Chemical reactions are rarely instantaneous. Instead, they pass through a series of energetic changes as reactants transform into products. Understanding this process is essential for predicting how fast reactions occur and why certain reactions proceed more easily than others.
Transition State Theory (TST) provides a framework for modeling these processes. Developed in the 1930s, it helps chemists estimate reaction rates by examining the short-lived, high-energy configuration known as the transition state. This theory plays a key role in fields such as chemical kinetics, catalysis, atmospheric chemistry, and enzyme reactions.
What Is Transition State Theory?
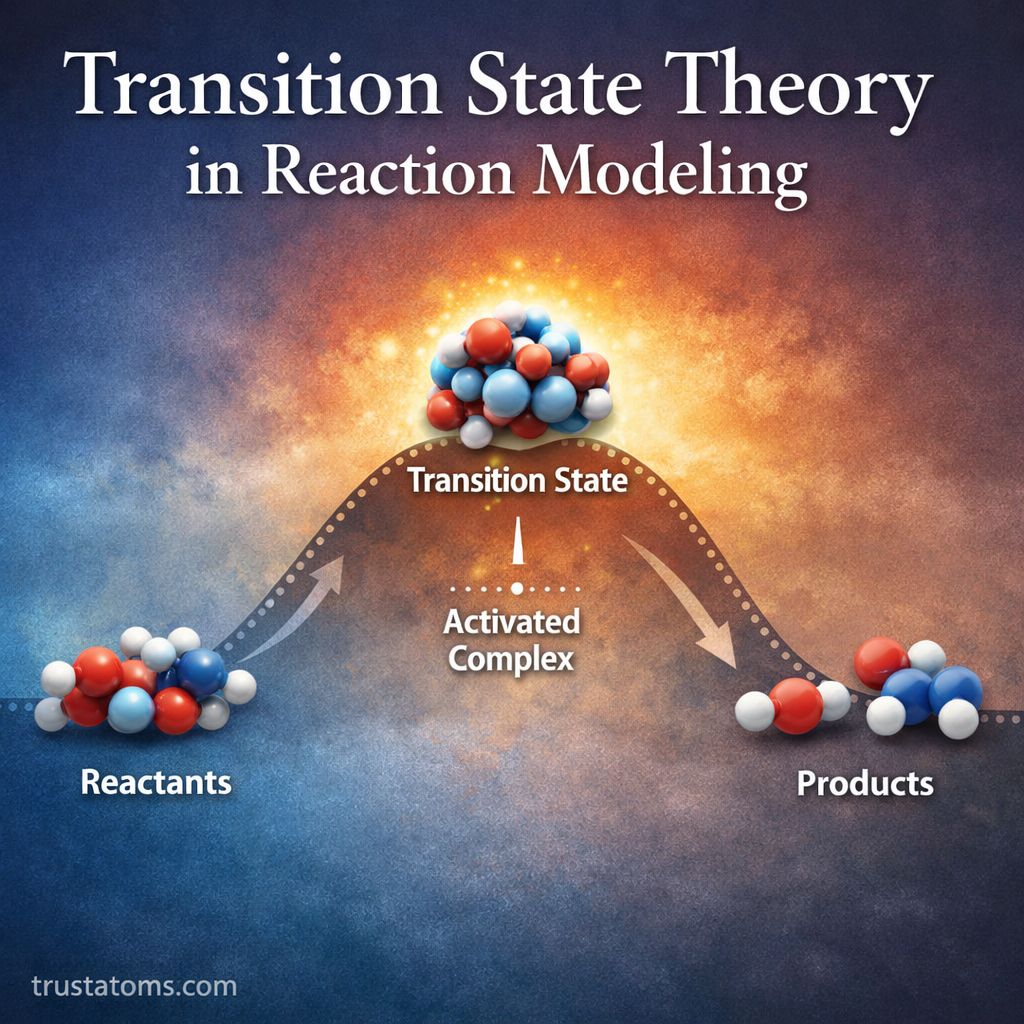
Transition State Theory describes how molecules move from reactants to products by passing through a high-energy intermediate configuration called the transition state.
The transition state represents the highest energy point along the reaction pathway. It is not a stable molecule but rather a temporary arrangement of atoms that exists for only a fraction of a second.
Key characteristics include:
- It occurs at the peak of the reaction energy barrier
- Bonds may be partially broken and partially formed
- It cannot be isolated or directly observed under normal conditions
- Its energy determines how fast the reaction proceeds
In reaction modeling, identifying this state allows chemists to calculate the probability that reactants will successfully convert into products.
The Concept of the Activated Complex

The structure formed at the transition state is often referred to as the activated complex.
This configuration represents a moment when molecules are rearranging their atomic structure. For example:
- In bond-breaking reactions, the bond may be stretched but not fully broken.
- In bond-forming reactions, atoms begin to connect but have not yet formed a stable bond.
The activated complex sits at the top of the energy barrier separating reactants from products.
Important properties of the activated complex include:
- Extremely short lifetime
- Higher potential energy than reactants or products
- Unstable and constantly rearranging atomic positions
Because it cannot be directly observed, scientists infer its structure using theoretical calculations and computational chemistry methods.
Energy Barriers and Activation Energy
A key concept in Transition State Theory is activation energy.
Activation energy is the minimum amount of energy required for reactants to reach the transition state. If molecules do not possess enough energy, they cannot overcome the barrier and the reaction will not occur.
The height of this barrier determines the reaction rate:
- Low activation energy → faster reactions
- High activation energy → slower reactions
Factors that influence activation energy include:
- Molecular structure
- Reaction environment
- Temperature
- Catalysts
Catalysts are especially important because they provide an alternative reaction pathway with a lower activation energy, allowing reactions to occur more quickly.
Reaction Coordinates and Energy Profiles
Transition State Theory is often visualized using a reaction coordinate diagram.
This diagram shows how the energy of a system changes as the reaction progresses from reactants to products.
Typical features include:
- Reactants – starting molecules with a certain energy level
- Rising energy curve – representing bond rearrangements
- Transition state peak – highest energy point in the reaction
- Descending curve – formation of products
- Products – final molecules with a new energy level
The difference between the reactants and the transition state corresponds to the activation energy.
These diagrams help chemists understand how reaction mechanisms work and where catalysts exert their influence.
The Transition State Theory Rate Expression
Transition State Theory provides a mathematical way to estimate reaction rates.
In simplified terms, the reaction rate depends on:
- How often molecules reach the transition state
- The probability that the activated complex proceeds to products rather than returning to reactants
The theory assumes that:
- Reactants and the activated complex exist in a quasi-equilibrium
- Once the activated complex forms, it quickly converts to products
This approach allows scientists to calculate reaction rate constants using thermodynamic and molecular parameters.
Assumptions Behind Transition State Theory
While extremely useful, Transition State Theory relies on several important assumptions.
1. Quasi-Equilibrium
Reactants and the activated complex are assumed to be in a temporary equilibrium. This simplifies the mathematics used to estimate reaction rates.
2. Single Reaction Pathway
The theory assumes that reactions proceed through one dominant pathway and one main transition state.
3. Immediate Product Formation
Once the activated complex forms, it is assumed to move forward into products rather than returning to reactants.
These assumptions make the model practical, though real chemical reactions can sometimes involve more complex dynamics.
Applications in Reaction Modeling
Transition State Theory is widely used in both theoretical and applied chemistry.
Common applications include:
Catalysis Research
Chemists analyze transition states to understand how catalysts lower activation energy and accelerate reactions.
Enzyme Mechanisms
Biochemists study transition states to determine how enzymes stabilize high-energy intermediates.
Atmospheric Chemistry
Reactions in the atmosphere, including pollutant formation and ozone chemistry, are often modeled using transition state calculations.
Computational Chemistry
Modern software can estimate transition state structures using quantum chemical methods, helping researchers predict reaction rates before experiments are performed.
How Catalysts Affect the Transition State
Catalysts speed up reactions by stabilizing the transition state.
Instead of increasing the energy of reactants, catalysts lower the peak energy required to reach the activated complex. This effectively reduces the activation energy barrier.
Catalysts can accomplish this by:
- Providing an alternative reaction pathway
- Orienting molecules into favorable positions
- Temporarily forming intermediate complexes
Enzymes in biological systems are particularly effective catalysts because they bind substrates in precise orientations that stabilize the transition state.
Limitations of Transition State Theory
Although widely used, Transition State Theory has limitations.
Some reaction systems behave differently than the theory predicts, especially when molecular motion becomes important.
Limitations include:
- Difficulty modeling extremely fast reactions
- Challenges with reactions involving multiple pathways
- Less accuracy in systems with strong molecular collisions
- Assumptions that may not hold for complex biological environments
To address these issues, researchers sometimes combine Transition State Theory with other approaches such as molecular dynamics simulations.
Why Transition State Theory Matters
Transition State Theory remains one of the most important tools for understanding chemical kinetics. By focusing on the highest energy point in a reaction, scientists gain insight into why reactions occur at certain speeds and how they can be controlled.
This understanding allows chemists to design better catalysts, predict reaction mechanisms, and model complex chemical systems. From industrial chemical production to biological enzyme reactions, the principles of Transition State Theory continue to shape modern chemistry.