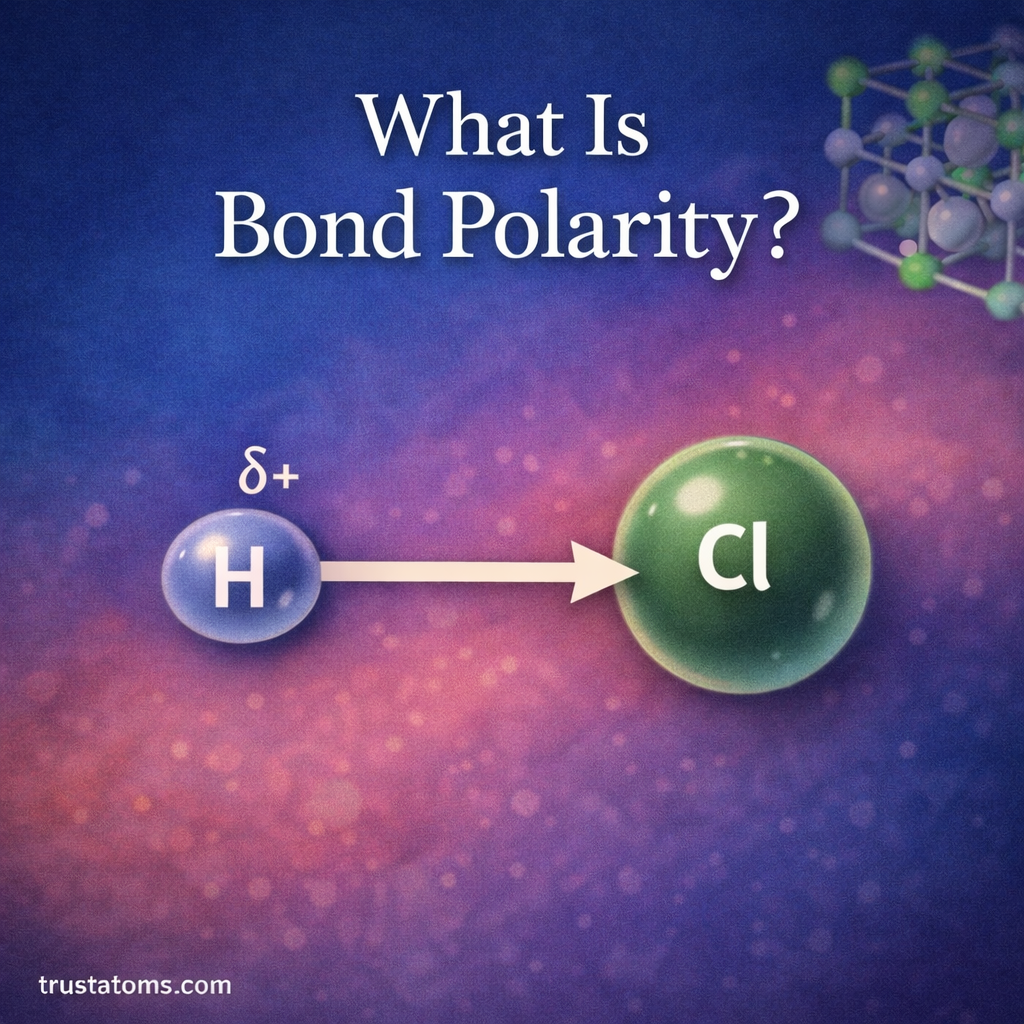
Bond polarity is a fundamental concept in chemistry that explains how electrons are shared between atoms in a chemical bond. Some atoms pull shared electrons more strongly than others, creating an uneven distribution of electron density.
This unequal sharing produces partial positive and partial negative regions within molecules, which affects how substances interact, dissolve, and react chemically.
Understanding bond polarity helps explain many chemical behaviors, including why water dissolves salt, how molecules attract each other, and why some substances mix while others do not.
What Is Bond Polarity?
Bond polarity describes the unequal sharing of electrons between two atoms in a chemical bond.
When atoms bond, they share electrons to achieve more stable electron configurations. However, if one atom attracts electrons more strongly than the other, the shared electrons spend more time near that atom.
This creates:
- A partial negative charge (δ−) on the more electronegative atom
- A partial positive charge (δ+) on the less electronegative atom
This separation of charge forms a polar bond.
If electrons are shared evenly, the bond is considered nonpolar.
Why Bond Polarity Occurs
Bond polarity occurs because atoms have different abilities to attract electrons.
This ability is called electronegativity.
Electronegativity measures how strongly an atom pulls electrons toward itself in a chemical bond.
When two bonded atoms have:
- Similar electronegativity values, electrons are shared equally
- Different electronegativity values, electrons shift toward the more electronegative atom
The greater the electronegativity difference, the more polar the bond becomes.
Types of Chemical Bonds Based on Polarity
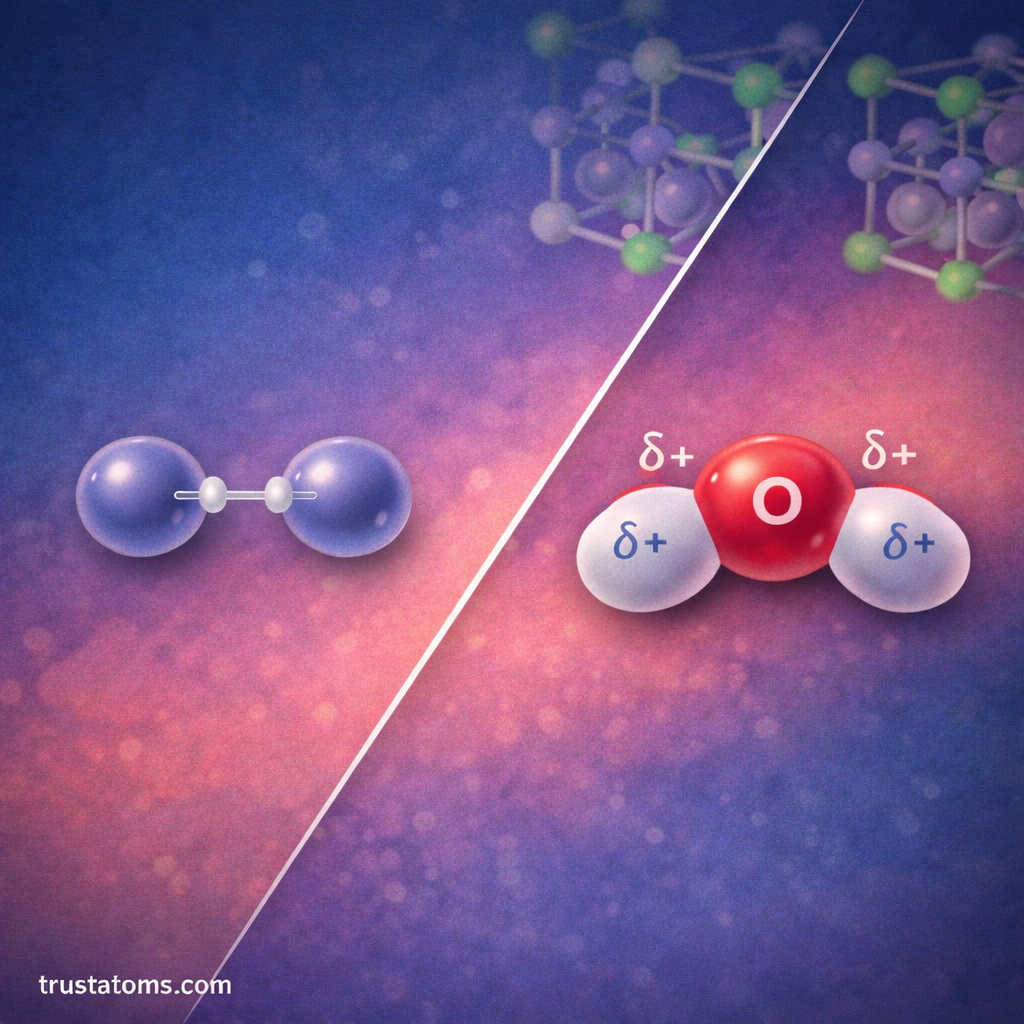
Chemists often categorize bonds based on how electrons are shared between atoms.
Nonpolar Covalent Bonds
In a nonpolar covalent bond, electrons are shared equally.
This usually happens when atoms have very similar electronegativity values.
Characteristics include:
- No significant charge separation
- Even distribution of electron density
- Nonpolar molecules
Examples:
- Hydrogen gas (H₂)
- Oxygen gas (O₂)
- Nitrogen gas (N₂)
Because the atoms are identical, neither atom pulls electrons more strongly.
Polar Covalent Bonds
In a polar covalent bond, electrons are shared unequally.
One atom pulls electrons more strongly, creating partial charges.
Characteristics include:
- Uneven electron distribution
- Partial positive and negative charges
- Molecular dipoles
Examples:
- Water (H₂O)
- Hydrogen chloride (HCl)
- Ammonia (NH₃)
These molecules contain bonds where one atom attracts electrons more strongly than the other.
Ionic Bonds
Ionic bonds represent the extreme case of bond polarity.
Instead of sharing electrons, one atom transfers electrons to another.
This produces:
- Positively charged ions (cations)
- Negatively charged ions (anions)
Example:
Sodium chloride (NaCl)
Sodium loses an electron while chlorine gains one, forming oppositely charged ions that attract each other.
Partial Charges and Dipoles
Bond polarity creates partial charges, which lead to something called a dipole.
A dipole forms when a molecule has two regions with different electrical charges.
In a polar bond:
- The more electronegative atom becomes partially negative (δ−)
- The other atom becomes partially positive (δ+)
Chemists often represent dipoles using an arrow that points toward the more electronegative atom.
These dipoles influence how molecules interact with each other.
Bond Polarity vs Molecular Polarity
Bond polarity and molecular polarity are related but not identical concepts.
Bond Polarity
Refers to the electron distribution between two atoms in a bond.
It depends primarily on:
- Electronegativity differences
Molecular Polarity
Refers to the overall charge distribution of an entire molecule.
It depends on:
- Bond polarity
- Molecular shape
Sometimes polar bonds cancel each other out.
Example: Carbon Dioxide (CO₂)
The carbon–oxygen bonds are polar.
However, CO₂ has a linear structure, so the bond dipoles cancel.
Result:
- The molecule is nonpolar overall.
Example: Water (H₂O)
Water has polar O–H bonds.
Because the molecule is bent in shape, the dipoles do not cancel.
Result:
- The molecule is polar overall.
This polarity explains many important properties of water.
How to Predict Bond Polarity
Chemists predict bond polarity by comparing electronegativity values.
Step-by-Step Method
- Identify the two atoms in the bond
- Look up their electronegativity values
- Subtract the smaller value from the larger one
- Interpret the difference
General guidelines:
- 0.0–0.4 → Nonpolar covalent
- 0.5–1.7 → Polar covalent
- 1.7 or greater → Ionic
These ranges are approximate but widely used.
Why Bond Polarity Matters
Bond polarity plays a major role in many chemical and physical properties.
1. Solubility
Polar molecules dissolve better in polar solvents.
Example:
- Salt dissolves in water
- Oil does not mix with water
This principle is often summarized as “like dissolves like.”
2. Intermolecular Forces
Polar molecules experience stronger attractions between molecules.
These forces include:
- Dipole–dipole interactions
- Hydrogen bonding
These interactions affect boiling points, melting points, and viscosity.
3. Chemical Reactivity
Regions with partial charges can influence chemical reactions.
Electrons may move toward positively charged areas or away from negatively charged regions.
This affects:
- Reaction mechanisms
- Reaction rates
- Product formation
4. Biological Chemistry
Bond polarity is essential in biological molecules.
It influences the behavior of:
- Proteins
- DNA
- Enzymes
- Cell membranes
Many biological interactions rely on polar bonds and intermolecular forces.
Common Examples of Polar Bonds
Here are some well-known examples of polar covalent bonds:
O–H bond
- Found in water and alcohols
- Oxygen pulls electrons more strongly than hydrogen
H–Cl bond
- Found in hydrogen chloride
- Chlorine attracts electrons more strongly
N–H bond
- Found in ammonia and many biological molecules
- Nitrogen attracts electrons more strongly than hydrogen
These polar bonds contribute to molecular polarity and chemical behavior.
Key Takeaways
Bond polarity describes how unevenly electrons are shared in a chemical bond.
Important points to remember:
- Bond polarity occurs when atoms have different electronegativities
- Unequal electron sharing creates partial charges
- Polar bonds produce molecular dipoles
- Molecular shape determines whether dipoles cancel
- Bond polarity influences solubility, intermolecular forces, and chemical reactions
By understanding bond polarity, chemists can predict how molecules interact and behave in both chemical reactions and everyday materials.