The human body must maintain a stable internal environment to function properly. One of the most critical aspects of this balance is acid–base regulation, which keeps blood pH within a narrow range. A key player in this system is carbon dioxide (CO₂), a waste product of metabolism that directly influences acidity.
Through a combination of respiratory and chemical processes, the body carefully regulates CO₂ levels to maintain optimal pH and support vital functions.
What Is Acid–Base Balance?
Acid–base balance refers to the regulation of hydrogen ion concentration (H⁺) in body fluids, which determines pH levels.
Normal Blood pH Range
- 7.35 to 7.45 (slightly alkaline)
Even small deviations outside this range can disrupt cellular processes and become life-threatening.
The Role of CO₂ in pH Regulation
Carbon dioxide is closely linked to acidity in the body.
How CO₂ Affects pH
- CO₂ reacts with water to form carbonic acid
- Carbonic acid releases hydrogen ions (H⁺)
- Increased H⁺ lowers pH (more acidic)
Key Concept
- High CO₂ = Lower pH (acidic)
- Low CO₂ = Higher pH (alkaline)
This relationship makes CO₂ a central factor in acid–base balance.
The Carbonic Acid–Bicarbonate Buffer System
The body uses buffering systems to stabilize pH, with the most important being the carbonic acid–bicarbonate buffer system.
How the Buffer System Works
- CO₂ combines with water to form carbonic acid
- Carbonic acid breaks down into:
- Hydrogen ions (H⁺)
- Bicarbonate (HCO₃⁻)
- The system can shift in either direction to maintain balance
This reversible process allows the body to quickly respond to changes in acidity.
The Role of the Respiratory System
The lungs play a major role in regulating CO₂ levels.
How Breathing Controls pH
- Faster breathing (hyperventilation)
- Removes more CO₂
- Raises pH (less acidic)
- Slower breathing (hypoventilation)
- Retains CO₂
- Lowers pH (more acidic)
Why This Matters
Breathing adjustments provide a rapid way to correct pH imbalances.
The Role of the Kidneys
While the lungs act quickly, the kidneys provide long-term regulation.
Kidney Functions in Acid–Base Balance
- Excrete excess hydrogen ions (H⁺)
- Reabsorb or produce bicarbonate (HCO₃⁻)
- Adjust blood pH over hours to days
The kidneys complement the respiratory system for sustained balance.
Types of Acid–Base Imbalances
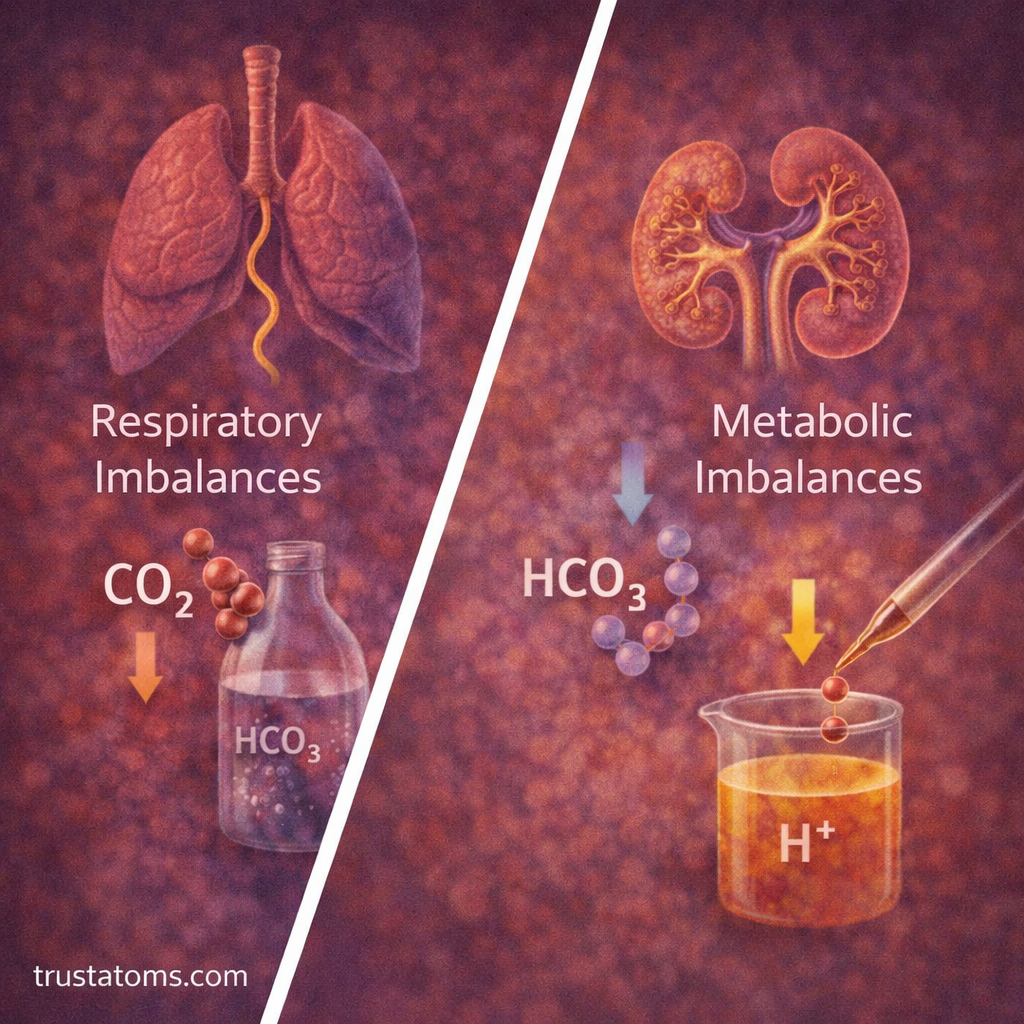
Disruptions in CO₂ regulation can lead to different types of imbalances.
Respiratory Imbalances
- Respiratory acidosis
- Caused by high CO₂
- Results from slow or impaired breathing
- Respiratory alkalosis
- Caused by low CO₂
- Often due to rapid breathing
Metabolic Imbalances
- Metabolic acidosis
- Caused by excess acid or loss of bicarbonate
- Metabolic alkalosis
- Caused by excess bicarbonate or loss of acid
How the Body Maintains Balance
The body uses multiple systems to keep pH stable.
Coordinated Response
- Chemoreceptors detect changes in CO₂ and pH
- The brainstem adjusts breathing rate
- The lungs regulate CO₂ levels
- The kidneys fine-tune long-term balance
This coordination ensures rapid and sustained control.
Real-World Examples
Acid–base balance and CO₂ regulation are constantly at work in everyday life.
Common Situations
- Exercise
- Increased CO₂ → faster breathing
- Anxiety or panic
- Rapid breathing → low CO₂ → dizziness
- High altitude
- Lower oxygen → increased breathing
Why Acid–Base Balance Is Essential
Maintaining proper pH is critical for nearly every function in the body.
Key Benefits
- Supports enzyme activity
- Maintains proper nerve and muscle function
- Ensures efficient oxygen delivery
- Prevents cellular damage
Final Thoughts
Acid–base balance and CO₂ regulation are essential for maintaining stability within the body. Through the combined efforts of the lungs, kidneys, and buffering systems, the body continuously adjusts to keep pH within a safe range.
Understanding this balance highlights the importance of respiration not just for oxygen intake, but also for controlling acidity and supporting overall health.