A chemical equation is a symbolic way of representing a chemical reaction. It shows the substances involved in the reaction and how they change into new substances.
Chemists use chemical equations to describe reactions clearly and concisely. Instead of writing long explanations, they use chemical formulas, symbols, and arrows to show how reactants transform into products.
Understanding chemical equations is essential for studying chemistry because they help explain how matter changes during reactions.
What Is a Chemical Equation?
A chemical equation is a written representation of a chemical reaction using chemical formulas and symbols.
It shows:
- The substances that start the reaction (reactants)
- The substances that form after the reaction (products)
- The direction of the reaction
The basic format of a chemical equation is:
Reactants → Products
The arrow indicates that the reactants undergo a reaction and form new substances.
Parts of a Chemical Equation
Chemical equations contain several key components that help describe what happens during a reaction.
Reactants
Reactants are the substances that start the chemical reaction.
They appear on the left side of the equation.
During the reaction, reactants are broken apart and rearranged to form new substances.
Example reactants might include:
- Hydrogen gas
- Oxygen gas
- Sodium metal
Products
Products are the new substances formed during the reaction.
They appear on the right side of the equation.
Products have different chemical properties from the reactants.
Examples of products include:
- Water
- Carbon dioxide
- Sodium chloride
Reaction Arrow
The arrow (→) in a chemical equation means “produces” or “yields.”
It shows the direction in which the reaction proceeds.
In reversible reactions, a double arrow (⇌) may be used to indicate that the reaction can occur in both directions.
Plus Signs
Plus signs (+) separate different substances in the equation.
They indicate that multiple reactants or products are involved.
Example:
Hydrogen + Oxygen → Water
Example of a Chemical Equation
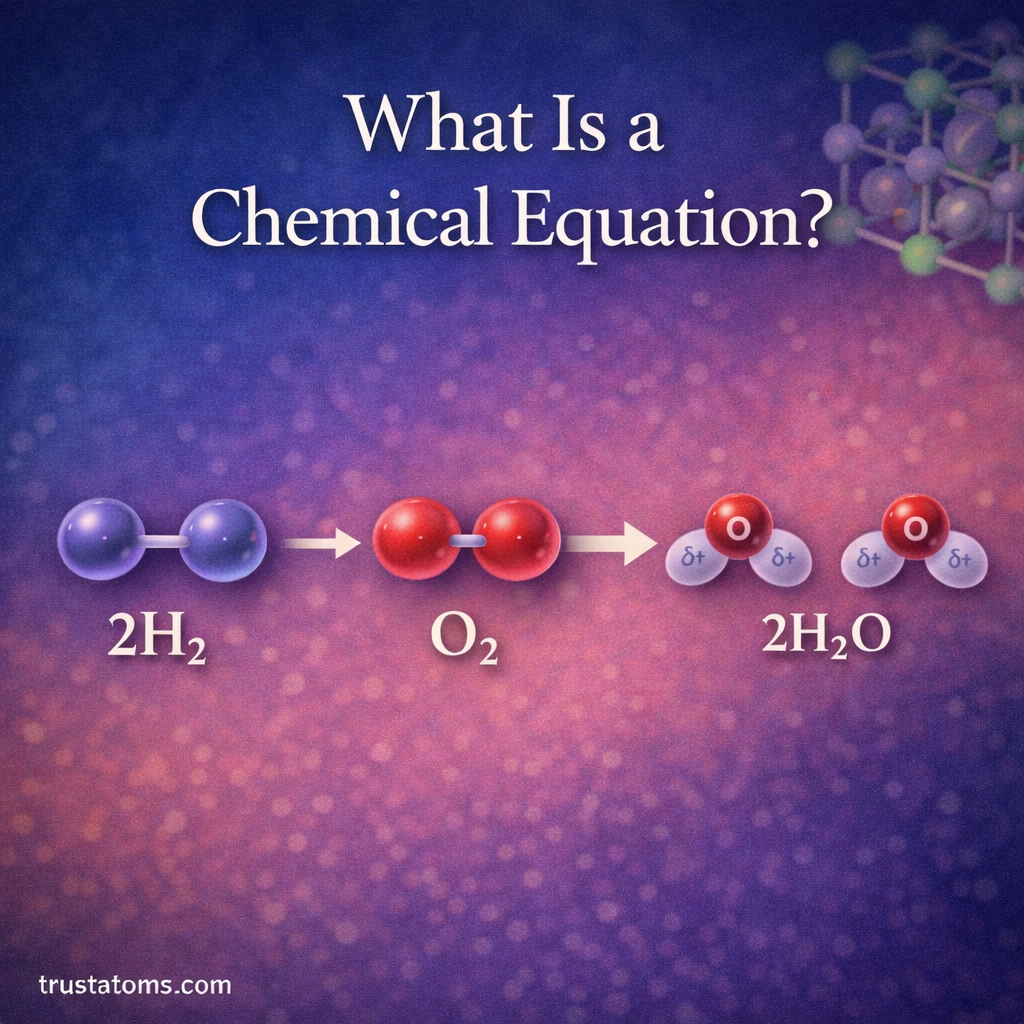
One of the most well-known chemical equations represents the formation of water.
Hydrogen reacts with oxygen to produce water.
This reaction can be written as:
2H₂ + O₂ → 2H₂O
This equation shows:
- Two hydrogen molecules reacting
- One oxygen molecule reacting
- Two water molecules forming
The numbers in front of formulas are called coefficients and indicate how many molecules participate in the reaction.
What Chemical Equations Show
Chemical equations communicate several important pieces of information about reactions.
They show:
- Which substances react
- Which substances form
- The relative amounts of each substance
- The direction of the reaction
However, chemical equations do not usually show:
- The speed of the reaction
- The reaction mechanism
- The physical conditions unless specifically indicated
Additional symbols may be added to show conditions such as heat or catalysts.
Types of Chemical Reactions in Equations
Chemical equations can represent many different kinds of reactions.
Here are some common types.
Synthesis Reactions
In a synthesis reaction, two or more substances combine to form a single product.
General form:
A + B → AB
Example:
Hydrogen + Oxygen → Water
These reactions build larger molecules from smaller ones.
Decomposition Reactions
In decomposition reactions, a single compound breaks down into simpler substances.
General form:
AB → A + B
Example:
Water → Hydrogen + Oxygen
These reactions often require energy input such as heat or electricity.
Single Replacement Reactions
In single replacement reactions, one element replaces another element in a compound.
General form:
A + BC → AC + B
Example:
Zinc + Hydrochloric acid → Zinc chloride + Hydrogen
Double Replacement Reactions
In double replacement reactions, parts of two compounds exchange places.
General form:
AB + CD → AD + CB
Example:
Silver nitrate + Sodium chloride → Silver chloride + Sodium nitrate
These reactions often produce a precipitate or solid product.
Balancing Chemical Equations
Chemical equations must follow the law of conservation of mass.
This law states that matter cannot be created or destroyed during a chemical reaction.
To satisfy this rule, equations must be balanced, meaning:
- The same number of each type of atom appears on both sides of the equation.
Balancing is done by adjusting coefficients in front of formulas.
Steps for Balancing Equations
- Write the unbalanced equation
- Count the atoms of each element on both sides
- Adjust coefficients to balance the atoms
- Verify that all elements are balanced
Balancing ensures the equation accurately reflects the reaction.
Symbols Used in Chemical Equations
Chemists often include additional symbols to provide more information about the reaction.
Common symbols include:
(s) — solid
(l) — liquid
(g) — gas
(aq) — aqueous solution (dissolved in water)
Other symbols may indicate conditions.
Examples:
Δ — heat added
→ — reaction direction
⇌ — reversible reaction
These symbols help chemists understand the physical state and conditions of the reaction.
Why Chemical Equations Are Important
Chemical equations are fundamental tools in chemistry because they help scientists understand and predict reactions.
They are used in many areas, including:
1. Predicting Reaction Products
Chemists can determine what substances will form during a reaction.
2. Calculating Quantities
Balanced equations allow chemists to calculate how much reactant is needed or how much product will form.
This process is called stoichiometry.
3. Industrial Chemistry
Chemical equations guide manufacturing processes for products such as:
- Medicines
- Fertilizers
- Fuels
- Plastics
4. Environmental Chemistry
Chemical equations help explain environmental processes like:
- Atmospheric reactions
- Water purification
- Pollution control
Understanding these reactions helps scientists protect ecosystems and public health.
Key Takeaways
Chemical equations are symbolic representations of chemical reactions.
Important points include:
- Reactants appear on the left side of the equation
- Products appear on the right side
- The arrow shows the direction of the reaction
- Coefficients indicate the number of molecules involved
- Balanced equations obey the law of conservation of mass
Learning to read and write chemical equations is a foundational skill in chemistry that helps scientists describe how substances transform during reactions.