
Photochemical reactions occur when molecules absorb light and undergo chemical transformations. These reactions are fundamental in processes ranging from photosynthesis and atmospheric chemistry to solar energy technologies and advanced materials science.
One of the most important concepts used to evaluate the efficiency of photochemical reactions is quantum yield. This measurement helps scientists understand how effectively absorbed light leads to chemical change. By studying quantum yield, chemists can determine how efficiently light energy drives molecular reactions.
What Is Quantum Yield?
Quantum yield is a measure of the efficiency of a photochemical process. It describes the ratio between the number of molecules that undergo a specific reaction and the number of photons absorbed by the system.
In simple terms, quantum yield answers the question:
How many chemical events occur for each photon of light absorbed?
If every absorbed photon leads to a chemical reaction, the quantum yield is equal to one. If fewer reactions occur than photons absorbed, the quantum yield will be less than one.
Why Quantum Yield Matters
Quantum yield plays a critical role in understanding photochemical processes because it quantifies reaction efficiency.
Scientists use quantum yield measurements to:
- Evaluate the effectiveness of photochemical reactions
- Compare different reaction pathways
- Optimize light-driven chemical systems
- Improve photochemical technologies
High quantum yields indicate that absorbed light energy is being used efficiently to drive chemical change.
How Quantum Yield Is Defined
Quantum yield is defined as the ratio of the number of events occurring to the number of photons absorbed.
These events may include:
- Molecules reacting chemically
- Fluorescence emission
- Phosphorescence emission
- Molecular dissociation
- Formation of excited states
Because different processes may occur after light absorption, a system can have different quantum yields for different outcomes.
Types of Quantum Yield in Photochemistry
In photochemical systems, several types of quantum yield are commonly studied depending on the process being analyzed.
Photochemical Reaction Quantum Yield
This describes how efficiently absorbed photons produce chemical reactions.
For example:
- Photodecomposition of molecules
- Formation of photochemical products
- Light-driven catalytic reactions
A high value indicates efficient conversion of light energy into chemical transformation.
Fluorescence Quantum Yield
Fluorescence quantum yield measures the efficiency of light emission following photon absorption.
In fluorescence:
- A molecule absorbs light.
- It enters an excited electronic state.
- It releases energy by emitting a photon.
Fluorescence quantum yield compares the number of emitted photons to the number absorbed.
Phosphorescence Quantum Yield
Phosphorescence is similar to fluorescence but occurs through a different electronic transition.
This process involves:
- Longer excited-state lifetimes
- Energy transitions through intermediate states
- Slower emission of light
Phosphorescence quantum yields are typically lower than fluorescence yields due to energy losses.
Factors That Influence Quantum Yield
Several factors influence how efficiently a photochemical reaction converts light energy into chemical change.
Molecular Structure
The structure of a molecule strongly affects how it absorbs and redistributes energy.
Important factors include:
- Electronic configuration
- Bond strength
- Molecular geometry
- Presence of functional groups
These features determine how excited states behave after photon absorption.
Light Wavelength
Different molecules absorb light at specific wavelengths. If the wavelength does not match the molecule’s absorption characteristics, the reaction efficiency may decrease.
Proper wavelength selection is essential for maximizing quantum yield.
Solvent Environment
The surrounding solvent can influence molecular motion and energy transfer.
Solvent effects may include:
- Stabilization of excited states
- Collisional energy transfer
- Solvent polarity effects
These factors can either enhance or reduce reaction efficiency.
Temperature
Temperature affects molecular motion and energy dissipation.
Higher temperatures may:
- Increase nonradiative energy loss
- Promote competing reactions
- Influence excited-state lifetimes
These effects can change the measured quantum yield.
Quantum Yield Greater Than One
In some photochemical reactions, the quantum yield can exceed one. This may seem counterintuitive but occurs when chain reactions are triggered by a single photon.
In these cases:
- One photon initiates a reactive intermediate.
- That intermediate triggers additional reactions.
- Multiple product molecules are formed from a single photon event.
Such chain mechanisms can significantly amplify photochemical efficiency.
Measuring Quantum Yield
Experimental measurement of quantum yield requires careful control of light exposure and reaction conditions.
Typical steps include:
- Determining the number of photons absorbed by the system.
- Measuring the amount of product formed or light emitted.
- Comparing these values to calculate the reaction efficiency.
Scientists often use specialized instruments such as:
- Spectrophotometers
- Fluorometers
- Laser light sources
- Photochemical reactors
Accurate measurement ensures reliable evaluation of photochemical systems.
Applications of Quantum Yield
Quantum yield measurements are essential in many areas of science and technology.
Photosynthesis Research
Photosynthetic systems convert sunlight into chemical energy. Measuring quantum yield helps scientists evaluate how efficiently plants and algae perform this conversion.
Solar Energy Technologies
Photochemical efficiency is critical in solar energy systems such as:
- Solar cells
- Photocatalytic water splitting
- Light-driven fuel production
Optimizing quantum yield improves energy conversion efficiency.
Photodynamic Therapy
Medical treatments sometimes use light-activated compounds to destroy harmful cells. Quantum yield measurements help determine how effectively these compounds generate reactive species.
Environmental Chemistry
Sunlight drives many chemical reactions in the atmosphere. Studying quantum yield helps scientists understand:
- Pollutant degradation
- Ozone formation
- Atmospheric photochemistry
Competing Processes After Light Absorption
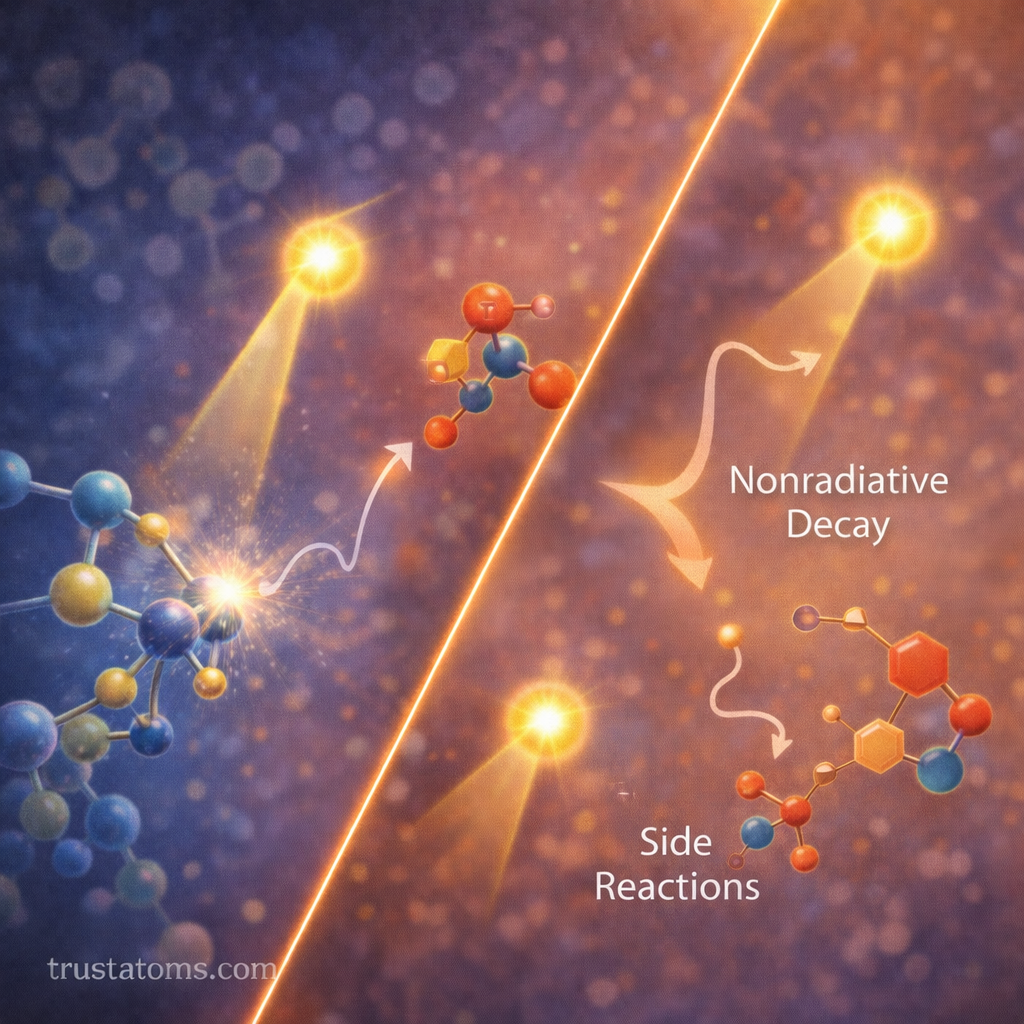
Not every absorbed photon leads to the desired reaction. Several competing processes may occur.
Common pathways include:
- Nonradiative relaxation
- Internal conversion
- Energy transfer
- Collisional quenching
- Photochemical side reactions
These processes reduce the fraction of photons that produce the desired outcome, lowering the overall quantum yield.
Improving Quantum Yield in Photochemical Systems
Scientists often attempt to improve quantum yield by controlling reaction conditions.
Strategies include:
- Selecting optimal light wavelengths
- Designing molecules with favorable excited states
- Using catalysts or sensitizers
- Controlling temperature and solvent environment
- Reducing competing energy loss pathways
These approaches help maximize the efficiency of photochemical reactions.
Final Thoughts
Quantum yield is a central concept in photochemistry because it quantifies how efficiently light energy drives chemical change. By measuring the relationship between absorbed photons and resulting chemical events, scientists can evaluate and optimize photochemical systems.
From biological photosynthesis to solar energy technologies and medical treatments, quantum yield provides valuable insight into how light interacts with molecules. As photochemical research continues to expand, understanding quantum yield remains essential for developing more efficient light-driven processes.




