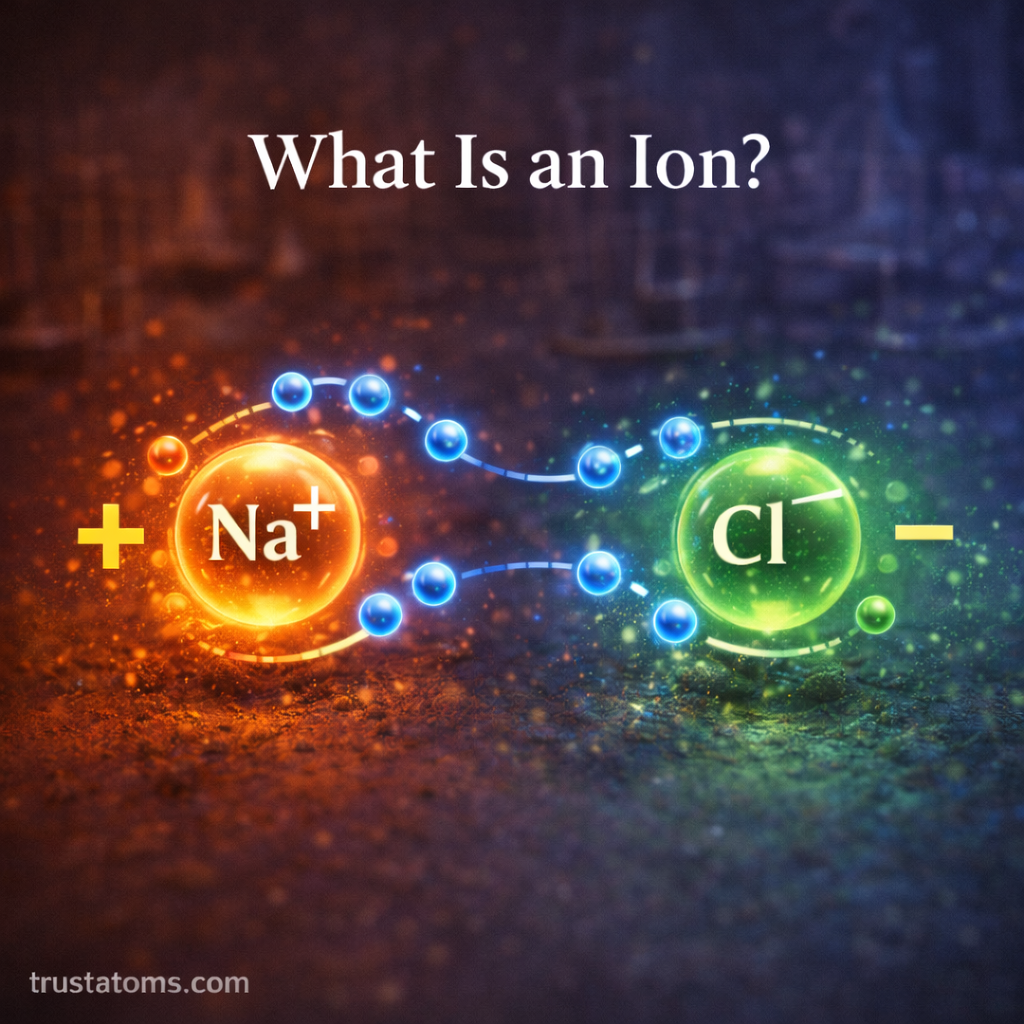A precipitation reaction is a type of chemical reaction in which two dissolved substances react to form a solid that separates from the solution. The solid that forms is called a precipitate.
These reactions commonly occur when two aqueous (water-based) solutions are mixed together and an insoluble compound forms. The solid particles appear cloudy at first and then settle at the bottom of the container.
Precipitation reactions are important in chemistry because they help scientists identify substances, remove impurities, and understand how compounds interact in solution.
Understanding the Basic Idea
A precipitation reaction occurs when ions in solution combine to form a compound that does not dissolve in water.
When this happens:
- Two solutions containing dissolved ions are mixed.
- Certain ions bond together to form a solid compound.
- The solid separates from the liquid and becomes visible.
The newly formed solid is the precipitate.
For example, if two clear solutions are mixed and suddenly become cloudy or form a solid deposit, a precipitation reaction has likely occurred.
What Is a Precipitate?
A precipitate is the solid product that forms during a precipitation reaction.
Characteristics of precipitates include:
- They form from dissolved substances.
- They are insoluble or only slightly soluble in water.
- They often appear as a cloudy suspension before settling.
Precipitates may look like:
- Fine powder
- Cloudy particles
- Solid crystals forming at the bottom of a container
The appearance of a precipitate is a clear visual sign that a chemical reaction has taken place.
How Precipitation Reactions Work
Many substances dissolve in water by breaking into charged particles called ions.
For example:
- Sodium chloride dissolves into sodium ions and chloride ions.
- Silver nitrate dissolves into silver ions and nitrate ions.
When two solutions are mixed, the ions from each solution can interact with each other.
If a pair of ions forms a compound that does not dissolve well in water, they combine to create a solid precipitate.
The process can be summarized like this:
- Two ionic compounds dissolve in water.
- Their ions move freely in the solution.
- Oppositely charged ions combine.
- An insoluble compound forms and falls out of the solution.
Simple Example of a Precipitation Reaction
One classic example occurs when silver nitrate solution is mixed with sodium chloride solution.
Before mixing:
- Both solutions are clear liquids.
- Each compound is dissolved into ions.
After mixing:
- Silver ions combine with chloride ions.
- A solid compound called silver chloride forms.
- A white precipitate appears in the solution.
This reaction is often used in chemistry demonstrations because the precipitate forms quickly and is easy to observe.
Double Replacement Reactions
Most precipitation reactions are also classified as double replacement reactions (also called double displacement reactions).
In these reactions:
- Two compounds exchange ions.
- New compounds are formed.
The general pattern looks like this:
Compound AB + Compound CD → Compound AD + Compound CB
One of the new compounds may be insoluble and forms a precipitate.
This type of reaction is very common in aqueous chemistry.
Solubility Rules and Precipitation

Chemists often use solubility rules to predict whether a precipitation reaction will occur.
Solubility rules are guidelines that describe which ionic compounds dissolve in water and which do not.
For example:
Compounds that usually dissolve in water:
- Nitrates
- Alkali metal salts
- Most ammonium compounds
Compounds that are often insoluble:
- Silver salts
- Lead salts
- Many sulfides and carbonates
If a reaction forms a compound that is insoluble according to these rules, a precipitate will likely form.
Real-World Applications of Precipitation Reactions
Precipitation reactions are used in many scientific and industrial processes.
Water Treatment
Water treatment plants use precipitation reactions to remove harmful substances from drinking water.
Chemicals are added to water to cause unwanted materials to form solids that can be filtered out.
Environmental Chemistry
Scientists use precipitation reactions to remove toxic metals from wastewater before releasing it into the environment.
This helps reduce pollution and protect ecosystems.
Chemical Analysis
Precipitation reactions help chemists identify unknown substances.
By adding specific reagents, chemists can observe whether a precipitate forms, helping determine the presence of certain ions.
Medicine and Pharmaceuticals
Some medicines rely on precipitation reactions to purify compounds or separate substances during manufacturing.
This ensures medications are produced with high purity.
Signs That a Precipitation Reaction Has Occurred
Several observations may indicate a precipitation reaction is happening.
Common signs include:
- A cloudy appearance forming in a previously clear solution
- Solid particles appearing in the liquid
- Sediment settling at the bottom of the container
- A visible color change due to the solid compound
These changes show that a new substance has formed.
Why Precipitation Reactions Are Important
Precipitation reactions help chemists understand how substances interact in solution.
They are important for:
- Identifying chemical compounds
- Separating mixtures
- Cleaning water and wastewater
- Manufacturing chemicals and medicines
- Studying reaction processes
Because these reactions produce visible changes, they are also commonly used in chemistry education and laboratory experiments.
Final Thoughts
A precipitation reaction occurs when two dissolved substances react to form a solid that separates from the solution. This solid, called a precipitate, forms when ions combine to create a compound that does not dissolve in water.
These reactions are widely used in laboratories, environmental science, water treatment, and industrial chemistry. By understanding precipitation reactions, scientists can better predict chemical behavior and design processes that improve safety, efficiency, and environmental protection.