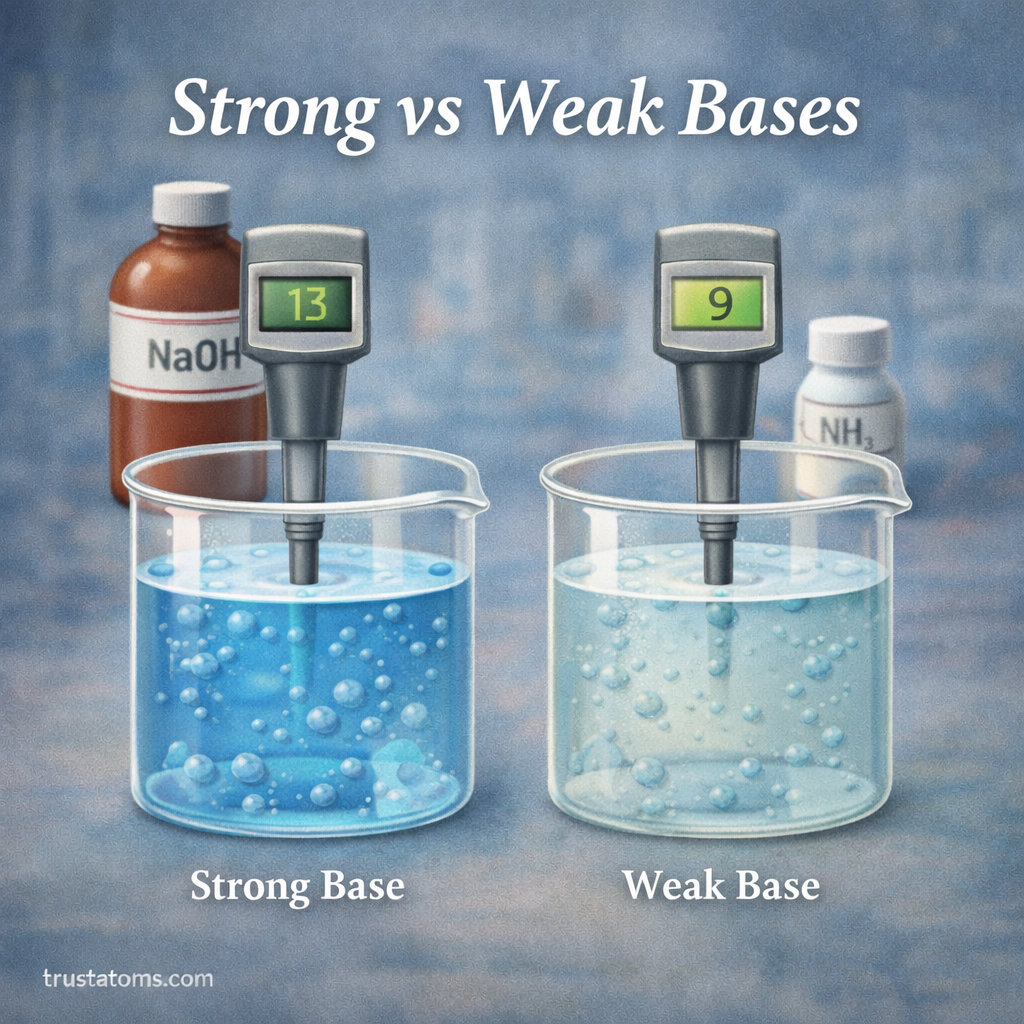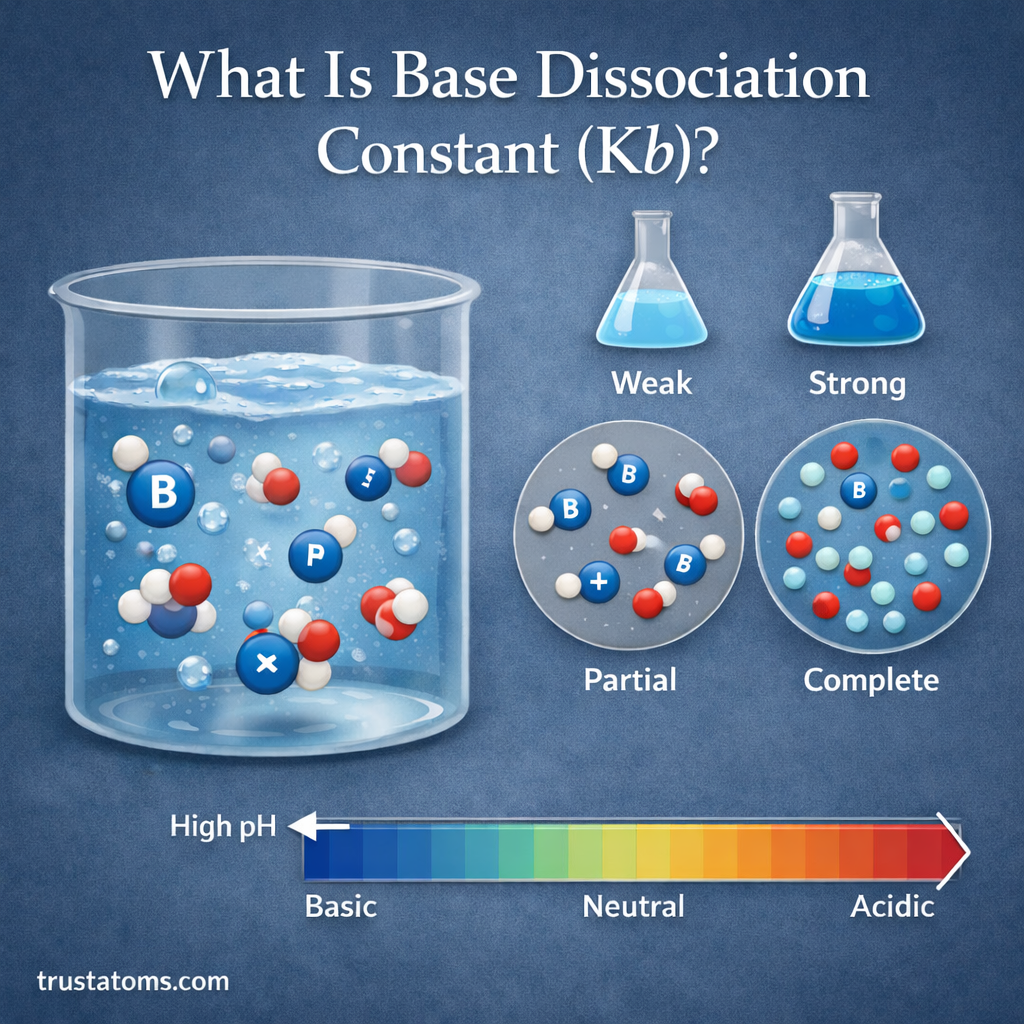
In chemistry, understanding how substances behave in water is essential for studying reactions, biological systems, and industrial processes. One important concept used to measure the strength of a base is the base dissociation constant, commonly written as Kb.
The base dissociation constant helps chemists determine how strongly a base reacts with water to produce hydroxide ions. The larger the Kb value, the stronger the base.
This guide explains what the base dissociation constant is, how it works, how it is calculated, and why it matters in chemistry.
Continue reading “What Is Base Dissociation Constant (Kb)?”