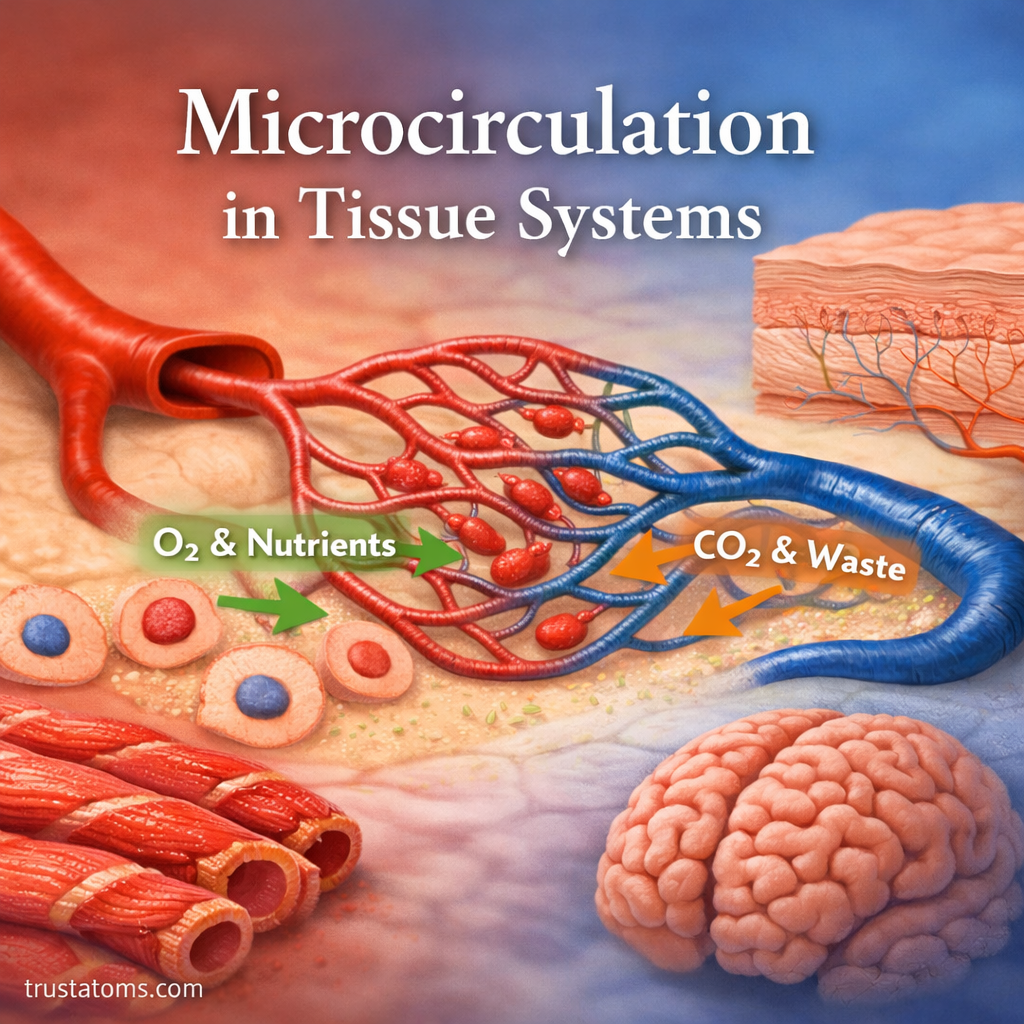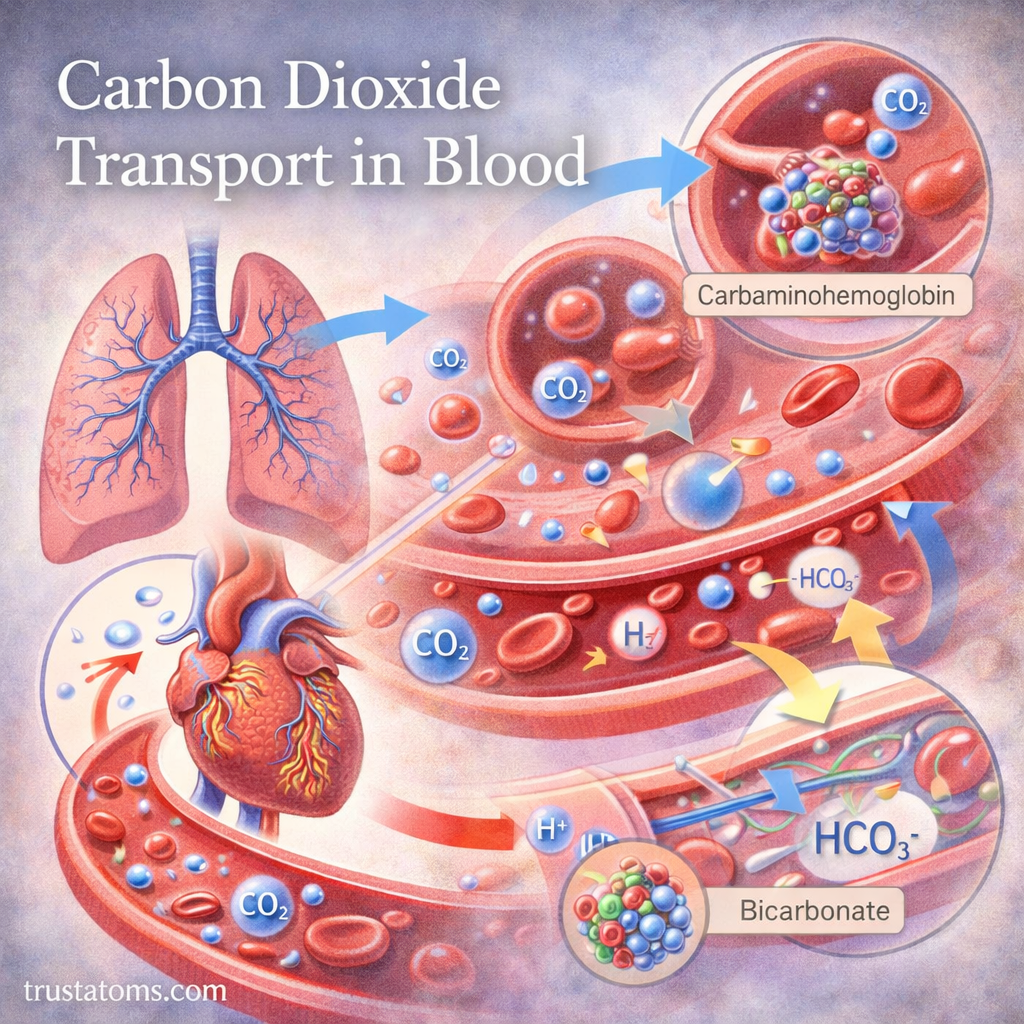
Carbon dioxide (CO₂) transport is a critical physiological process that helps maintain the body’s acid-base balance and supports efficient respiration. As cells produce carbon dioxide as a waste product of metabolism, it must be transported from tissues to the lungs for removal.
The body uses multiple mechanisms to move carbon dioxide through the bloodstream safely and efficiently.
What Is Carbon Dioxide?
Carbon dioxide is a waste gas produced during cellular respiration. When cells generate energy, they use oxygen and release carbon dioxide as a byproduct.
If CO₂ is not removed efficiently, it can accumulate and disrupt the body’s internal balance.
Why Carbon Dioxide Transport Is Important
Proper transport of carbon dioxide is essential for:
- Maintaining blood pH balance
- Supporting normal breathing
- Preventing toxic buildup of waste gases
- Ensuring efficient gas exchange in the lungs
Even small changes in CO₂ levels can significantly affect the body.
How Carbon Dioxide Is Transported in Blood
Carbon dioxide is transported in the blood in three main forms.
1. Dissolved in Plasma
A small portion of CO₂ dissolves directly in the blood plasma.
- Accounts for about 5–10% of total CO₂ transport
- Travels freely in the bloodstream
2. Bound to Hemoglobin (Carbaminohemoglobin)
Carbon dioxide can bind to hemoglobin in red blood cells.
- Forms carbaminohemoglobin
- Accounts for about 20–30% of CO₂ transport
This binding occurs at different sites than oxygen, allowing both gases to be carried simultaneously.
3. As Bicarbonate Ions (Primary Method)
Most carbon dioxide is transported as bicarbonate (HCO₃⁻).
- Accounts for about 60–70% of transport
- Occurs through a chemical reaction in red blood cells
The Conversion Process
- CO₂ enters red blood cells
- It reacts with water to form carbonic acid (H₂CO₃)
- Carbonic acid quickly dissociates into:
- Hydrogen ions (H⁺)
- Bicarbonate ions (HCO₃⁻)
This reaction is catalyzed by the enzyme carbonic anhydrase.
The Role of Hemoglobin
Hemoglobin plays a key role in carbon dioxide transport and regulation.
Buffering Hydrogen Ions
- Hemoglobin binds hydrogen ions produced during CO₂ conversion
- Helps maintain stable blood pH
Facilitating Gas Exchange
- When oxygen is released in tissues, hemoglobin more readily binds CO₂
- In the lungs, oxygen binding promotes CO₂ release
This interaction is known as the Haldane effect.
Gas Exchange in Tissues and Lungs
Carbon dioxide transport depends on efficient exchange at both ends of circulation.
In the Tissues
- Cells produce CO₂
- CO₂ diffuses into blood
- Conversion to bicarbonate occurs
In the Lungs
- Bicarbonate is converted back into CO₂
- CO₂ diffuses into alveoli
- CO₂ is exhaled
This reverse process ensures waste removal.
The Role of the Respiratory System
The lungs are responsible for eliminating carbon dioxide.
Ventilation
- Breathing rate adjusts based on CO₂ levels
- Increased CO₂ → faster breathing
- Decreased CO₂ → slower breathing
Gas Exchange
- CO₂ moves from blood into alveoli
- Oxygen moves into the blood
This exchange maintains proper gas balance.
Carbon Dioxide and Blood pH
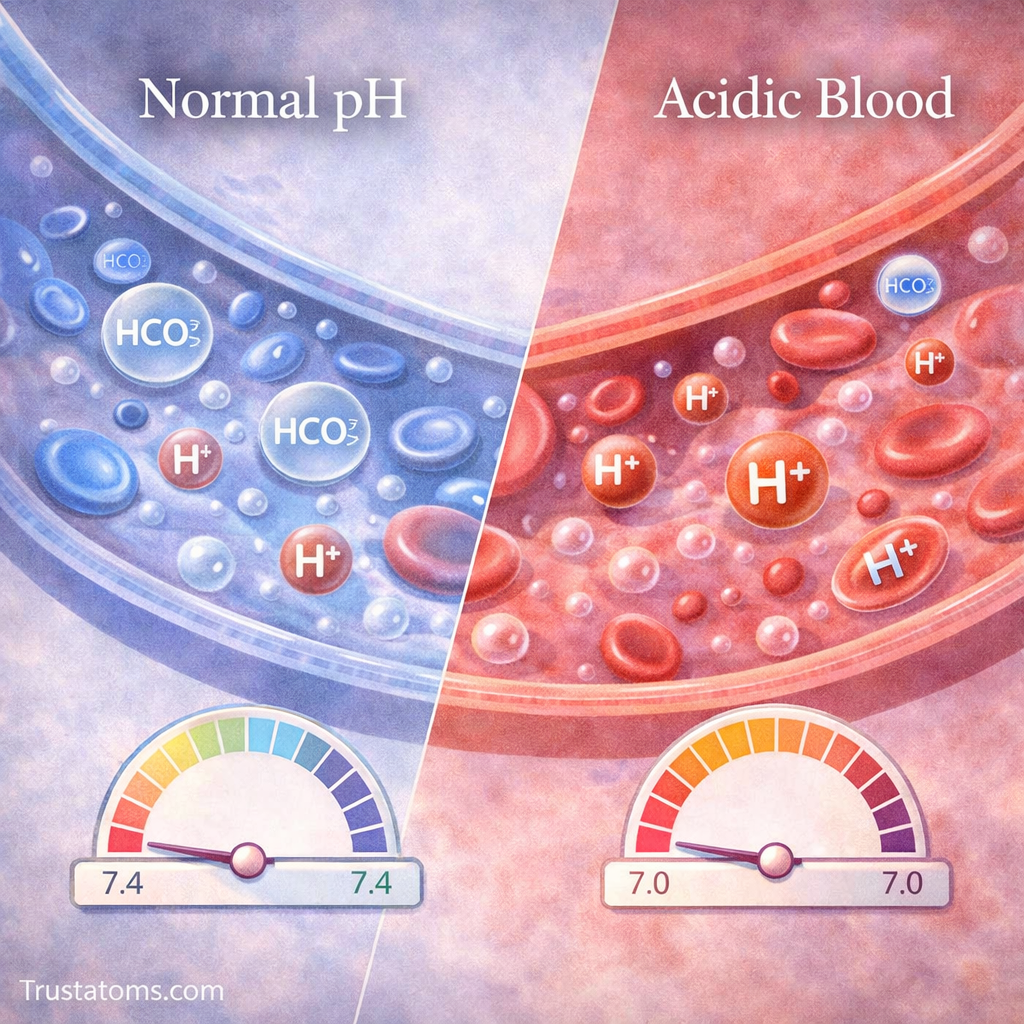
Carbon dioxide plays a major role in regulating blood acidity.
Acid-Base Balance
- Increased CO₂ → more hydrogen ions → lower pH (more acidic)
- Decreased CO₂ → higher pH (more alkaline)
The body tightly controls CO₂ levels to maintain a stable pH.
Factors That Affect Carbon Dioxide Transport
Several factors influence how effectively CO₂ is transported.
Physiological Factors
- Blood flow (circulation efficiency)
- Hemoglobin levels
- Enzyme activity (carbonic anhydrase)
Environmental and Health Factors
- Lung function
- Exercise level
- Respiratory diseases
Carbon Dioxide Transport During Exercise
Exercise increases the production of carbon dioxide.
Body’s Response
- Increased breathing rate
- Increased heart rate
- Enhanced CO₂ removal
Adaptation
With regular training:
- The body becomes more efficient at removing CO₂
- Respiratory and circulatory systems improve
Disorders Related to Carbon Dioxide Imbalance
Imbalances in CO₂ levels can affect health.
Hypercapnia (High CO₂ Levels)
- Caused by inadequate ventilation
- Symptoms: confusion, shortness of breath, fatigue
Hypocapnia (Low CO₂ Levels)
- Caused by excessive breathing (hyperventilation)
- Symptoms: dizziness, tingling, lightheadedness
How the Body Maintains CO₂ Balance
The body uses multiple systems to regulate carbon dioxide.
Key Control Systems
- Respiratory system (adjusts breathing)
- Nervous system (controls respiratory rate)
- Blood buffering systems
These systems work together to maintain equilibrium.
Tips for Supporting Healthy Gas Exchange
Maintaining healthy lungs and circulation supports effective CO₂ transport.
Practical Tips
- Stay physically active
- Practice deep breathing
- Avoid smoking
- Maintain good cardiovascular health
- Support lung health
Final Thoughts
Carbon dioxide transport in the blood is a vital process that ensures metabolic waste is removed and the body’s internal environment remains stable. Through a combination of dissolved transport, hemoglobin binding, and bicarbonate conversion, the body efficiently manages CO₂ levels.
Understanding this process highlights the importance of respiratory health, circulation, and balance in maintaining overall well-being.