
Stoichiometry is the branch of chemistry that deals with quantitative relationships between substances in chemical reactions. In simple terms, it helps you calculate how much of a reactant is needed or how much product will be formed during a reaction.
If chemistry is the language of matter, stoichiometry is the math that keeps everything balanced.
Understanding stoichiometry allows scientists, engineers, pharmacists, and manufacturers to predict outcomes accurately, minimize waste, and design efficient chemical processes.
Stoichiometry Definition
Stoichiometry is the calculation of the relative amounts of reactants and products in a chemical reaction based on a balanced chemical equation.
It relies on three core ideas:
- The Law of Conservation of Mass
- The mole concept
- Balanced chemical equations
These principles allow chemists to determine exact proportions in chemical reactions.
Why Balanced Equations Matter
A chemical equation must be balanced before you can perform stoichiometric calculations.
For example:
Hydrogen + Oxygen → Water
When balanced, it becomes:
2H₂ + O₂ → 2H₂O
This shows:
- 2 molecules (or moles) of hydrogen react
- With 1 molecule (or mole) of oxygen
- To produce 2 molecules (or moles) of water
The coefficients (the numbers in front) give the mole ratios, which are essential for stoichiometry.
The Role of the Mole in Stoichiometry
The mole is a counting unit used in chemistry, similar to how a dozen represents 12 items.
One mole represents:
6.022 × 10²³ particles (Avogadro’s number)
Stoichiometry works because balanced equations describe relationships in terms of moles, not grams.
To perform calculations, chemists often convert:
- Grams → moles
- Use mole ratio from balanced equation
- Convert moles → grams (if needed)
The 4 Basic Types of Stoichiometry Calculations
1. Mole-to-Mole Calculations
These are the simplest.
You use the mole ratio directly from the balanced equation.
Example:
If 3 moles of hydrogen react, how many moles of water are formed?
Using the ratio 2:2 (from 2H₂ → 2H₂O), the relationship is 1:1.
So 3 moles of hydrogen produce 3 moles of water.
2. Mass-to-Mass Calculations
This is the most common type.
Steps:
- Convert grams of given substance to moles
- Use mole ratio
- Convert moles to grams of desired substance
3. Limiting Reactant Problems
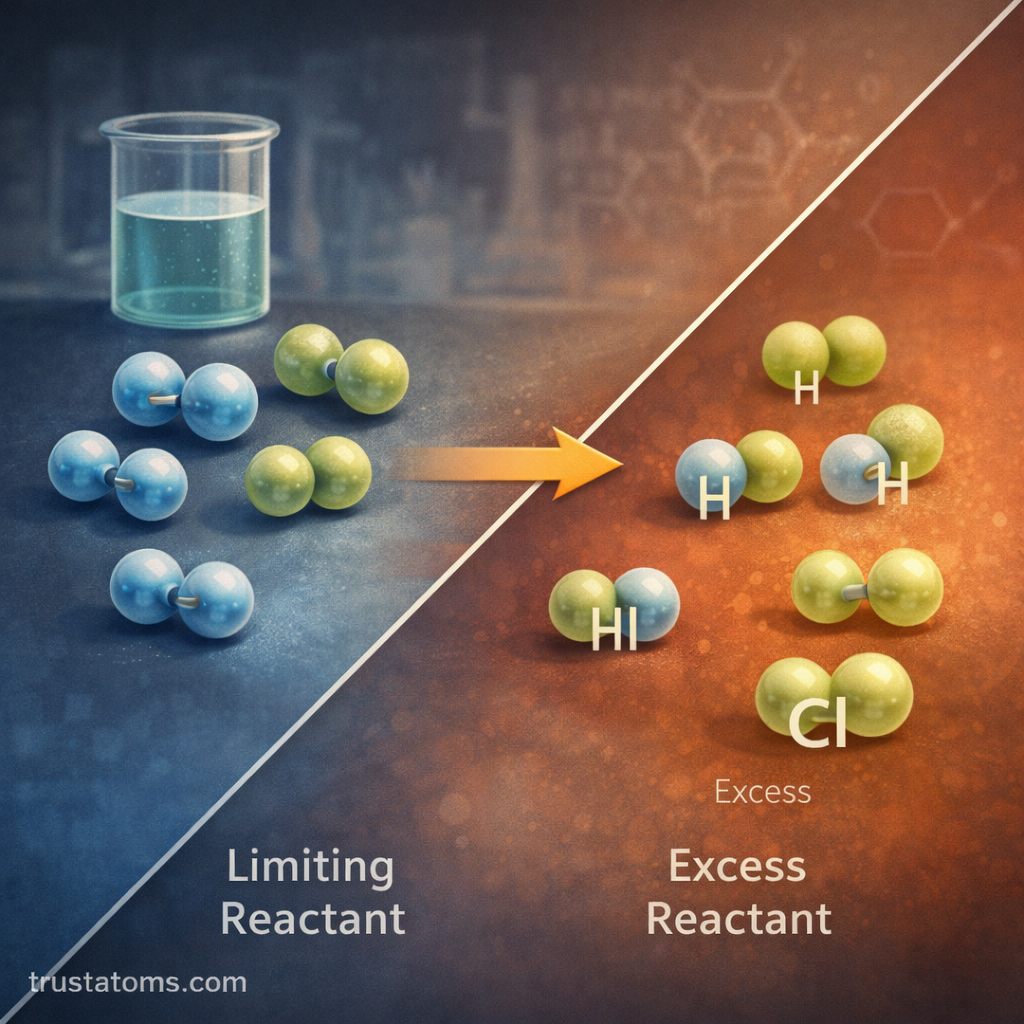
Sometimes one reactant runs out first. This is called the limiting reactant.
The limiting reactant:
- Determines how much product forms
- Stops the reaction once used up
The other reactant is called the excess reactant.
4. Percent Yield Calculations
In real experiments, reactions rarely produce 100% of the expected product.
Percent yield measures efficiency:
Percent Yield = (Actual Yield ÷ Theoretical Yield) × 100
A high percent yield means the reaction was efficient.
Real-World Applications of Stoichiometry
Stoichiometry is not just classroom math. It is essential in:
- Pharmaceutical drug production
- Environmental chemistry (pollution control)
- Food manufacturing
- Rocket fuel formulation
- Industrial chemical production
- Battery and energy systems
Without stoichiometry, chemical processes would be inefficient, wasteful, or even dangerous.
The Law of Conservation of Mass
Stoichiometry works because matter is conserved in chemical reactions.
The Law of Conservation of Mass states:
Matter cannot be created or destroyed in a chemical reaction.
That’s why:
- Atoms must balance
- Total mass of reactants equals total mass of products
Stoichiometry simply applies this principle using mathematical relationships.
Common Mistakes in Stoichiometry
Students often struggle with:
- Forgetting to balance the equation first
- Skipping unit conversions
- Mixing up mole ratios
- Using incorrect molar masses
- Rounding too early in calculations
Careful step-by-step work prevents most errors.
Why Stoichiometry Is Important in Chemistry
Stoichiometry is foundational to nearly every area of chemistry. It allows scientists to:
- Predict reaction outcomes
- Determine chemical quantities precisely
- Optimize industrial reactions
- Ensure laboratory safety
- Minimize material waste
From small lab experiments to massive chemical plants, stoichiometry ensures reactions are predictable and controlled.
Final Thoughts
Stoichiometry is the mathematical backbone of chemistry. By connecting balanced equations to real-world quantities, it allows us to measure, predict, and control chemical reactions with precision.
Once you understand mole ratios and unit conversions, stoichiometry becomes a powerful and practical tool — not just a classroom exercise, but a cornerstone of modern science and industry.




