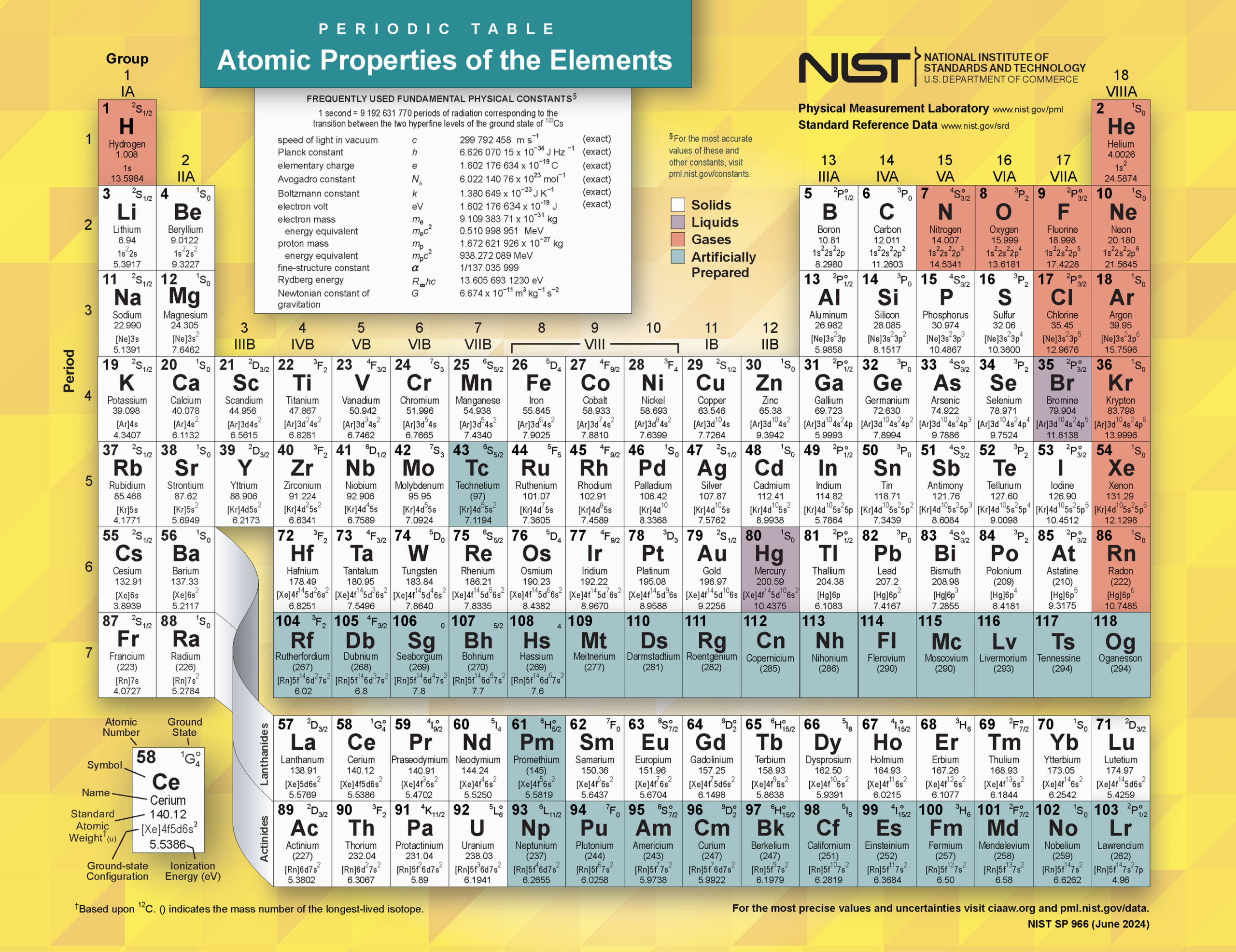
The periodic table is a structured chart that organizes all known chemical elements based on their atomic number, properties, and recurring patterns.
It is one of the most important tools in chemistry. Scientists use it to predict how elements behave, how they react, and how they combine to form compounds.
If you understand the periodic table, you understand the foundation of chemistry.
What Does the Periodic Table Show?
Each box on the periodic table represents one element.
Inside each element box, you typically find:
- Atomic number (number of protons)
- Chemical symbol (one or two letters)
- Element name
- Atomic mass
The elements are arranged in order of increasing atomic number, from hydrogen (1) to oganesson (118).
Why Is It Called “Periodic”?
The word periodic refers to repeating patterns.
When elements are arranged by atomic number, certain properties repeat at regular intervals. These repeating patterns are called periodic trends.
For example:
- Elements in the same column behave similarly.
- Reactivity patterns repeat across rows.
- Atomic size increases and decreases in predictable ways.
This repetition is what gives the table its name.
Rows and Columns: Periods and Groups

The periodic table is organized into rows and columns.
Periods (Rows)
Horizontal rows are called periods.
- There are 7 periods.
- Moving left to right across a period, atomic number increases.
- Elements gradually change from metals to nonmetals.
Groups (Columns)
Vertical columns are called groups or families.
- There are 18 groups.
- Elements in the same group have similar chemical properties.
- They have the same number of valence electrons.
Valence electrons largely determine how an element reacts.
Major Categories of Elements
The periodic table is divided into broad categories based on properties.
1. Metals
Metals are found on the left and center of the table.
Common properties:
- Good conductors of heat and electricity
- Shiny (lustrous)
- Malleable and ductile
Examples:
- Iron
- Copper
- Gold
2. Nonmetals
Nonmetals are located on the right side.
Common properties:
- Poor conductors of electricity
- Often gases or brittle solids
- Lower melting points (in many cases)
Examples:
- Oxygen
- Nitrogen
- Carbon
3. Metalloids
Metalloids lie along the “stair-step” line separating metals and nonmetals.
They have properties of both.
Examples:
- Silicon
- Boron
- Arsenic
Metalloids are important in electronics and semiconductors.
Important Groups on the Periodic Table
Some groups are especially significant in chemistry.
Alkali Metals (Group 1)
- Extremely reactive
- Soft metals
- React strongly with water
Examples: Lithium, sodium, potassium
Alkaline Earth Metals (Group 2)
- Reactive, but less than Group 1
- Found in many minerals
Examples: Magnesium, calcium
Halogens (Group 17)
- Very reactive nonmetals
- Often form salts with metals
Examples: Fluorine, chlorine
Noble Gases (Group 18)
- Very stable
- Rarely react
- Full outer electron shells
Examples: Helium, neon, argon
How the Periodic Table Was Developed
The modern periodic table evolved over time.
In 1869, Russian chemist Dmitri Mendeleev arranged elements by atomic mass and noticed repeating patterns. He even predicted the existence of elements that had not yet been discovered.
Later discoveries showed that atomic number — not atomic mass — is the true organizing principle.
Today’s periodic table is based on atomic number and electron configuration.
Periodic Trends: Patterns in Properties
The periodic table allows scientists to predict trends.
Some important trends include:
Atomic Radius
- Increases down a group
- Decreases across a period
Electronegativity
- Increases across a period
- Decreases down a group
Reactivity
- Metals become more reactive down Group 1
- Nonmetals become more reactive up Group 17
These trends explain why elements behave the way they do.
Why the Periodic Table Matters
The periodic table is not just a chart — it is a predictive tool.
It helps scientists:
- Predict chemical reactions
- Understand bonding behavior
- Design new materials
- Develop medicines
- Study environmental chemistry
Nearly every field of chemistry relies on it.
Summary: What Is the Periodic Table?
The periodic table is a structured chart that organizes elements by atomic number and repeating chemical properties.
It is arranged into:
- Periods (rows)
- Groups (columns)
- Metals, nonmetals, and metalloids
Because of its repeating patterns, the periodic table allows scientists to predict how elements will behave — making it one of the most powerful tools in science.
Image Source & Legal Reference
This image is the “Periodic Table of the Elements: Atomic Properties of the Elements” (NIST SP 966, June 2024) published by the National Institute of Standards and Technology (NIST), U.S. Department of Commerce.
NIST is a U.S. federal agency. Works created by U.S. government employees as part of their official duties are generally not subject to copyright protection under 17 U.S.C. § 105 and are in the public domain within the United States.
Source: National Institute of Standards and Technology (NIST)
Publication: NIST Special Publication 966 (June 2024)
Official site: https://www.nist.gov
Optional shorter footer version (if you want it minimal):
Source: National Institute of Standards and Technology (NIST), SP 966 (June 2024). Public domain (17 U.S.C. § 105).