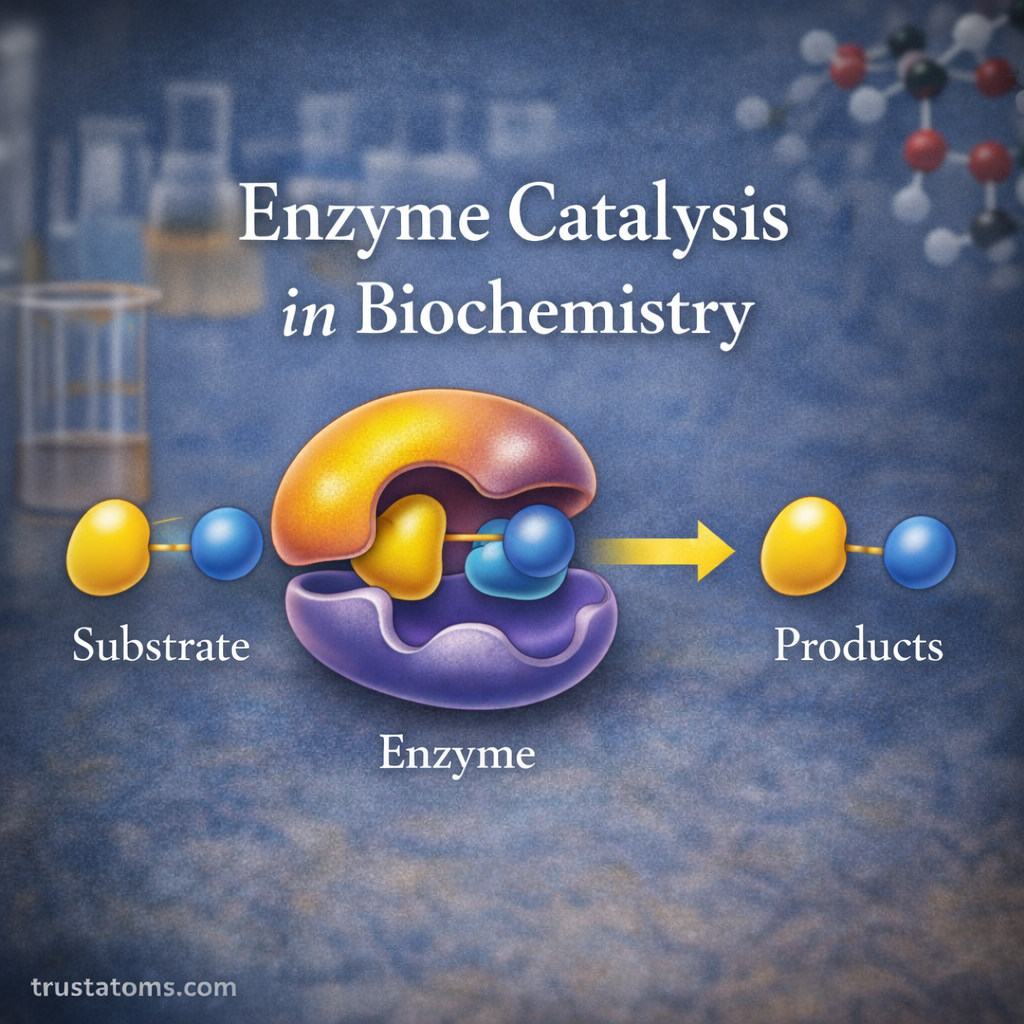
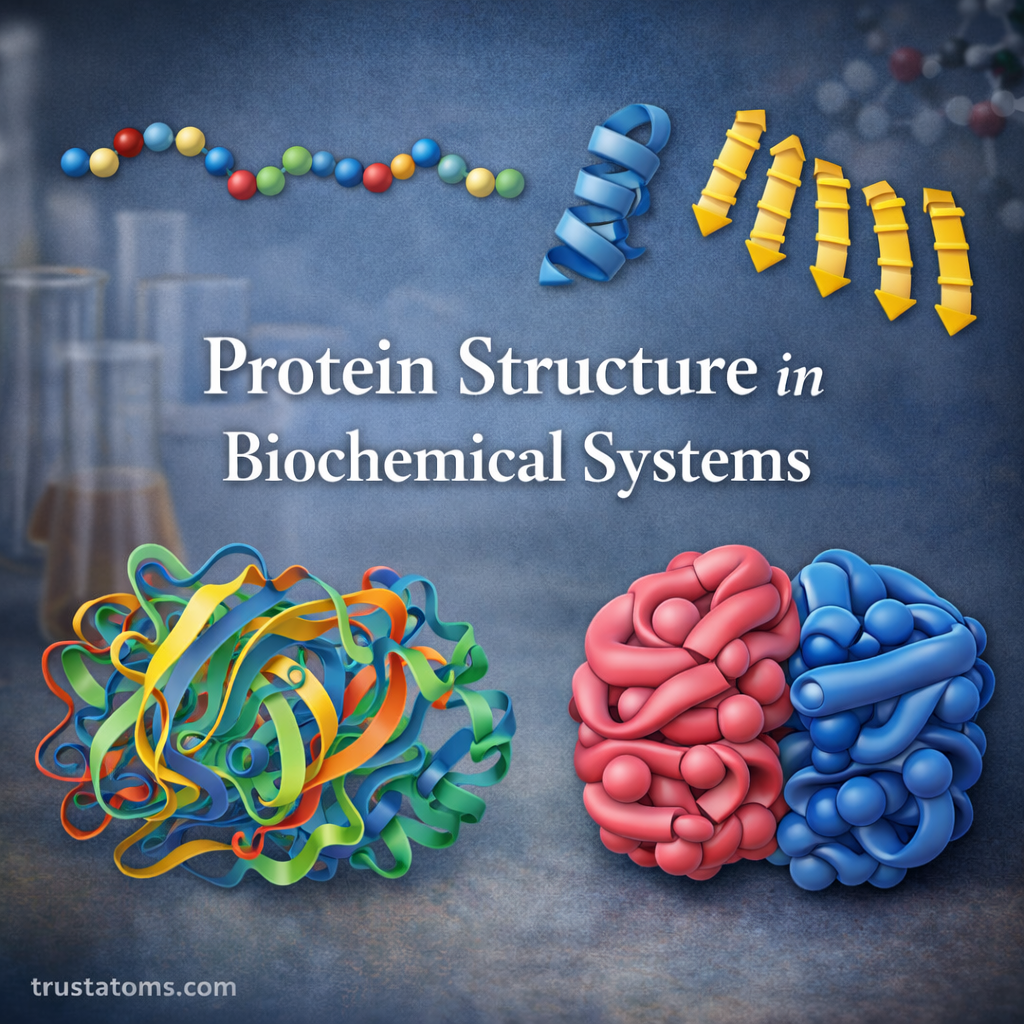
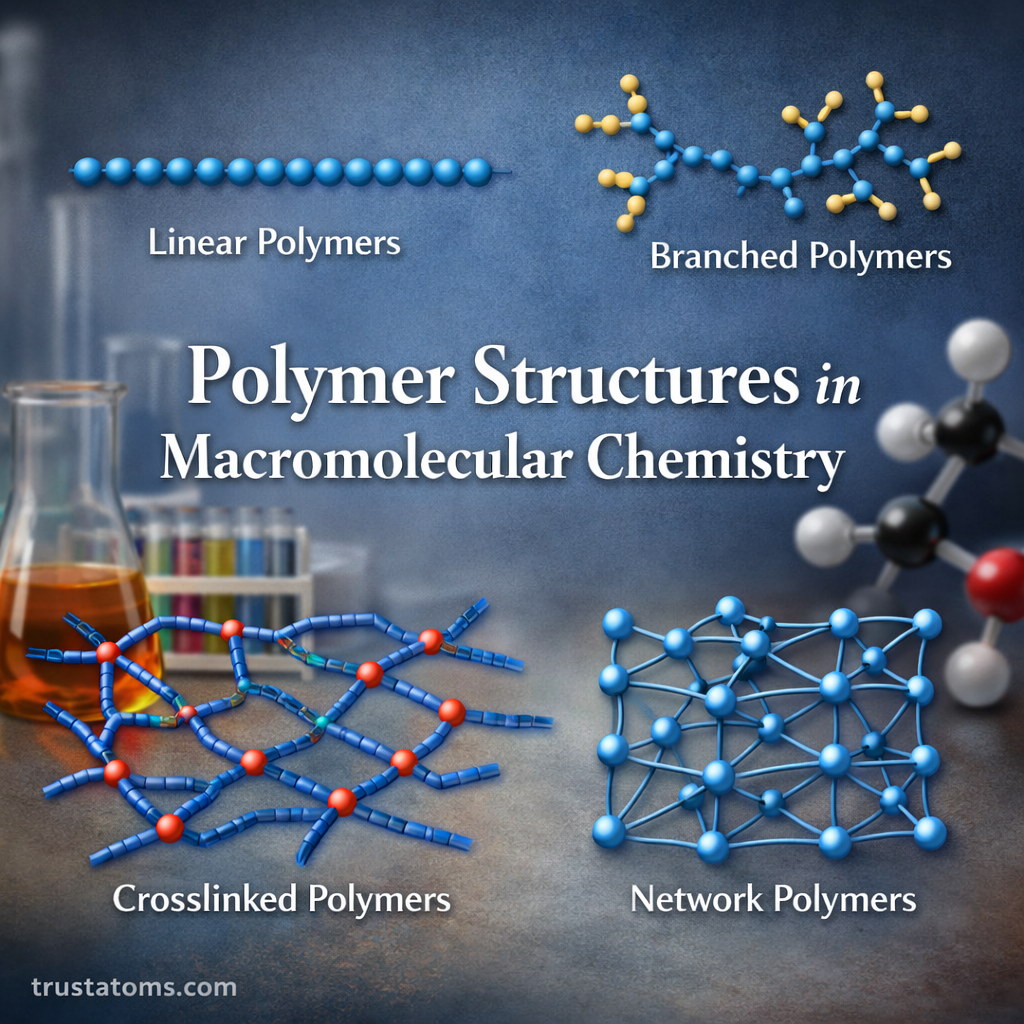
Chromatography is one of the most important techniques used in analytical chemistry to separate, identify, and analyze mixtures of chemical substances. It allows scientists to isolate individual components within complex samples, making it easier to study their properties and concentrations.
From pharmaceutical testing and environmental monitoring to food safety and forensic science, chromatography plays a central role in modern laboratory analysis.
This guide explains how chromatography works, the major types used in analytical chemistry, and why it remains one of the most reliable tools for chemical analysis.
Continue reading “Chromatography in Analytical Chemistry”