Understanding how atoms form chemical bonds requires more than simply knowing how many electrons they have. In many molecules, atoms rearrange their orbitals to form stronger and more stable bonds. This process is known as orbital hybridization.
Orbital hybridization explains how atoms combine atomic orbitals to create new hybrid orbitals that are better suited for bonding. These hybrid orbitals help explain molecular shapes, bond angles, and the strength of chemical bonds.
In this article, we’ll explore what orbital hybridization is, why it occurs, and how different hybridization types influence molecular bonding.
What Is Orbital Hybridization?
Orbital hybridization is a concept in chemistry that describes how atomic orbitals mix to form new hybrid orbitals used in bonding.
Atomic orbitals such as s, p, and sometimes d orbitals can combine to produce hybrid orbitals with new shapes and energy levels.
These hybrid orbitals:
- Allow atoms to form multiple bonds
- Create specific molecular geometries
- Provide stronger overlap between orbitals during bonding
Hybridization mainly occurs in the valence shell, which is the outermost energy level of an atom.
Why Hybridization Occurs
Atoms tend to form bonds that maximize stability and minimize energy. In many cases, the original atomic orbitals do not align properly for effective bonding.
Hybridization solves this problem by rearranging orbitals into shapes that allow better overlap between atoms.
This leads to:
- Stronger chemical bonds
- Predictable bond angles
- Stable molecular structures
For example, carbon atoms commonly hybridize their orbitals to form the stable bonding patterns seen in organic molecules.
Types of Orbital Hybridization
Several types of hybridization occur depending on how many orbitals mix together. The most common types include:
- sp hybridization
- sp² hybridization
- sp³ hybridization
- sp³d hybridization
- sp³d² hybridization
Each hybridization type corresponds to a specific molecular geometry.
sp Hybridization
sp hybridization occurs when one s orbital and one p orbital combine.
This produces two sp hybrid orbitals arranged in a linear geometry.
Key characteristics
- Number of hybrid orbitals: 2
- Bond angle: 180°
- Molecular geometry: linear
Common examples
- Carbon dioxide (CO₂)
- Acetylene (C₂H₂)
In sp hybridization, two unhybridized p orbitals remain available for forming π bonds, which are common in multiple bonds like double and triple bonds.
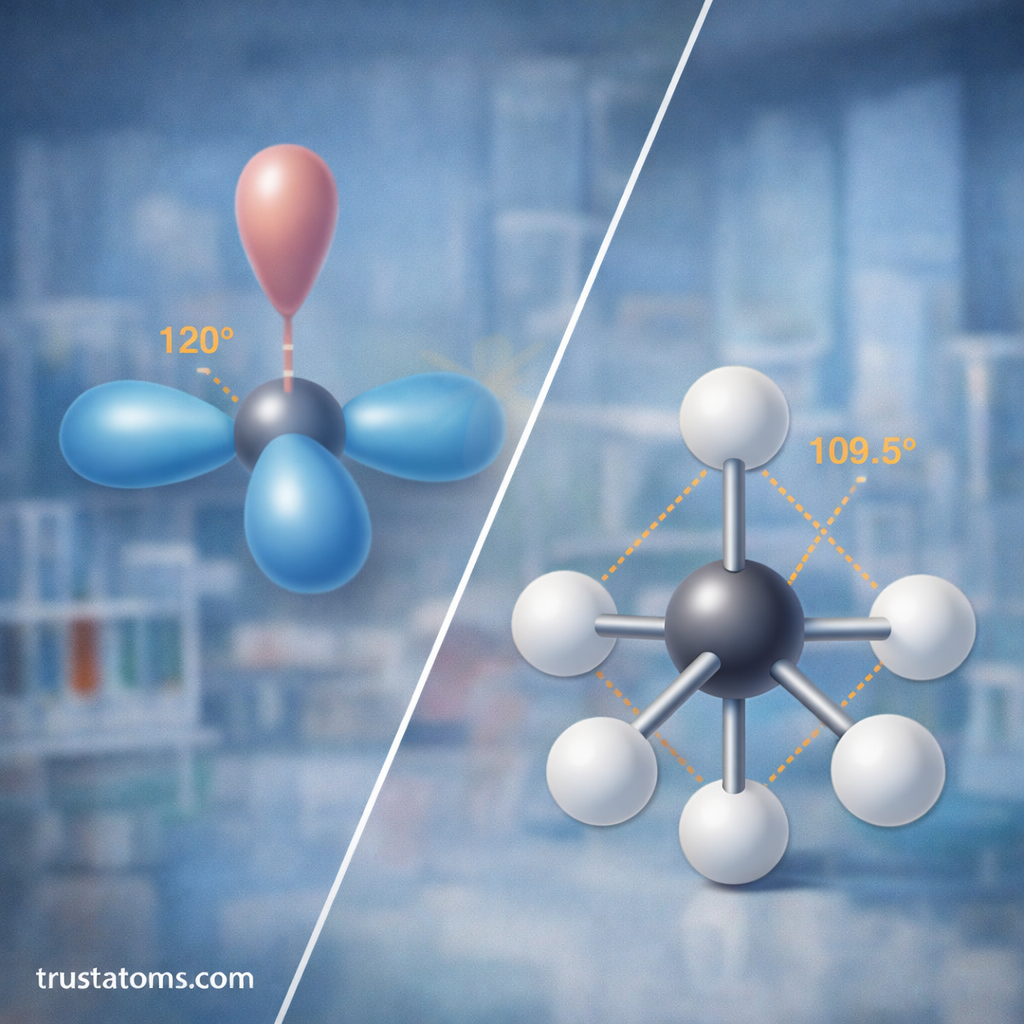
sp² Hybridization
sp² hybridization happens when one s orbital mixes with two p orbitals.
This produces three sp² hybrid orbitals arranged in a trigonal planar structure.
Key characteristics
- Number of hybrid orbitals: 3
- Bond angle: 120°
- Molecular geometry: trigonal planar
Common examples
- Ethene (C₂H₄)
- Boron trifluoride (BF₃)
One unhybridized p orbital remains perpendicular to the plane of the hybrid orbitals. This orbital often forms π bonds.
sp³ Hybridization
sp³ hybridization occurs when one s orbital mixes with three p orbitals.
This produces four sp³ hybrid orbitals arranged in a tetrahedral geometry.
Key characteristics
- Number of hybrid orbitals: 4
- Bond angle: 109.5°
- Molecular geometry: tetrahedral
Common examples
- Methane (CH₄)
- Ammonia (NH₃)
- Water (H₂O)
Even when lone pairs are present, the electron geometry still follows the tetrahedral arrangement predicted by hybridization.
sp³d Hybridization
sp³d hybridization occurs when one s orbital, three p orbitals, and one d orbital combine.
This produces five hybrid orbitals arranged in trigonal bipyramidal geometry.
Key characteristics
- Number of hybrid orbitals: 5
- Molecular geometry: trigonal bipyramidal
Common examples
- Phosphorus pentachloride (PCl₅)
This type of hybridization is typically found in atoms that have access to expanded valence shells, usually elements in the third period or below.
sp³d² Hybridization
sp³d² hybridization occurs when one s orbital, three p orbitals, and two d orbitals combine.
This produces six hybrid orbitals arranged in an octahedral geometry.
Key characteristics
- Number of hybrid orbitals: 6
- Molecular geometry: octahedral
Common examples
- Sulfur hexafluoride (SF₆)
This geometry places atoms evenly around the central atom, minimizing electron repulsion.
Hybridization and Molecular Geometry
Orbital hybridization works closely with molecular geometry concepts. While VSEPR theory predicts shapes based on electron pair repulsion, hybridization explains how orbitals rearrange to produce those shapes.
For example:
- sp hybridization → linear geometry
- sp² hybridization → trigonal planar geometry
- sp³ hybridization → tetrahedral geometry
Both models complement each other when explaining molecular structures.
Sigma Bonds and Pi Bonds
Orbital hybridization also helps explain the difference between sigma (σ) bonds and pi (π) bonds.
Sigma Bonds
Sigma bonds form when orbitals overlap directly along the bond axis.
Characteristics:
- Stronger than π bonds
- First bond formed between two atoms
- Allow rotation around the bond axis
Hybrid orbitals commonly form sigma bonds.
Pi Bonds
Pi bonds form when unhybridized p orbitals overlap sideways.
Characteristics:
- Found in double and triple bonds
- Weaker than sigma bonds
- Restrict rotational movement
For example:
- Double bond = 1 sigma + 1 pi bond
- Triple bond = 1 sigma + 2 pi bonds
How to Determine Hybridization
Chemists often determine hybridization using a simple method.
Step 1: Draw the Lewis structure
Identify bonding pairs and lone pairs around the central atom.
Step 2: Count electron regions
Each of the following counts as one region:
- Single bond
- Double bond
- Triple bond
- Lone pair
Step 3: Assign hybridization
Use the number of electron regions to determine hybridization.
| Electron Regions | Hybridization |
|---|---|
| 2 | sp |
| 3 | sp² |
| 4 | sp³ |
| 5 | sp³d |
| 6 | sp³d² |
This method works well for most simple molecules.
Importance of Orbital Hybridization
Orbital hybridization is essential for understanding chemical bonding in many fields of science.
Organic chemistry
Most carbon compounds rely on hybrid orbitals to form stable molecules.
Biochemistry
Molecular structures such as proteins, DNA, and enzymes depend on bonding patterns explained by hybridization.
Materials science
Hybridization influences the properties of materials like graphene, diamond, and polymers.
Understanding hybridization allows scientists to predict how molecules behave and interact.
Final Thoughts
Orbital hybridization provides a powerful explanation for how atoms form stable chemical bonds. By mixing atomic orbitals into new hybrid orbitals, atoms can arrange their bonds in ways that match observed molecular shapes and bond angles.
When combined with models like VSEPR theory, hybridization helps chemists visualize the structure of molecules and understand the forces that govern chemical bonding. This concept remains a cornerstone of modern chemistry and plays a crucial role in fields ranging from organic chemistry to materials science.