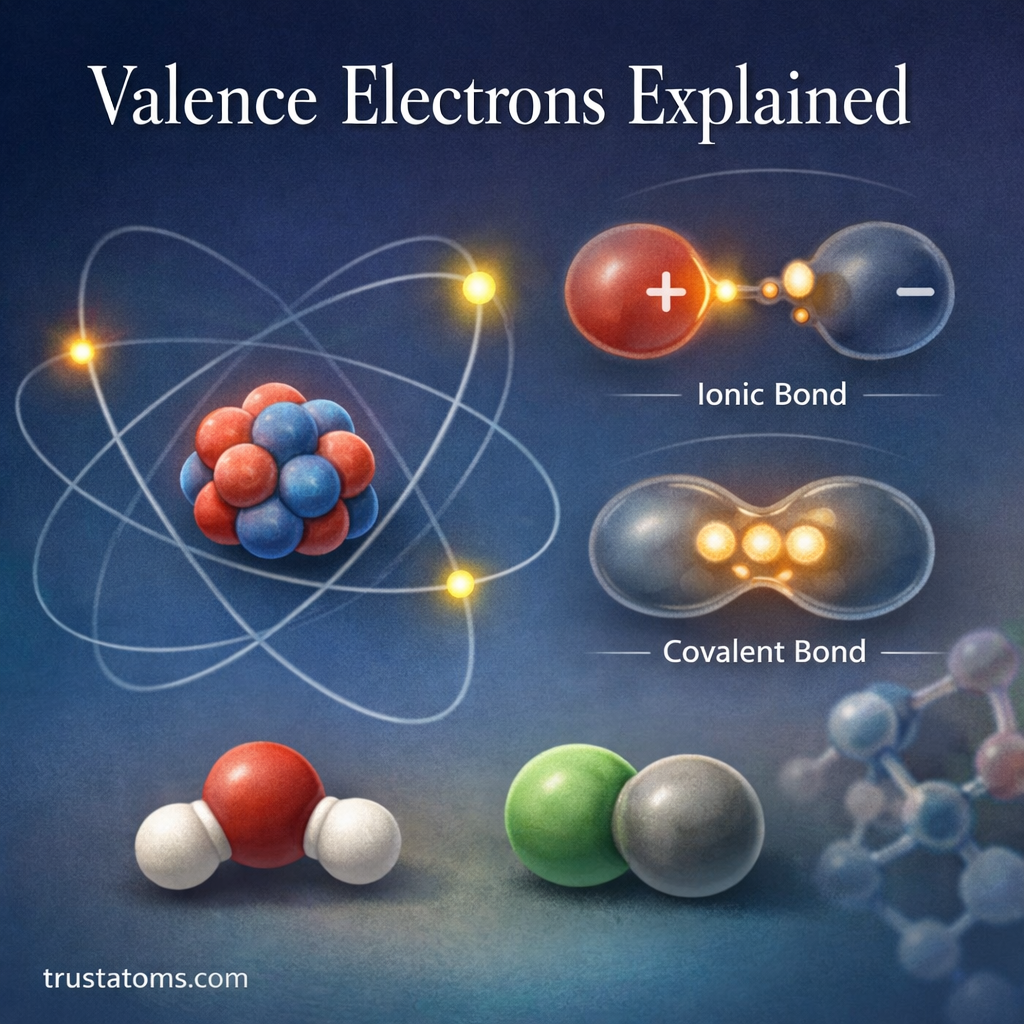
A molecule is formed when two or more atoms bond together. These bonded atoms act as a single unit and make up many of the substances around us — including water, oxygen, sugar, and carbon dioxide.
Understanding what a molecule is helps explain how matter is built, how chemical reactions work, and why substances have different properties.
Molecule Definition in Simple Terms
A molecule is a group of atoms chemically bonded together.
These atoms can be:
- The same element (like oxygen gas, O₂)
- Different elements (like water, H₂O)
The key idea is that the atoms are held together by chemical bonds and function as one stable unit.
Atoms vs. Molecules
To understand molecules, it helps to compare them with atoms.
Atom
- The smallest unit of an element
- Cannot be broken down by chemical means
- Example: a single oxygen atom
Molecule
- Two or more atoms bonded together
- Can consist of the same or different elements
- Example: two oxygen atoms bonded together (O₂)
In short:
Atoms are the building blocks.
Molecules are structures built from those blocks.
How Atoms Form Molecules
Atoms bond together because they seek stability. Most atoms are more stable when their outer energy level (valence shell) is filled with electrons.
They achieve this by:
- Sharing electrons (covalent bonds)
- Transferring electrons (ionic bonds)
- Attracting through weaker intermolecular forces
When atoms bond, they form molecules with new properties different from the individual atoms.
For example:
- Hydrogen gas is flammable.
- Oxygen supports combustion.
- Water (made from hydrogen and oxygen) is used to put out fires.
The molecule has completely different characteristics than its separate atoms.
Types of Molecules
Molecules can be grouped into different categories based on their composition.
1. Diatomic Molecules
These consist of two atoms.
Examples:
- Oxygen (O₂)
- Nitrogen (N₂)
- Hydrogen (H₂)
Some elements naturally exist as diatomic molecules.
2. Compound Molecules
These contain atoms from different elements.
Examples:
- Water (H₂O)
- Carbon dioxide (CO₂)
- Ammonia (NH₃)
These molecules form chemical compounds.
3. Large and Complex Molecules
Some molecules are small and simple. Others are extremely large and complex.
Examples:
- Glucose (a sugar molecule)
- Proteins (made of thousands of atoms)
- DNA (contains millions of atoms)
Large biological molecules are essential for life.
Molecular Bonds Explained
The way atoms bond determines the molecule’s structure and properties.
Covalent Bonds
- Atoms share electrons
- Strong and common in organic molecules
- Found in water, carbon dioxide, and sugars
Ionic Bonds
- One atom transfers electrons to another
- Creates charged particles (ions)
- Common in salts like sodium chloride
Hydrogen Bonds (Weaker Interactions)
- Occur between molecules
- Important in water and biological systems
The type of bond affects melting point, boiling point, reactivity, and structure.
Molecular Formulas
A molecular formula tells you:
- Which elements are present
- How many atoms of each element are in the molecule
For example:
- H₂O = 2 hydrogen atoms + 1 oxygen atom
- CO₂ = 1 carbon atom + 2 oxygen atoms
The numbers show the ratio of atoms in the molecule.
Why Molecules Matter
Molecules are responsible for the physical and chemical properties of substances.
They determine:
- Whether something is solid, liquid, or gas
- How substances react with one another
- Biological processes in living organisms
- Energy storage and transfer
Every chemical reaction involves molecules breaking apart and forming new molecules.
Molecules in Everyday Life

You interact with molecules constantly.
Examples include:
- The oxygen you breathe
- The water you drink
- The carbon dioxide you exhale
- The proteins in your body
- The fuels that power vehicles
Even the materials in smartphones, clothing, and food are made of molecules.
Key Takeaways
- A molecule is two or more atoms bonded together.
- Molecules can consist of the same or different elements.
- Atoms bond to become more stable.
- The type of bond affects molecular properties.
- All matter is made of molecules (except single-atom noble gases).
Understanding molecules is essential to chemistry because they explain how matter forms, changes, and interacts.