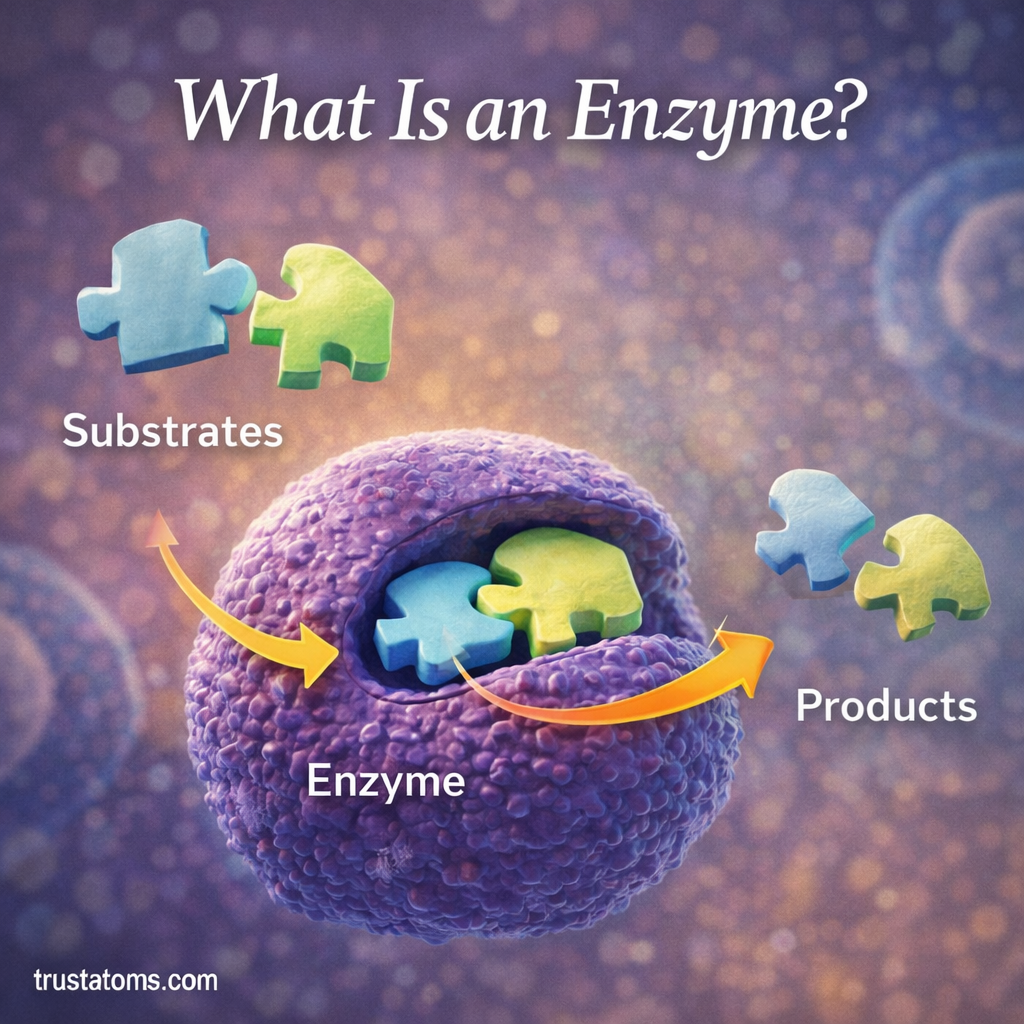
Enzymes are specialized biological molecules that make life possible by speeding up chemical reactions inside cells. Nearly every biological process—digestion, energy production, DNA replication, and cellular repair—depends on enzymes to occur efficiently.
In simple terms, an enzyme is a biological catalyst, meaning it accelerates chemical reactions without being consumed in the process. Most enzymes are proteins, although a small number are RNA molecules known as ribozymes.
Because chemical reactions inside cells would otherwise occur extremely slowly, enzymes are essential for maintaining life.
How Enzymes Work
Enzymes function by lowering the activation energy required for a chemical reaction to occur. Activation energy is the initial energy needed to start a reaction.
Without enzymes:
- Many biochemical reactions would take hours or days.
- Cells would not be able to maintain metabolism.
- Life processes would not occur fast enough to support living organisms.
By lowering activation energy, enzymes allow reactions to proceed rapidly under normal cellular conditions.
The Role of the Active Site
Each enzyme has a specific region called the active site, where the chemical reaction takes place.
Key characteristics of the active site include:
- A unique three-dimensional shape
- Chemical groups that interact with molecules
- A binding region for reactants
The molecules that interact with enzymes are called substrates.
When a substrate binds to the enzyme’s active site, an enzyme–substrate complex forms, allowing the reaction to proceed.
After the reaction occurs:
- The substrate is converted into products.
- The products leave the enzyme.
- The enzyme is free to catalyze another reaction.
The Lock-and-Key Model
One early model used to explain enzyme activity is the lock-and-key model.
In this model:
- The enzyme is the “lock.”
- The substrate is the “key.”
Only substrates with the correct shape fit into the enzyme’s active site.
This explains why enzymes are highly specific and usually catalyze only one type of reaction.
The Induced Fit Model
A more accurate explanation of enzyme behavior is the induced fit model.
In this model:
- The active site is flexible rather than rigid.
- When a substrate approaches, the enzyme changes shape slightly.
- This adjustment improves binding and increases reaction efficiency.
The induced fit model helps explain how enzymes stabilize reaction intermediates during chemical transformations.
Factors That Affect Enzyme Activity
Several environmental conditions influence how well enzymes function.
Temperature
Temperature affects enzyme activity in two ways:
- Increasing temperature speeds up molecular motion and reaction rates.
- Extremely high temperatures can cause enzyme denaturation, where the protein loses its shape.
Most human enzymes function best near 37°C (98.6°F).
pH Levels
Enzymes also depend on proper pH levels.
Different enzymes operate best at specific pH ranges.
Examples include:
- Pepsin in the stomach, which works best in acidic conditions
- Trypsin in the small intestine, which functions in a more alkaline environment
If the pH becomes too acidic or too basic, the enzyme structure can change and reduce activity.
Substrate Concentration
The amount of substrate available can affect enzyme reaction rates.
At low substrate levels:
- Reaction rates increase as substrate concentration increases.
At high substrate levels:
- Enzymes become saturated.
- The reaction rate reaches a maximum level.
This limit is known as the maximum reaction rate (Vmax).
Enzyme Concentration
The number of enzymes present also affects reaction speed.
More enzyme molecules mean:
- More active sites available
- More reactions occurring simultaneously
As a result, increasing enzyme concentration generally increases reaction rates, provided enough substrate is available.
Types of Enzymes
Thousands of different enzymes exist in biological systems, each performing specific tasks.
Scientists classify enzymes based on the reactions they catalyze.
Major enzyme categories include:
Oxidoreductases
These enzymes catalyze oxidation–reduction reactions involving electron transfer.
Examples include enzymes used in cellular respiration.
Transferases
Transferases move chemical groups from one molecule to another.
Examples include enzymes that transfer:
- Methyl groups
- Phosphate groups
- Amino groups
Hydrolases
Hydrolases break chemical bonds using water in hydrolysis reactions.
Common examples include digestive enzymes such as:
- Proteases
- Lipases
- Amylases
Lyases
Lyases break or form chemical bonds without using water or ATP.
These enzymes often participate in metabolic pathways.
Isomerases
Isomerases rearrange atoms within a molecule, converting it into an isomer.
This allows cells to reorganize molecules for metabolic processes.
Ligases
Ligases join two molecules together, often using energy from ATP.
These enzymes are essential in processes such as:
- DNA replication
- DNA repair
- Cellular metabolism
Enzymes in Metabolism
Metabolism refers to all chemical reactions occurring within living organisms.
These reactions fall into two categories:
Catabolism
Catabolic reactions break down large molecules into smaller ones.
Examples include:
- Digestion of food
- Breakdown of glucose during cellular respiration
Enzymes involved in catabolism release energy.
Anabolism
Anabolic reactions build complex molecules from smaller building blocks.
Examples include:
- Protein synthesis
- DNA replication
- Fat synthesis
These processes require energy and rely heavily on enzymes.
Enzyme Inhibition
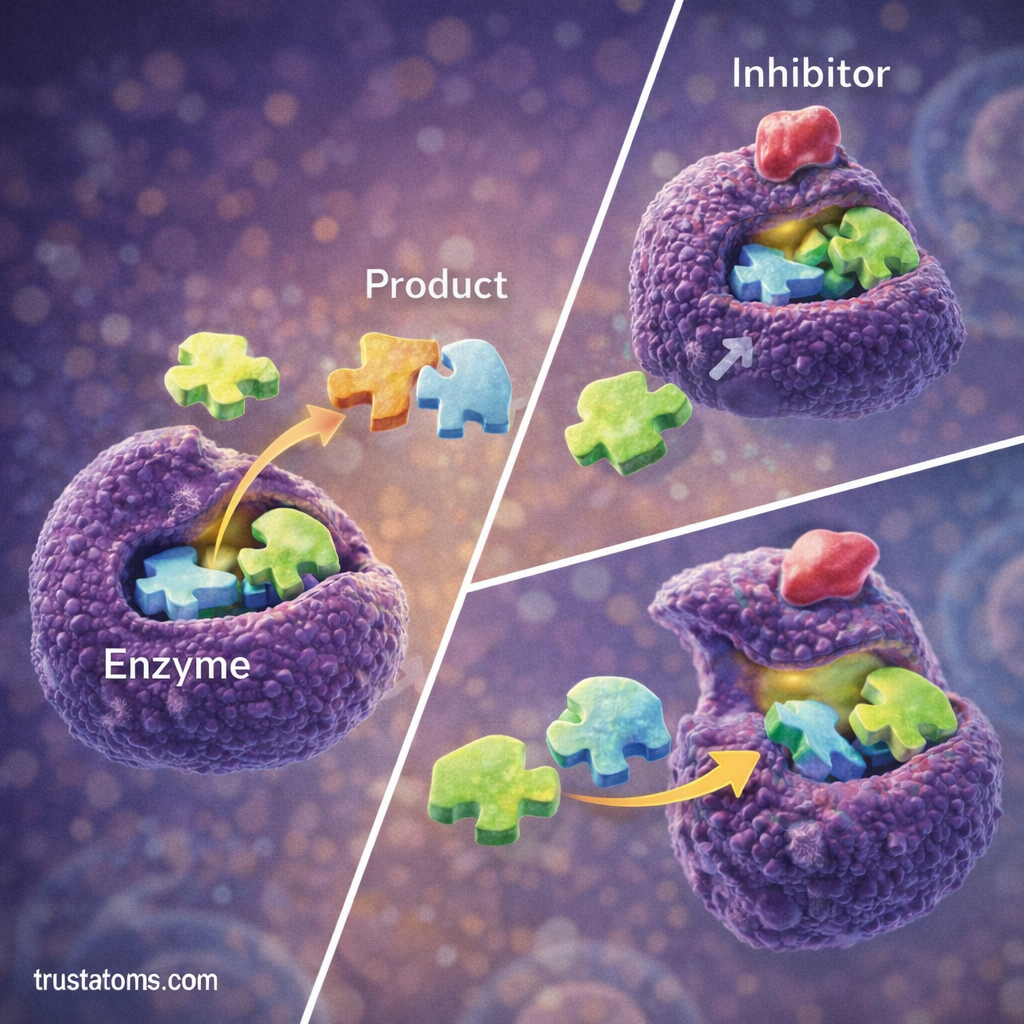
Enzyme activity can be reduced or stopped by molecules known as inhibitors.
Two major types of inhibition exist.
Competitive Inhibition
In competitive inhibition:
- The inhibitor competes with the substrate for the enzyme’s active site.
- If the inhibitor binds first, the reaction cannot occur.
Increasing substrate concentration can often overcome this inhibition.
Noncompetitive Inhibition
In noncompetitive inhibition:
- The inhibitor binds to a different location on the enzyme.
- This changes the enzyme’s shape and reduces its activity.
Because the active site changes shape, increasing substrate concentration does not restore full activity.
Enzymes in Everyday Life
Enzymes are not only important in biology but also widely used in industry and daily life.
Common applications include:
- Food production – fermentation in bread, cheese, and yogurt
- Detergents – enzymes that break down stains
- Medicine – diagnostic tests and drug development
- Biotechnology – genetic engineering and DNA analysis
- Agriculture – improving crop production and animal digestion
Modern biotechnology relies heavily on enzymes to manipulate DNA and produce biological products.
Why Enzymes Are Essential for Life
Enzymes are critical because they allow biological reactions to occur quickly and efficiently under normal conditions.
Without enzymes:
- Metabolic reactions would proceed too slowly.
- Cells could not generate energy efficiently.
- Growth and repair would be impossible.
From digestion to DNA replication, enzymes serve as the molecular machinery that powers life.
Final Thoughts
Enzymes are biological catalysts that drive the chemical reactions necessary for life. By lowering activation energy and binding specific substrates, enzymes allow cells to carry out complex biochemical processes efficiently.
Understanding enzymes helps scientists study metabolism, develop medicines, and design new biotechnology applications. As research continues, enzymes remain central to fields ranging from molecular biology to industrial biotechnology.