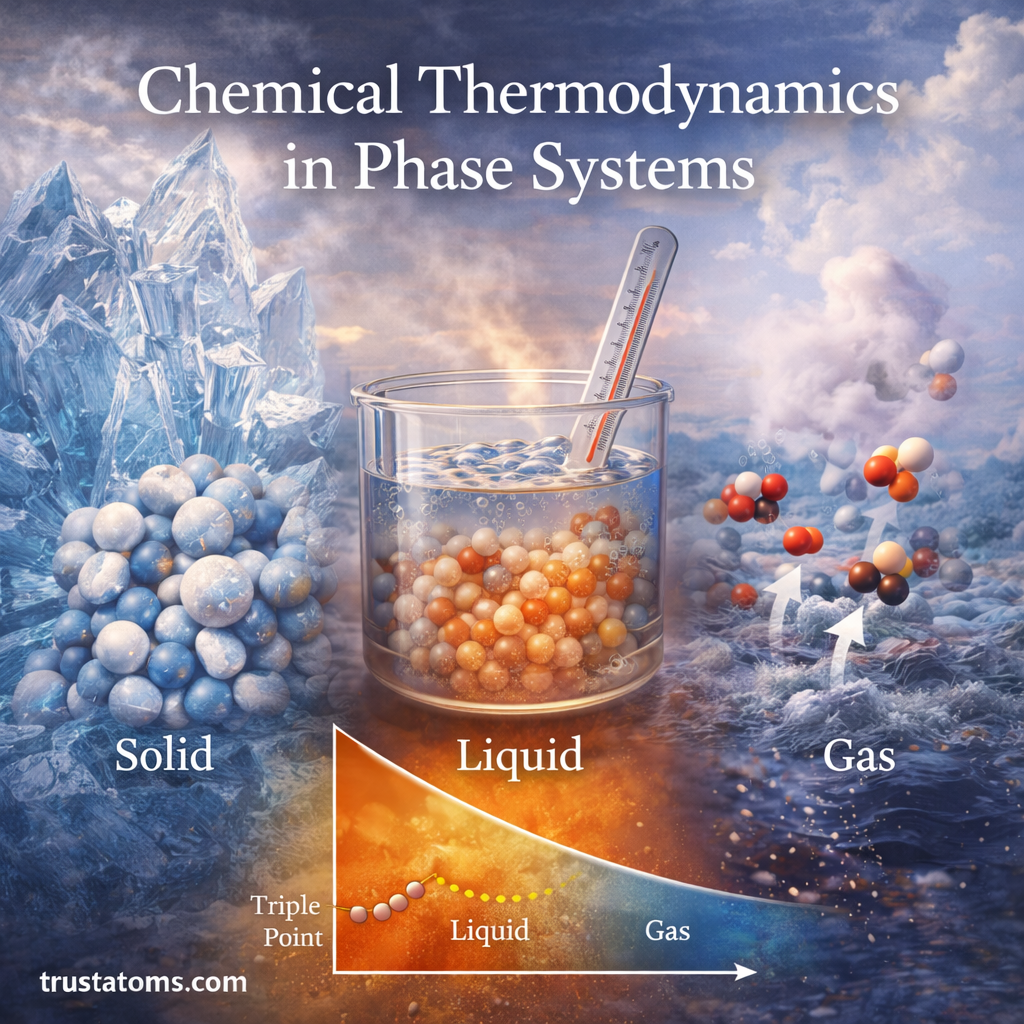
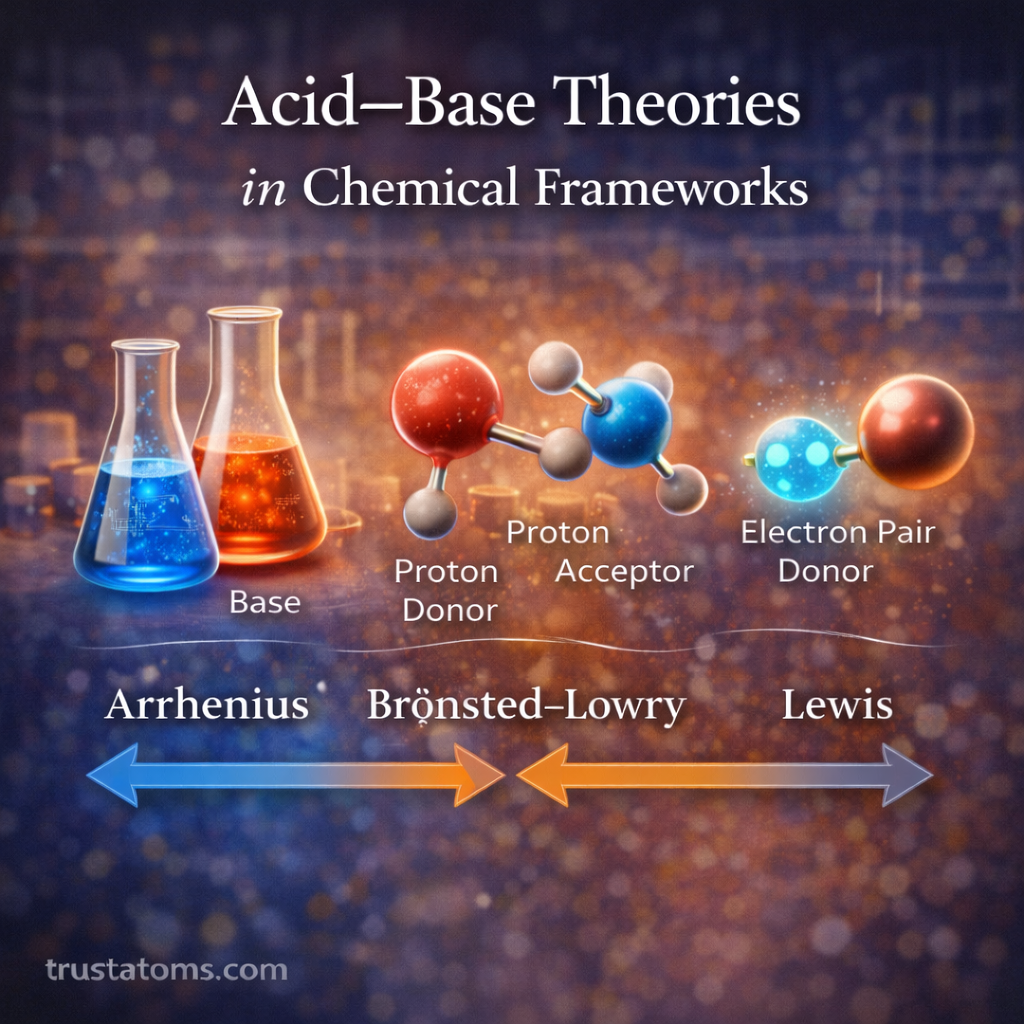
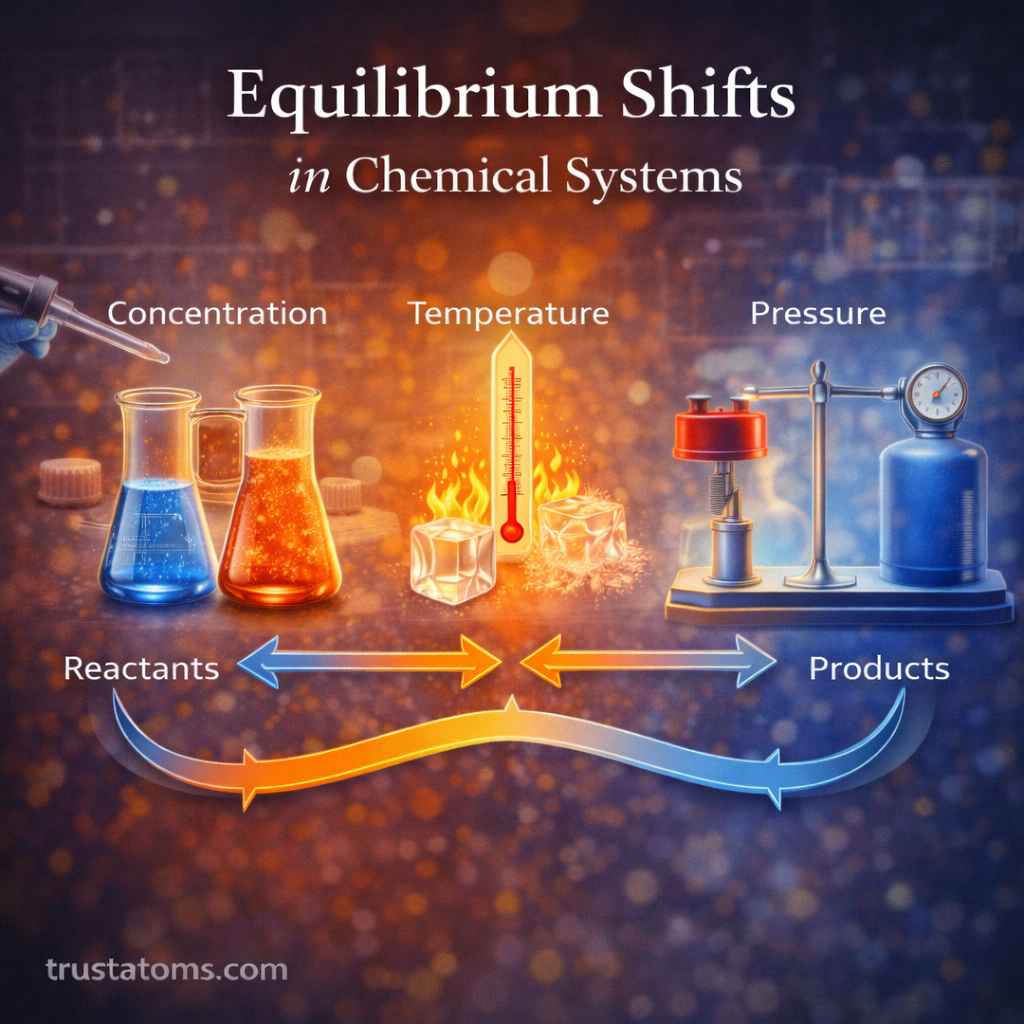
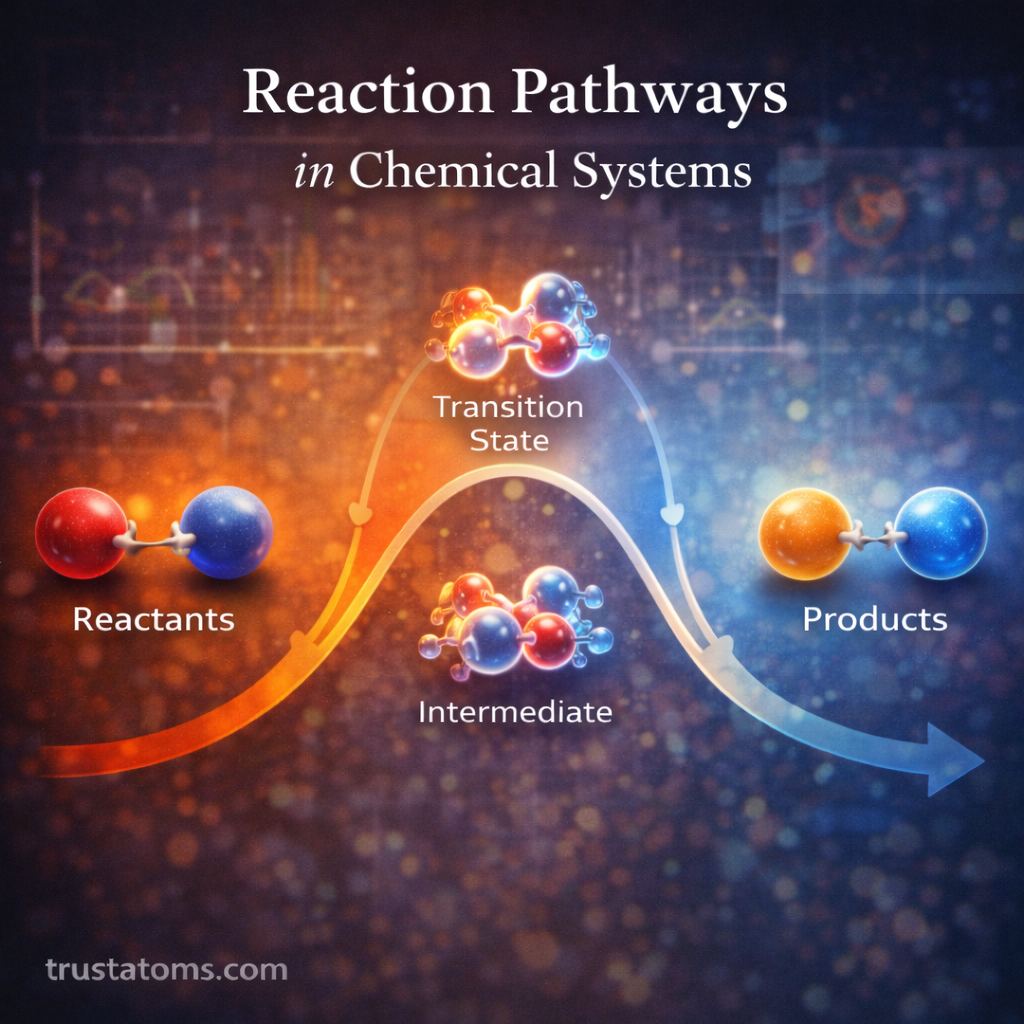
Chemical reactions often involve the transfer of electrons between substances. These reactions, known as oxidation–reduction (redox) reactions, are fundamental to chemistry because they drive processes such as corrosion, combustion, battery operation, and cellular respiration.
Balancing redox equations ensures that both mass and electrical charge are conserved during a chemical reaction. While balancing simple equations can be done by inspection, redox reactions typically require a structured method due to the movement of electrons.
Understanding how to balance redox equations helps chemists analyze reaction mechanisms, predict reaction outcomes, and apply chemistry principles in real-world systems.
Continue reading “Redox Balancing in Chemical Equations”