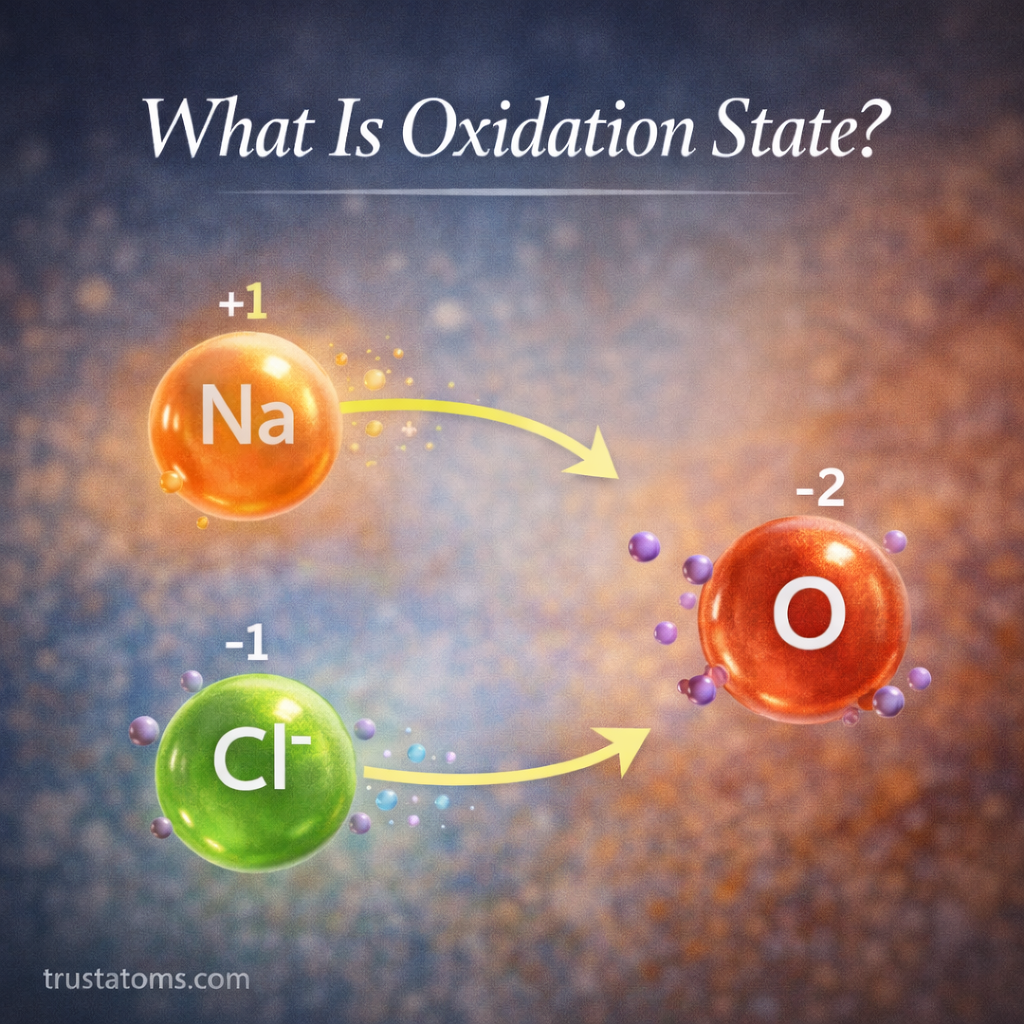
Oxidation state, also called oxidation number, is a concept in chemistry used to track how electrons are transferred between atoms during chemical reactions. It helps chemists understand how elements interact, particularly in reactions involving electron transfer.
In simple terms, the oxidation state of an atom represents the number of electrons it has gained, lost, or shared when forming chemical bonds. This idea is especially important in studying oxidation–reduction reactions, also known as redox reactions.
By assigning oxidation states to atoms in compounds, scientists can analyze how substances react and predict the products of chemical reactions.
Definition of Oxidation State
The oxidation state of an atom is the hypothetical charge it would have if all bonds to other atoms were completely ionic.
This means chemists imagine that:
- Electrons in bonds belong entirely to the more electronegative atom.
- The difference in electron ownership determines the oxidation state.
Oxidation states help represent how electrons shift during reactions.
For example:
- If an atom loses electrons, its oxidation state becomes more positive.
- If an atom gains electrons, its oxidation state becomes more negative.
Why Oxidation States Are Important
Oxidation states provide a useful framework for understanding chemical behavior.
They help chemists:
- Identify oxidation and reduction processes
- Track electron transfer in reactions
- Balance redox equations
- Predict chemical reactivity
- Determine the structure of compounds
Without oxidation states, it would be much more difficult to analyze how complex chemical reactions occur.
Oxidation and Reduction in Chemistry
Oxidation states are closely connected to oxidation and reduction reactions.
These reactions involve the movement of electrons between atoms.
Oxidation
Oxidation occurs when an atom:
- Loses electrons
- Experiences an increase in oxidation state
Example:
- Iron atoms losing electrons during rust formation.
Reduction
Reduction occurs when an atom:
- Gains electrons
- Experiences a decrease in oxidation state
Example:
- Oxygen gaining electrons when forming water.
Because oxidation and reduction always occur together, they are known as redox reactions.
Common Rules for Assigning Oxidation States
Chemists use several standard rules to determine oxidation states in compounds.
Rule 1: Elements in Their Natural Form
Atoms in their pure elemental form have an oxidation state of zero.
Examples:
- O₂
- H₂
- Fe
- Cl₂
Rule 2: Alkali Metals
Alkali metals in compounds usually have an oxidation state of +1.
Examples include:
- Sodium (Na)
- Potassium (K)
- Lithium (Li)
Rule 3: Alkaline Earth Metals
These elements typically have an oxidation state of +2.
Examples:
- Magnesium (Mg)
- Calcium (Ca)
Rule 4: Oxygen
Oxygen usually has an oxidation state of −2 in compounds.
Exceptions exist in certain molecules like peroxides.
Rule 5: Hydrogen
Hydrogen generally has an oxidation state of:
- +1 when bonded to nonmetals
- −1 when bonded to metals
Rule 6: Sum of Oxidation States
The total oxidation states in a compound must equal the overall charge of the molecule or ion.
For example:
- In neutral molecules, the total equals zero
- In ions, the total equals the ionic charge
Example of Oxidation State in Water
Water provides a simple example of oxidation states.
Water contains:
- Two hydrogen atoms
- One oxygen atom
Applying the rules:
- Hydrogen typically has an oxidation state of +1
- Oxygen typically has an oxidation state of −2
When combined:
- +1 + +1 + −2 = 0
This matches the neutral charge of the water molecule.
Example of Oxidation States in Sodium Chloride
Sodium chloride (table salt) also demonstrates oxidation states clearly.
In NaCl:
- Sodium loses one electron
- Chlorine gains one electron
The oxidation states become:
- Sodium: +1
- Chlorine: −1
This electron transfer forms an ionic bond between the atoms.
Oxidation States in Redox Reactions
In redox reactions, oxidation states change as electrons move between substances.
Chemists track these changes to determine which substance is oxidized and which is reduced.
Example
Consider a reaction between zinc and copper ions.
During the reaction:
- Zinc loses electrons and its oxidation state increases.
- Copper ions gain electrons and their oxidation state decreases.
This electron exchange drives the chemical reaction.
Tracking oxidation states helps chemists understand the direction and mechanism of such reactions.
Oxidation States in Everyday Chemistry

Oxidation states appear in many chemical processes that occur in daily life and industry.
Examples include:
- Rusting of iron
- Combustion reactions
- Battery chemistry
- Photosynthesis
- Industrial chemical production
In each case, oxidation states help scientists track electron movement and chemical change.
Common Misconceptions About Oxidation States
Although oxidation states resemble charges, they are not always actual charges on atoms.
Important points to remember:
- Oxidation states are a bookkeeping tool for electrons.
- They help describe how electrons are distributed in molecules.
- Real electron sharing in covalent bonds may differ from the simplified oxidation-state model.
Despite this simplification, oxidation states remain one of the most powerful tools in chemistry.
Final Thoughts
Oxidation state is a fundamental concept that allows chemists to track how electrons move during chemical reactions. By assigning oxidation numbers to atoms, scientists can analyze redox reactions, predict chemical behavior, and balance complex equations.
From rusting metals to battery chemistry, oxidation states help explain many processes that shape both everyday life and industrial technology.
Understanding oxidation states provides an essential foundation for studying chemical reactions and the behavior of elements in compounds.