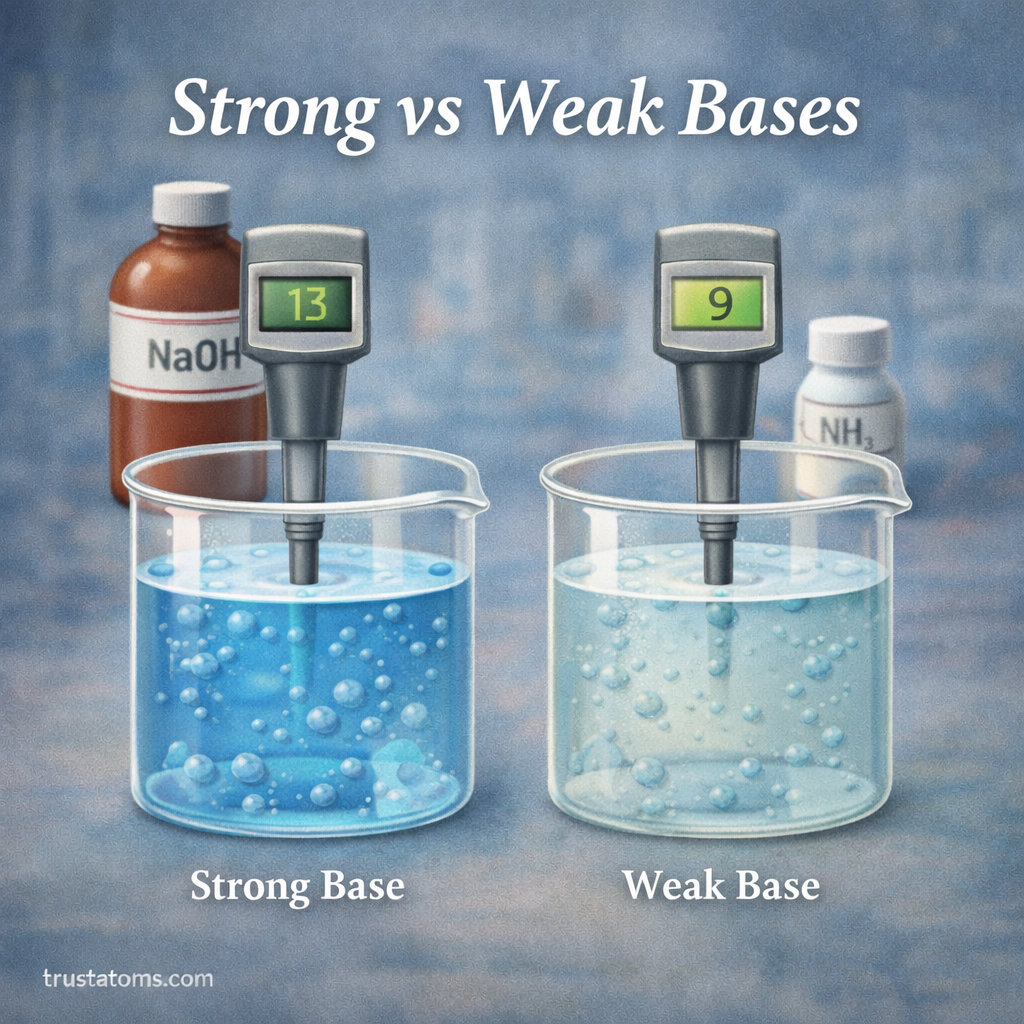
Bases are an important category of chemical substances that play a role in many natural and industrial processes. From cleaning products and food preparation to biological systems and chemical manufacturing, bases influence countless reactions.
One of the most important ways chemists classify bases is by their strength, which depends on how completely they produce hydroxide ions in water.
In chemistry, bases are commonly divided into strong bases and weak bases. Understanding the difference between them helps explain pH behavior, chemical reactions, and how substances interact in solutions.
What Is a Base?
A base is a substance that can produce hydroxide ions (OH⁻) when dissolved in water or accept hydrogen ions in a chemical reaction.
According to the Arrhenius definition:
- Acids produce hydrogen ions (H⁺) in water.
- Bases produce hydroxide ions (OH⁻) in water.
When a base dissolves in water, it may break apart into ions. The degree to which it does so determines whether the base is strong or weak.
What Determines Base Strength?
The strength of a base depends on how completely it ionizes in water.
- Strong bases completely dissociate into ions.
- Weak bases only partially dissociate.
This means strong bases release more hydroxide ions, which leads to higher pH values and stronger alkaline behavior.
Factors that influence base strength include:
- The compound’s molecular structure
- Stability of the ions formed
- Bond strength within the molecule
- Solubility in water
These factors determine how easily a substance can produce hydroxide ions.
What Are Strong Bases?
Strong bases are bases that fully dissociate in water, producing a high concentration of hydroxide ions.
Because nearly every molecule breaks apart into ions, strong bases create highly alkaline solutions.
Common Strong Bases
Most strong bases belong to the alkali metal hydroxides and some alkaline earth metal hydroxides.
Examples include:
- Sodium hydroxide (NaOH)
- Potassium hydroxide (KOH)
- Lithium hydroxide (LiOH)
- Calcium hydroxide (Ca(OH)₂)
- Barium hydroxide (Ba(OH)₂)
These substances are commonly used in industrial processes and laboratory chemistry.
Characteristics of Strong Bases
Strong bases typically have several recognizable properties:
- Complete ionization in water
- Very high pH values
- Strong electrical conductivity in solution
- Highly reactive with acids
- Often corrosive to organic materials and skin
Because of their strength, they must be handled with proper safety precautions.
What Are Weak Bases?
Weak bases are bases that only partially ionize in water. This means most of the base molecules remain intact, while only a small fraction forms hydroxide ions.
As a result, weak bases produce lower concentrations of hydroxide ions and have more moderate pH levels.
Common Weak Bases
Many common substances are weak bases.
Examples include:
- Ammonia (NH₃)
- Methylamine (CH₃NH₂)
- Aniline (C₆H₅NH₂)
- Pyridine (C₅H₅N)
These compounds are often used in biological chemistry and industrial reactions.
Characteristics of Weak Bases
Weak bases usually show the following characteristics:
- Partial ionization in water
- Moderate alkaline pH
- Reversible reactions with water
- Lower electrical conductivity than strong bases
- Often less corrosive than strong bases
Although weaker than strong bases, they still participate in many important chemical reactions.
Strong vs Weak Bases: Key Differences
The primary difference between strong and weak bases lies in their degree of ionization.
Ionization in Water
Strong bases:
- Dissociate completely
- Produce many hydroxide ions
Weak bases:
- Dissociate partially
- Produce fewer hydroxide ions
Chemical Equilibrium
Strong bases:
- Reaction proceeds almost entirely toward ion formation
Weak bases:
- Establish an equilibrium between molecules and ions
This equilibrium is described by the base dissociation constant (Kb), which measures the strength of a weak base.
pH Range
Strong bases typically produce:
- Very high pH values (often above 12)
Weak bases generally produce:
- Moderate pH values (often between 8 and 11)
However, pH also depends on the concentration of the base.
Why Some Bases Are Stronger Than Others
The strength of a base depends on how easily it can produce hydroxide ions or accept hydrogen ions.
Several chemical factors influence base strength.
Ion Stability
If the ions produced after dissociation are stable, the base is more likely to ionize completely.
Molecular Structure
The arrangement of atoms in a molecule can affect how easily the compound interacts with water to form hydroxide ions.
Solubility in Water
Some compounds may technically be strong bases but are limited by low solubility in water, which affects how much hydroxide ion is produced.
Real-World Examples of Strong and Weak Bases

Bases appear in many everyday environments, from household cleaning products to biological systems.
Household Products
Many cleaning products rely on strong bases to break down grease and organic materials.
Examples include:
- Drain cleaners (sodium hydroxide)
- Oven cleaners
- Industrial degreasers
These substances are effective but must be handled carefully due to their corrosive nature.
Biological Systems
Weak bases play important roles in biological chemistry.
For example:
- Ammonia helps regulate nitrogen cycles in nature
- Amines are essential components of many biological molecules
- Buffer systems in cells help maintain stable pH levels
These reactions allow organisms to maintain chemical balance.
Industrial Applications
Strong bases are widely used in manufacturing.
Examples include:
- Soap and detergent production
- Paper manufacturing
- Petroleum refining
- Chemical synthesis
Weak bases are also important in industrial chemistry, particularly in pharmaceutical development and organic reactions.
Bases in Acid–Base Reactions
Bases frequently react with acids in neutralization reactions.
In these reactions:
- An acid reacts with a base
- The products are water and a salt
Strong bases often react quickly and completely with strong acids, while weak bases participate in equilibrium reactions.
Understanding base strength helps chemists predict how these reactions will proceed.
Final Thoughts
Strong and weak bases differ primarily in how completely they produce hydroxide ions in water. Strong bases fully dissociate, creating highly alkaline solutions, while weak bases only partially ionize and establish equilibrium in solution.
These differences affect pH levels, chemical reactivity, and how bases behave in reactions. From industrial manufacturing to biological chemistry, understanding the distinction between strong and weak bases is essential for studying chemical processes and predicting reaction outcomes.