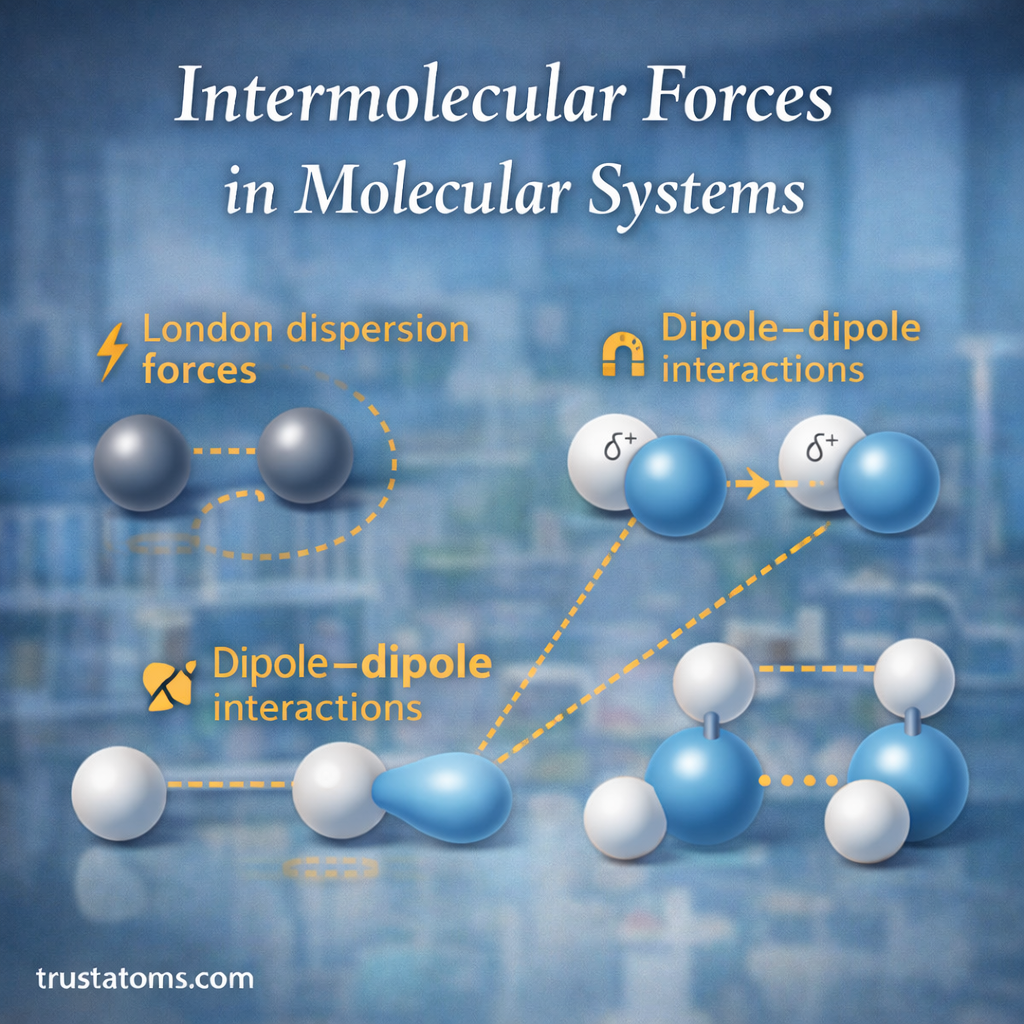
Hydrogen bonding is one of the most important intermolecular forces in chemistry. Although it is weaker than a covalent bond, hydrogen bonding has a major influence on how molecules behave, interact, and organize themselves.
From the structure of water to the shape of DNA, hydrogen bonds help determine the physical properties and stability of many chemical systems. Understanding hydrogen bonding is essential for studying molecular chemistry, biology, and materials science.
Continue reading “Hydrogen Bonding in Chemical Structures”