Proteins are among the most important molecules in living organisms. They perform a vast range of biological functions, including catalyzing chemical reactions, supporting cellular structure, transporting molecules, and regulating biological processes.
In biochemical systems, the structure of a protein determines its function. The way a protein folds and organizes in three-dimensional space controls how it interacts with other molecules and carries out biological tasks.
Understanding protein structure is a core concept in biochemistry, molecular biology, and structural biology, and it plays a key role in fields such as medicine, biotechnology, and drug development.
What Are Proteins?
Proteins are large biological macromolecules made from smaller building blocks called amino acids. These amino acids are linked together through peptide bonds, forming long chains known as polypeptides.
There are 20 standard amino acids commonly found in proteins. Each amino acid has unique chemical properties that influence how the protein folds and behaves.
Proteins are involved in nearly every cellular activity, including:
- Enzyme catalysis
- Cellular signaling
- Immune defense
- Molecular transport
- Structural support
- Gene regulation
The ability of proteins to perform these functions depends heavily on their structural organization.
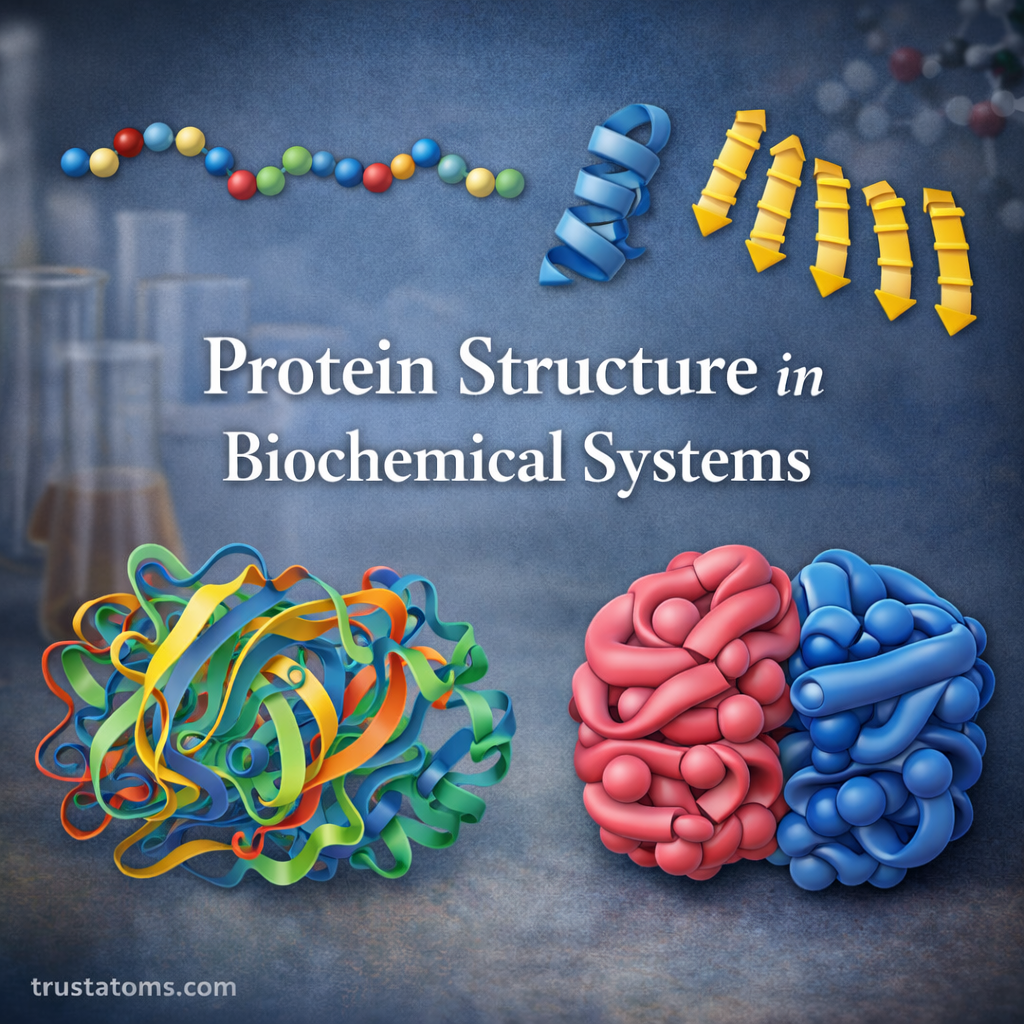
The Four Levels of Protein Structure
Protein structure is typically described using four hierarchical levels, each representing increasing complexity in how the molecule is organized.
These levels include:
- Primary structure
- Secondary structure
- Tertiary structure
- Quaternary structure
Each level builds upon the previous one and contributes to the protein’s final shape and function.
Primary Structure of Proteins
The primary structure refers to the linear sequence of amino acids in a polypeptide chain.
This sequence is determined by the genetic code stored in DNA and translated through the process of protein synthesis.
Important characteristics of primary structure include:
- The exact order of amino acids
- The number of amino acids in the chain
- The position of specific residues that influence folding
Even a small change in the amino acid sequence can dramatically affect how the protein functions.
For example, certain genetic mutations alter protein sequences and can lead to diseases or functional changes in biological systems.
Secondary Structure of Proteins
Secondary structure describes local folding patterns within a protein chain, stabilized primarily by hydrogen bonding.
Two major types of secondary structures occur frequently in proteins.
Alpha-Helix
The alpha-helix is a spiral-shaped structure where the polypeptide backbone coils around itself.
Key features include:
- Regular hydrogen bonding between nearby amino acids
- Compact helical arrangement
- Stability through repeated bonding patterns
Alpha-helices are common in proteins involved in cell membranes and structural components.
Beta-Sheet
The beta-sheet structure forms when segments of the protein chain align side by side.
Characteristics include:
- Sheet-like arrangement of polypeptide strands
- Hydrogen bonds between adjacent strands
- Either parallel or antiparallel orientation
Beta-sheets often contribute to the mechanical strength of proteins, especially in structural tissues.
Tertiary Structure of Proteins
The tertiary structure represents the overall three-dimensional shape of a single protein molecule.
This structure results from interactions between the side chains of amino acids. Several forces stabilize tertiary structure.
These include:
- Hydrogen bonds
- Ionic interactions
- Hydrophobic interactions
- Van der Waals forces
- Disulfide bonds
The tertiary structure determines:
- The protein’s functional regions
- Binding sites for other molecules
- Enzyme active sites
Because of this, even slight changes in tertiary structure can affect how proteins perform biological tasks.
Quaternary Structure of Proteins
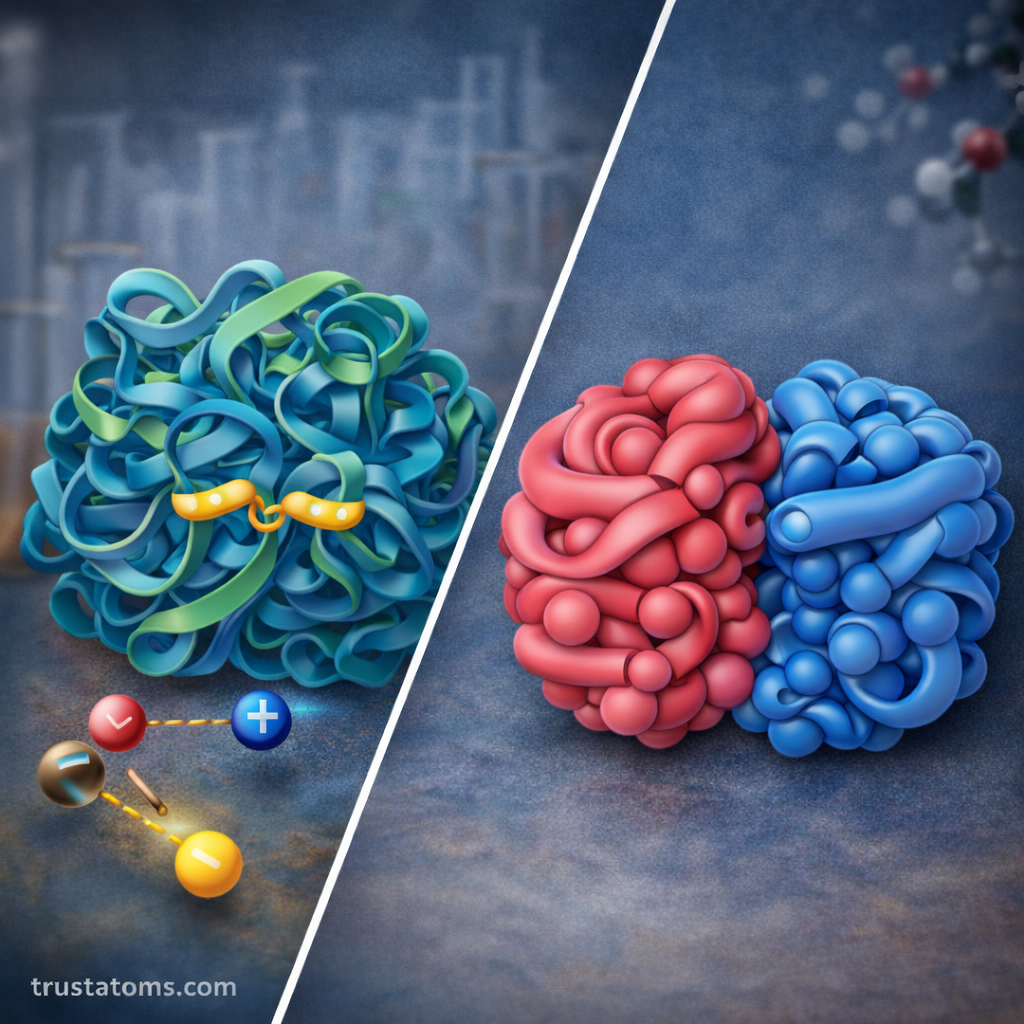
Some proteins consist of multiple polypeptide chains working together as a functional unit. This level of organization is called quaternary structure.
Each individual chain is known as a subunit, and the arrangement of these subunits creates the final functional protein.
Examples include:
- Hemoglobin, which contains four protein subunits
- Many enzyme complexes
- Antibody molecules
Quaternary structure allows proteins to perform more complex biological functions through cooperative interactions between subunits.
Forces That Stabilize Protein Structure
Several chemical interactions help maintain protein stability and shape.
Important stabilizing forces include:
Hydrogen Bonds
Hydrogen bonding occurs between atoms within the protein backbone or between side chains.
These bonds help stabilize secondary and tertiary structures.
Hydrophobic Interactions
Nonpolar amino acids tend to cluster together inside the protein, away from water.
This hydrophobic effect is one of the main drivers of protein folding.
Ionic Bonds
Charged amino acid side chains can attract one another, forming electrostatic interactions that stabilize protein structure.
Disulfide Bridges
Disulfide bonds form between sulfur atoms of cysteine residues.
These strong covalent bonds provide extra stability, especially in proteins exposed to harsh environments.
Protein Folding and Biological Function
Proteins do not remain as straight chains. Instead, they fold into specific three-dimensional shapes shortly after being synthesized in the cell.
Protein folding occurs through a complex process driven by:
- Chemical interactions between amino acids
- Cellular conditions such as temperature and pH
- Assistance from specialized molecules called molecular chaperones
Correct folding is essential because the protein’s structure determines its function.
If folding occurs incorrectly, the protein may lose its biological activity.
Protein Misfolding and Disease
Improper protein folding can cause serious biological problems.
Misfolded proteins may:
- Lose normal function
- Aggregate into harmful structures
- Disrupt cellular processes
Protein misfolding has been linked to several diseases, including:
- Neurodegenerative disorders
- Certain metabolic conditions
- Some genetic diseases
Understanding protein structure helps scientists study how these conditions develop and how they might be treated.
Methods for Studying Protein Structure
Scientists use several advanced techniques to analyze protein structure at the molecular level.
Common methods include:
X-ray Crystallography
This technique determines protein structure by analyzing how X-rays scatter when they pass through protein crystals.
It provides highly detailed structural information.
Nuclear Magnetic Resonance (NMR) Spectroscopy
NMR spectroscopy studies proteins in solution and reveals information about their structure and dynamics.
Cryo-Electron Microscopy
Cryo-electron microscopy allows researchers to visualize large protein complexes at near-atomic resolution without crystallization.
These techniques have greatly expanded our understanding of biological molecules.
Importance of Protein Structure in Biochemistry
Studying protein structure helps scientists understand how biological systems operate at the molecular level.
Applications include:
- Drug design and pharmaceutical development
- Enzyme engineering
- Biotechnology innovation
- Disease research
- Synthetic biology
By analyzing protein structure, researchers can develop therapies, improve industrial enzymes, and design new biological technologies.
Final Thoughts
Protein structure is a foundational concept in biochemical systems. From the sequence of amino acids to the complex three-dimensional shapes formed by folding and subunit interactions, every level of structure contributes to a protein’s biological role.
Understanding these structural levels allows scientists to explore how living systems function, how diseases arise from molecular changes, and how new therapies and technologies can be developed.
As research continues, the study of protein structure remains central to advancements in biochemistry, medicine, and biotechnology.




