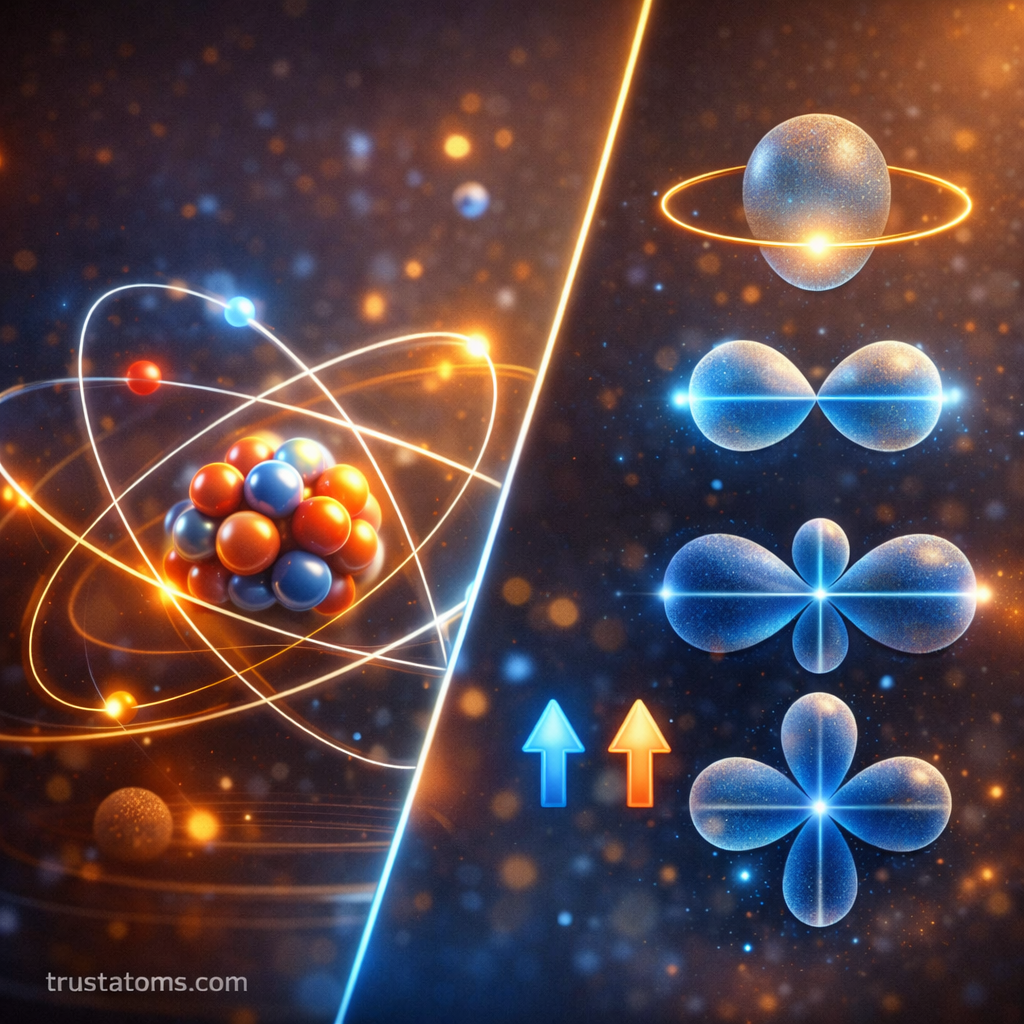
Understanding atoms requires more than simply knowing how many electrons they contain. In quantum mechanics, quantum numbers are used to describe the exact state and position of electrons within an atom. These numbers determine how electrons behave, where they are likely to be found, and how atoms interact with each other.
Quantum numbers are essential for understanding atomic structure, chemical bonding, spectroscopy, and modern chemistry. Every electron in an atom is described by a unique set of four quantum numbers.
This guide explains what quantum numbers are, what each number represents, and why they are critical in atomic theory.
What Are Quantum Numbers?
Quantum numbers are mathematical values that describe the properties and energy of electrons in atoms. They originate from solutions to the Schrödinger equation, which models how electrons behave as probability waves rather than fixed particles.
Each electron in an atom is defined by four quantum numbers:
- Principal quantum number (n)
- Angular momentum quantum number (l)
- Magnetic quantum number (mₗ)
- Spin quantum number (mₛ)
Together, these numbers specify an electron’s energy level, orbital shape, orientation, and spin.
One important rule in quantum mechanics is the Pauli Exclusion Principle, which states:
- No two electrons in the same atom can have the exact same set of four quantum numbers.
This rule helps determine how electrons fill orbitals and explains the structure of the periodic table.
The Principal Quantum Number (n)
The principal quantum number (n) describes the main energy level of an electron and its average distance from the nucleus.
Possible values:
n = 1, 2, 3, 4, …
Key ideas about the principal quantum number:
- Larger values of n mean higher energy levels
- Electrons with higher n values are farther from the nucleus
- Each energy level contains multiple orbitals
Examples of energy levels:
- n = 1 → first shell (closest to nucleus)
- n = 2 → second shell
- n = 3 → third shell
As n increases, orbitals become larger and electrons have greater potential energy.
The Angular Momentum Quantum Number (l)
The angular momentum quantum number (l) determines the shape of an electron’s orbital.
Possible values depend on the principal quantum number:
l = 0 to (n − 1)
For example:
- If n = 1 → l = 0
- If n = 2 → l = 0 or 1
- If n = 3 → l = 0, 1, or 2
Each value of l corresponds to a specific orbital type.
| l Value | Orbital Type | Shape |
|---|---|---|
| 0 | s | spherical |
| 1 | p | dumbbell-shaped |
| 2 | d | clover-shaped |
| 3 | f | complex multi-lobed |
Examples:
- 2p orbital → n = 2, l = 1
- 3d orbital → n = 3, l = 2
These orbital shapes determine how atoms bond and interact chemically.
The Magnetic Quantum Number (mₗ)
The magnetic quantum number (mₗ) describes the orientation of an orbital in space.
Possible values:
mₗ = −l to +l
Examples:
If l = 1 (p orbital):
mₗ can be:
- −1
- 0
- +1
This corresponds to the three p orbitals:
- pₓ
- pᵧ
- p_z
If l = 2 (d orbital):
mₗ values are:
- −2, −1, 0, +1, +2
This means five possible d orbitals.
The magnetic quantum number explains why orbitals exist in multiple spatial orientations.
The Spin Quantum Number (mₛ)
The spin quantum number (mₛ) describes the intrinsic spin of an electron.
Electrons behave like tiny magnets due to their spin.
Possible values:
mₛ = +½ or −½
Key rules:
- Each orbital can hold two electrons
- These electrons must have opposite spins
This rule arises from the Pauli Exclusion Principle.
Example:
A single orbital can contain:
- one electron with spin +½
- one electron with spin −½
Opposite spins allow electrons to occupy the same orbital without violating quantum rules.
How Quantum Numbers Work Together
Each electron in an atom is described by a unique combination of the four quantum numbers.
Example:
An electron in a 2p orbital could have:
- n = 2
- l = 1
- mₗ = 0
- mₛ = +½
Another electron in the same orbital would differ in spin:
- n = 2
- l = 1
- mₗ = 0
- mₛ = −½
This system ensures that electrons occupy orbitals in a structured way.
Quantum Numbers and Electron Configuration
Quantum numbers help explain how electrons fill orbitals in atoms.
Three important rules govern electron placement:
1. Aufbau Principle
Electrons fill lowest-energy orbitals first.
Example order:
1s → 2s → 2p → 3s → 3p → 4s → 3d
2. Pauli Exclusion Principle
- No two electrons share the same four quantum numbers.
- Maximum two electrons per orbital.
3. Hund’s Rule
Electrons occupy empty orbitals before pairing up.
Example in p orbitals:
Instead of:
↑↓ ↑ _
Electrons fill as:
↑ ↑ ↑
before pairing occurs.
Example: Quantum Numbers for a Carbon Electron
Carbon has 6 electrons.
Electron configuration:
1s² 2s² 2p²
For one of the 2p electrons:
Possible quantum numbers could be:
- n = 2
- l = 1
- mₗ = −1
- mₛ = +½
Another electron might be:
- n = 2
- l = 1
- mₗ = 0
- mₛ = +½
The different orientations explain how electrons distribute themselves within orbitals.
Why Quantum Numbers Matter in Chemistry
Quantum numbers are foundational to modern chemistry and physics.
They help scientists understand:
- atomic structure
- electron behavior
- chemical bonding
- spectroscopy
- magnetic properties of materials
- periodic table organization
Without quantum numbers, it would be impossible to explain why elements behave differently or why atoms form molecules in predictable ways.
They also play a critical role in fields like quantum chemistry, materials science, and semiconductor physics.
Final Thoughts
Quantum numbers provide a precise way to describe electrons inside atoms. By defining energy levels, orbital shapes, spatial orientation, and electron spin, they form the backbone of modern atomic theory.
Every electron in every atom has a unique set of these four numbers, allowing scientists to understand and predict the structure of matter.
From chemical bonding to modern electronics, quantum numbers help explain how the microscopic world shapes the physical universe.