electrons occupy orbitals singly before pairing.
These rules ensure that electrons distribute in the most stable arrangement possible.
Molecular Orbital Theory vs Valence Bond Theory
Molecular Orbital Theory and Valence Bond (VB) Theory are both used to describe chemical bonding, but they approach the problem differently.
Valence Bond Theory
- Electrons are localized between atoms
- Bonds form from overlapping atomic orbitals
- Works well for simple molecules
Molecular Orbital Theory
- Electrons are delocalized across the molecule
- Orbitals extend over the entire molecular structure
- Explains magnetic and electronic properties better
Both models are useful, but molecular orbital theory often provides more detailed explanations for complex molecular behavior.
Explaining Magnetism with Molecular Orbital Theory

Molecular orbital theory helps explain why certain molecules are paramagnetic or diamagnetic.
Paramagnetic Molecules
Paramagnetic molecules contain unpaired electrons.
These molecules are attracted to magnetic fields.
Example:
Oxygen (O₂) is paramagnetic because molecular orbital theory predicts unpaired electrons in antibonding orbitals.
Diamagnetic Molecules
Diamagnetic molecules have all electrons paired.
They are weakly repelled by magnetic fields.
This difference cannot always be explained using simpler bonding theories, which highlights the strength of the MO model.
Applications of Molecular Orbital Theory
Molecular orbital theory is widely used in many scientific fields.
Organic Chemistry
- Explaining conjugation and aromaticity
- Predicting reactivity of molecules
Materials Science
- Understanding semiconductor behavior
- Designing conductive polymers
Spectroscopy
- Interpreting light absorption and emission
- Studying electronic transitions
Biochemistry
- Explaining electron transfer in enzymes
- Understanding photosynthesis and respiration
These applications show how molecular orbital theory connects atomic structure to real-world chemical behavior.
Limitations of Molecular Orbital Theory
While molecular orbital theory is powerful, it also has limitations.
Challenges include:
- Complex calculations for large molecules
- Requires quantum mechanical mathematics
- Often relies on computational methods
Despite these challenges, modern chemistry frequently uses computer models to apply molecular orbital theory to complex molecular systems.
Final Thoughts
Molecular Orbital Theory provides a deeper understanding of chemical bonding by describing electrons as occupying orbitals that extend across an entire molecule. This model explains bond strength, molecular stability, magnetism, and electronic behavior in ways that simpler theories cannot.
By combining atomic orbitals into bonding and antibonding molecular orbitals, chemists can predict how molecules form and how they behave under different conditions. As computational chemistry continues to advance, molecular orbital theory remains a central framework for understanding the quantum nature of chemical bonds.