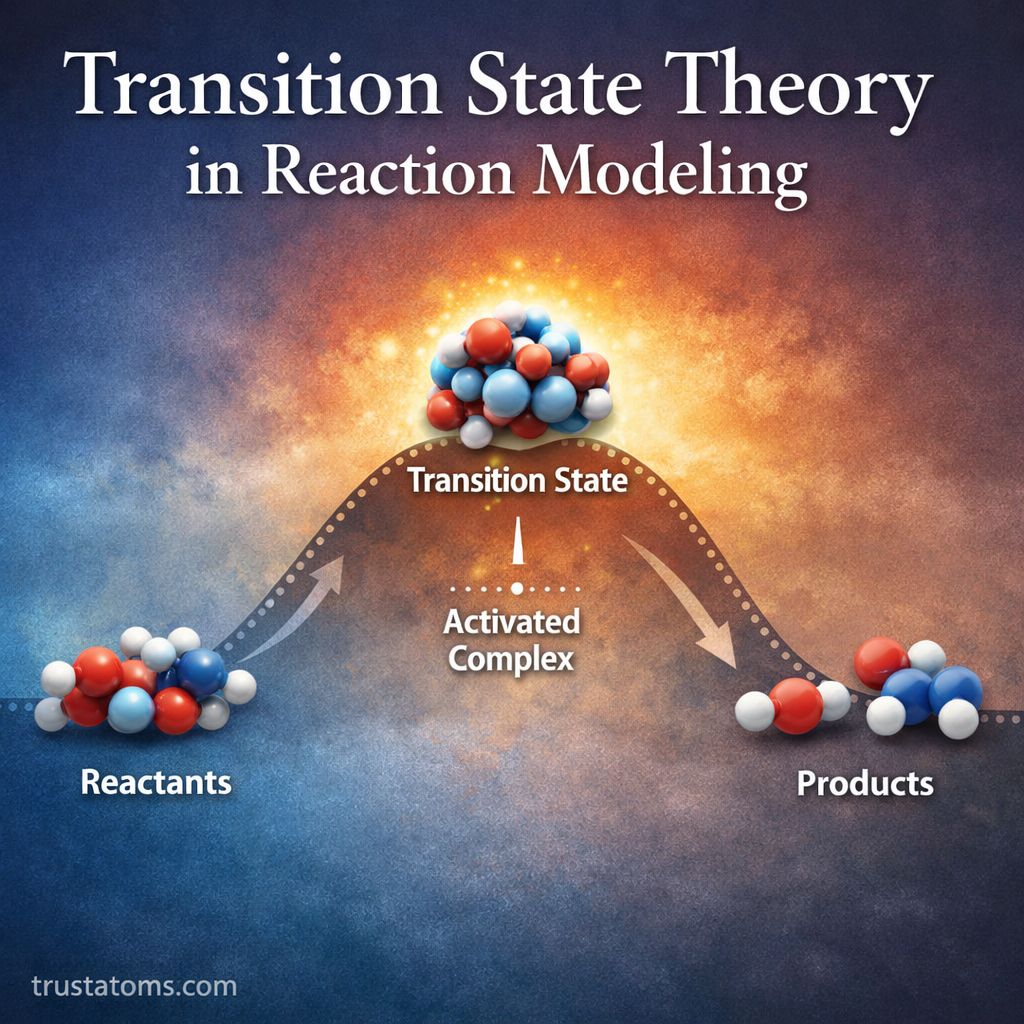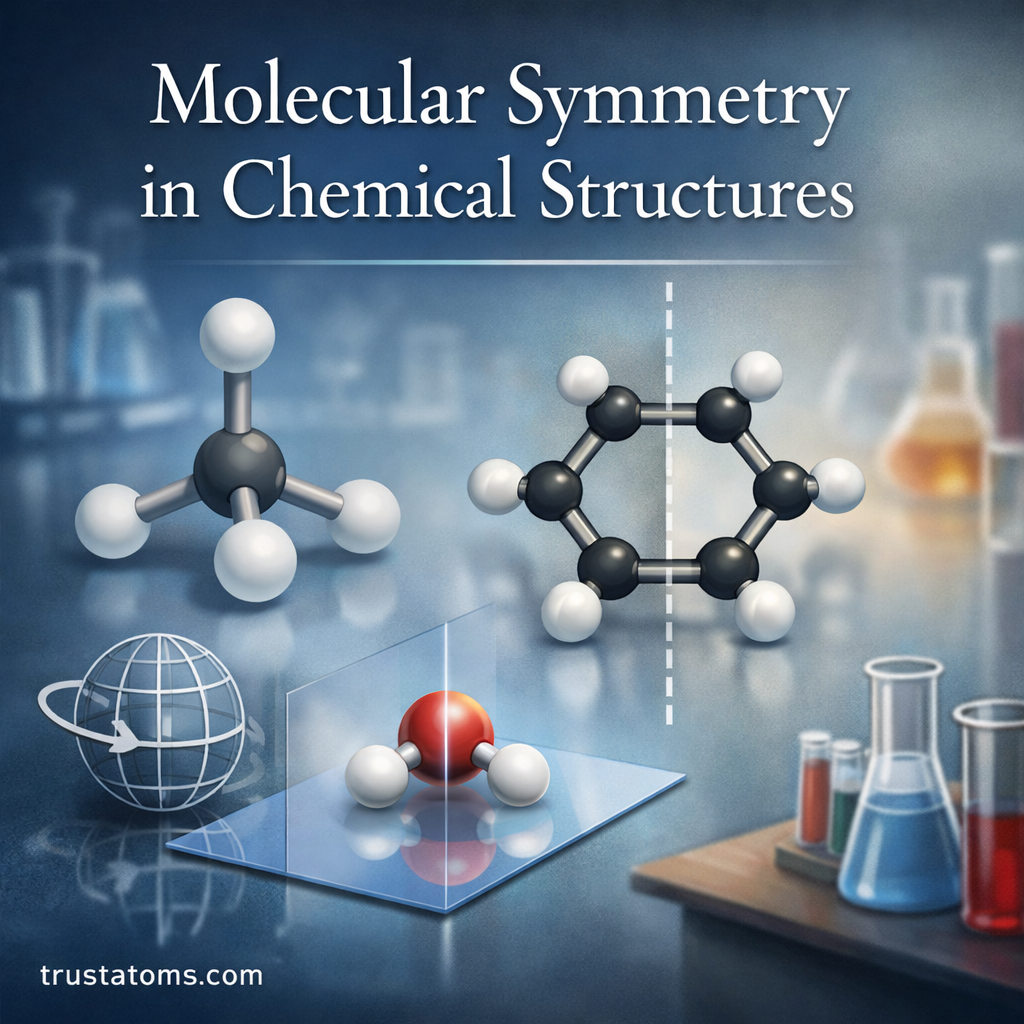
Molecular symmetry is one of the most elegant concepts in chemistry. It describes how atoms in a molecule are arranged in space and how that arrangement repeats through specific symmetry operations.
Understanding symmetry helps chemists predict molecular behavior, physical properties, chemical reactions, and spectroscopic patterns. From simple molecules like methane to complex organic compounds and crystalline materials, symmetry provides a powerful framework for analyzing chemical structures.
In this guide, we’ll explore what molecular symmetry is, the key symmetry elements, how molecules are classified into symmetry groups, and why symmetry matters in chemistry.
What Is Molecular Symmetry?
Molecular symmetry refers to the balanced arrangement of atoms within a molecule that allows certain geometric operations to produce an identical configuration.
If you can rotate, reflect, or invert a molecule and it appears unchanged, the molecule possesses symmetry.
Chemists analyze symmetry using symmetry operations and symmetry elements, which together define the molecule’s overall symmetry.
Examples of symmetrical molecules include:
- Methane (CH₄)
- Benzene (C₆H₆)
- Carbon dioxide (CO₂)
These molecules have repeating spatial patterns that remain unchanged under specific transformations.
Why Molecular Symmetry Matters
Symmetry is more than just visual balance—it has real chemical consequences.
Chemists use symmetry to understand and predict:
- Molecular vibrations
- Spectroscopy results
- Chemical bonding
- Reaction mechanisms
- Optical activity
- Crystal structures
For example, infrared and Raman spectroscopy depend heavily on molecular symmetry because only certain vibrational modes are spectroscopically active.
Symmetry also simplifies complex mathematical calculations in quantum chemistry and molecular orbital theory.
The Five Main Symmetry Elements
Symmetry in molecules is defined by specific geometric features called symmetry elements. Each symmetry element corresponds to a symmetry operation.
1. Identity (E)
The identity operation means doing nothing to the molecule.
Every molecule possesses this symmetry element because leaving the molecule unchanged still counts as a symmetry operation.
Although it seems trivial, the identity element is required in symmetry classification.
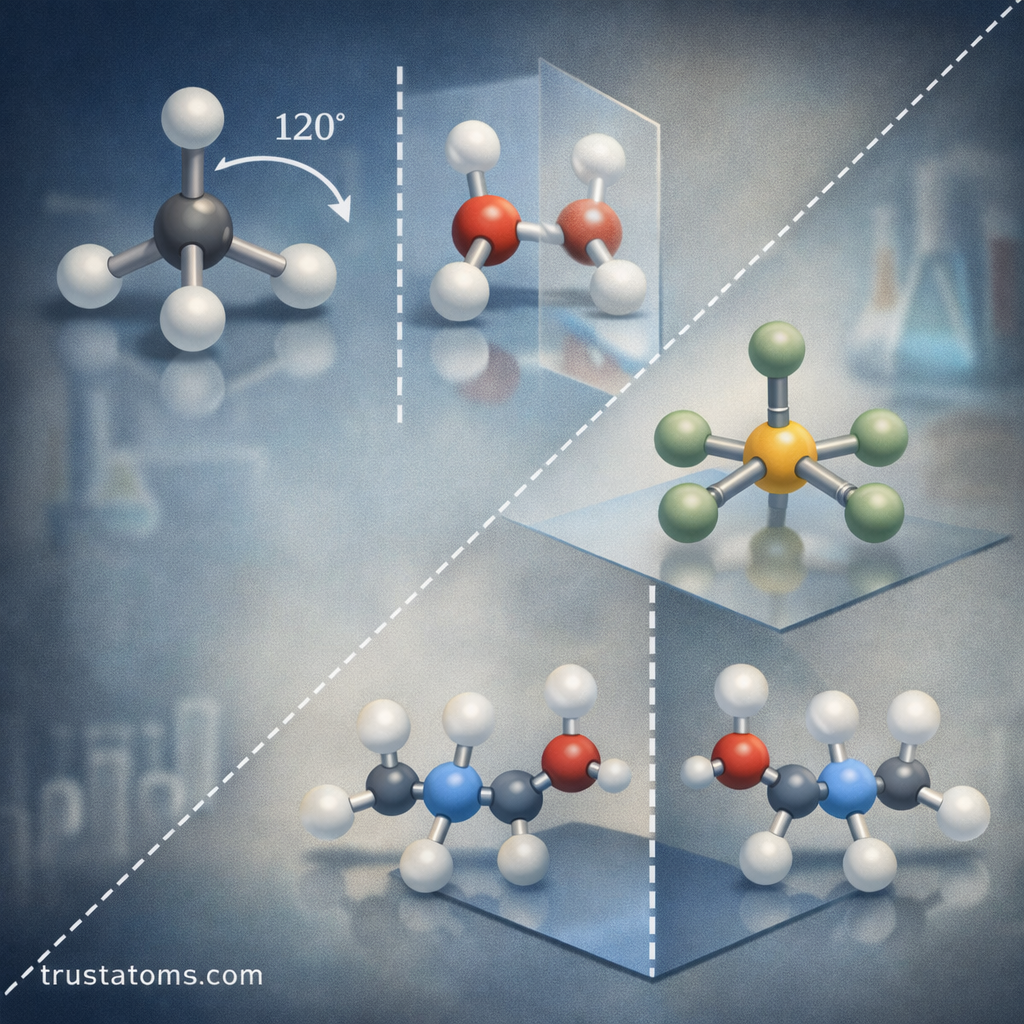
2. Rotation Axis (Cn)
A rotation axis exists when a molecule can be rotated around an axis and still appear identical.
The notation Cn represents rotation by:
360° / n
Examples:
- C₂ axis: 180° rotation
- C₃ axis: 120° rotation
- C₄ axis: 90° rotation
Methane contains multiple rotational symmetry axes due to its tetrahedral geometry.
3. Mirror Plane (σ)
A mirror plane divides a molecule into two halves that are mirror images of each other.
There are three common types of mirror planes:
- σv – vertical plane through the rotation axis
- σh – horizontal plane perpendicular to the axis
- σd – diagonal mirror plane
Water (H₂O), for example, contains vertical mirror planes that reflect one hydrogen atom onto the other.
4. Center of Inversion (i)
A center of inversion exists when every atom at coordinates (x, y, z) corresponds to another atom at (-x, -y, -z).
If the molecule remains identical after inversion through a central point, it has inversion symmetry.
Carbon dioxide (CO₂) is a classic example with a center of inversion at the carbon atom.
5. Improper Rotation Axis (Sn)
An improper rotation axis combines two operations:
- Rotation around an axis
- Reflection through a plane perpendicular to that axis
This combined transformation must produce an identical molecule.
Improper rotations appear frequently in molecules with complex three-dimensional symmetry.
Symmetry Operations Explained
A symmetry operation is the movement performed on a molecule that results in an indistinguishable configuration.
Common symmetry operations include:
- Rotation
- Reflection
- Inversion
- Improper rotation
For example:
- Rotating benzene by 60° produces the same structure
- Reflecting water across its symmetry plane produces the same molecule
These operations are fundamental for identifying a molecule’s symmetry group.
Point Groups: Classifying Molecular Symmetry
Chemists categorize molecules into point groups, which represent all symmetry operations that apply to a molecule.
A point group describes the full symmetry of a molecule relative to a central point.
Common point groups include:
C₁
- No symmetry except identity
- Asymmetrical molecules
Example: most complex organic molecules
C₂v
- One C₂ rotation axis
- Two vertical mirror planes
Example: water (H₂O)
D₃h
- Multiple rotation axes
- Horizontal mirror plane
Example: boron trifluoride (BF₃)
Td
- Highly symmetrical tetrahedral molecules
Example: methane (CH₄)
Oh
- Extremely symmetrical octahedral molecules
Example: sulfur hexafluoride (SF₆)
How Chemists Determine Molecular Symmetry
Identifying symmetry involves a systematic approach.
Chemists typically follow these steps:
1. Visualize the Molecular Geometry
First determine the 3D shape of the molecule using bonding theory or VSEPR geometry.
Examples:
- Linear
- Trigonal planar
- Tetrahedral
- Octahedral
2. Identify All Symmetry Elements
Look for:
- Rotation axes
- Mirror planes
- Inversion centers
- Improper rotation axes
3. Match the Pattern to a Point Group
Once symmetry elements are identified, the molecule can be assigned to a known point group using symmetry flowcharts or tables.
Examples of Molecular Symmetry
Water (H₂O)
Symmetry elements:
- Identity (E)
- One C₂ axis
- Two vertical mirror planes
Point group: C₂v
Methane (CH₄)
Symmetry elements:
- Multiple C₃ axes
- Several mirror planes
- Improper rotations
Point group: Td
Methane is one of the most symmetrical molecules in chemistry.
Carbon Dioxide (CO₂)
Symmetry elements:
- C∞ rotation axis
- Multiple mirror planes
- Center of inversion
Point group: D∞h
Linear molecules often have very high symmetry.
Applications of Molecular Symmetry
Molecular symmetry plays a major role across many branches of chemistry.
Spectroscopy
Symmetry determines which vibrational modes appear in:
- Infrared spectroscopy
- Raman spectroscopy
Some vibrations are active while others are forbidden based on symmetry.
Quantum Chemistry
Symmetry simplifies complex calculations by reducing the number of unique interactions in molecular orbitals.
This makes quantum models more computationally efficient.
Chemical Reactions
Reaction pathways often depend on symmetry rules.
A famous example is the Woodward–Hoffmann rules, which predict whether pericyclic reactions are allowed.
Crystallography
Symmetry principles also extend to crystal lattices and solid materials.
Crystal systems rely heavily on symmetry classification.
Common Misconceptions About Molecular Symmetry
Symmetrical molecules are always nonpolar
Not always.
While many symmetrical molecules cancel out dipole moments, some structures can still be polar depending on atom arrangement.
Large molecules cannot have symmetry
Even complex molecules may contain partial symmetry elements.
Symmetry analysis often focuses on the dominant repeating pattern.
Symmetry is only visual
Symmetry is mathematically defined and deeply tied to quantum mechanics, group theory, and spectroscopy.
It’s far more than just a geometric observation.
Final Thoughts
Molecular symmetry provides chemists with a powerful way to understand how atoms are arranged and how molecules behave. By identifying symmetry elements and classifying molecules into point groups, scientists can predict physical properties, analyze spectroscopic data, and understand chemical reactions more effectively.
Although symmetry may initially seem abstract, it simplifies complex chemical systems and reveals the underlying order within molecular structures. From small inorganic molecules to large biological compounds, symmetry remains a central tool in modern chemistry.