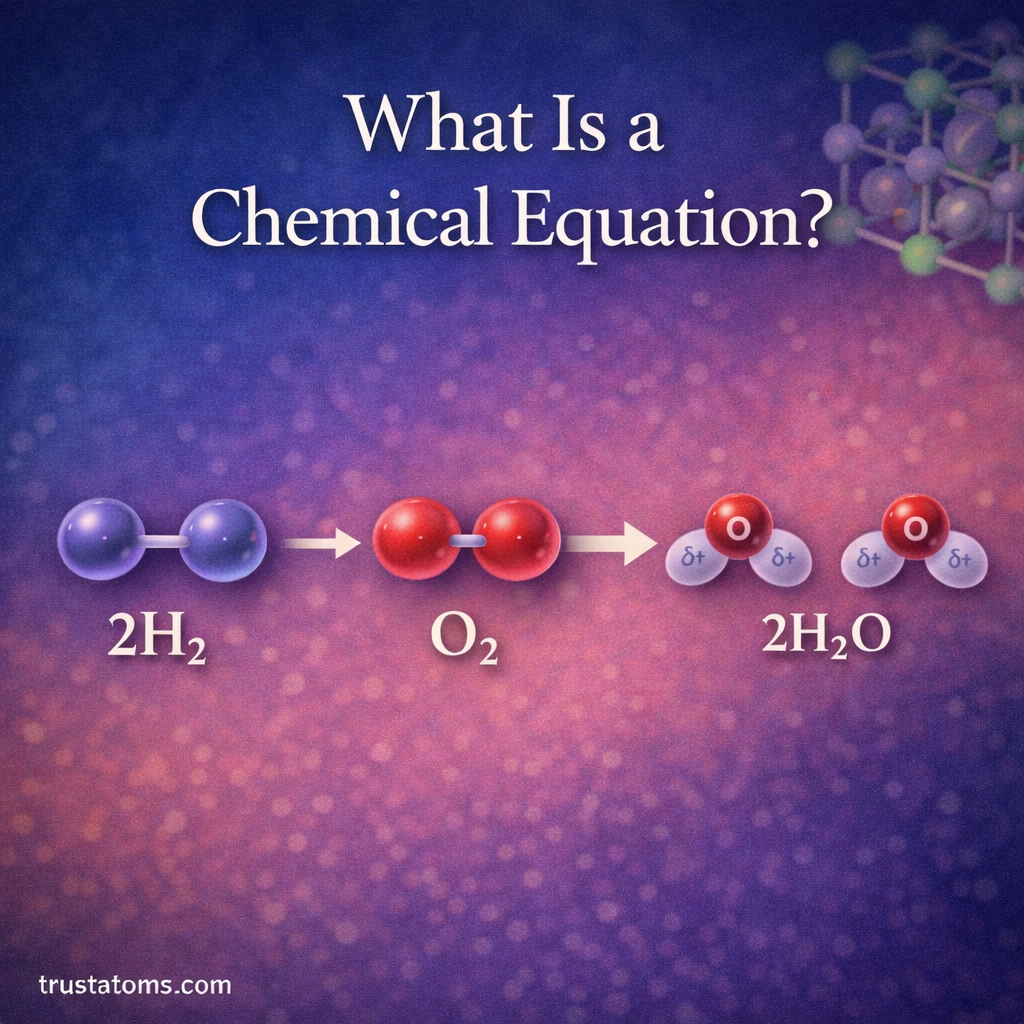
Electronegativity is one of the most important concepts in chemistry because it helps explain why atoms bond the way they do. It influences the formation of molecules, the behavior of chemical reactions, and the physical properties of substances.
In simple terms, electronegativity describes how strongly an atom attracts electrons when it forms a chemical bond. Some atoms pull electrons toward themselves very strongly, while others exert a weaker pull.
Understanding electronegativity allows chemists to predict how atoms interact, what types of bonds will form, and how molecules behave.
The Basic Definition of Electronegativity
Electronegativity is the ability of an atom to attract shared electrons toward itself in a chemical bond.
When two atoms bond together, they share electrons. However, the electrons are rarely shared equally. The atom with the higher electronegativity pulls the electrons closer to itself.
Key points about electronegativity:
- It applies to atoms involved in chemical bonds
- It describes relative attraction for electrons
- It helps determine bond polarity
- It influences chemical reactivity
Electronegativity is not a directly measurable physical quantity like mass or charge. Instead, scientists determine it using scales based on experimental observations and calculations.
The Electronegativity Scale
The most widely used system is the Pauling scale, developed by chemist Linus Pauling.
On this scale:
- Higher numbers indicate stronger attraction for electrons
- Lower numbers indicate weaker attraction
Typical values range roughly from 0.7 to 4.0.
Examples of common elements:
- Fluorine: 3.98 (highest electronegativity)
- Oxygen: 3.44
- Nitrogen: 3.04
- Carbon: 2.55
- Hydrogen: 2.20
- Sodium: 0.93
- Cesium: 0.79 (very low)
Fluorine sits at the top of the scale because it strongly attracts electrons in chemical bonds.
Why Electronegativity Matters in Chemical Bonds

Electronegativity determines how electrons are distributed between atoms when they bond.
The difference in electronegativity between two atoms helps predict the type of bond formed.
Three Common Bond Types
- Nonpolar Covalent Bonds
- Electrons are shared nearly equally
- Occurs when electronegativity values are very similar
- Polar Covalent Bonds
- Electrons are shared unequally
- One atom pulls electrons closer to itself
- Ionic Bonds
- One atom effectively takes electrons from another
- Occurs when the electronegativity difference is large
For example, in a water molecule, oxygen has a much higher electronegativity than hydrogen. As a result, oxygen pulls the shared electrons closer, creating a polar molecule.
How Electronegativity Influences Molecular Behavior
Electronegativity affects several important chemical and physical properties.
Molecular Polarity
When electrons are unevenly distributed, molecules develop partial positive and negative regions. This is known as polarity.
Polar molecules often:
- Dissolve well in water
- Have higher boiling points
- Form hydrogen bonds
Chemical Reactivity
Atoms with high electronegativity often:
- Gain electrons in reactions
- Form negative ions
- Act as oxidizing agents
Atoms with low electronegativity often:
- Lose electrons easily
- Form positive ions
- React strongly with highly electronegative elements
Intermolecular Forces
Electronegativity differences can create dipole interactions, which influence how molecules attract each other. These forces affect properties like:
- Melting point
- Boiling point
- Solubility
Trends of Electronegativity in the Periodic Table
Electronegativity follows clear patterns across the periodic table.
Across a Period (Left to Right)
Electronegativity increases.
Reasons include:
- Increasing nuclear charge
- Stronger pull on electrons
- Smaller atomic radius
Down a Group (Top to Bottom)
Electronegativity decreases.
Reasons include:
- Larger atomic size
- More electron shells
- Greater shielding from the nucleus
This explains why elements in the upper right corner of the periodic table have the highest electronegativity values.
Factors That Affect Electronegativity
Several atomic properties influence how strongly an atom attracts electrons.
Atomic Size
Smaller atoms pull electrons more strongly because the nucleus is closer to the bonding electrons.
Nuclear Charge
Atoms with more protons exert a stronger attraction on electrons.
Electron Shielding
Inner electron shells partially block the nucleus’s pull on outer electrons, reducing electronegativity.
Examples of Electronegativity in Everyday Chemistry
Electronegativity helps explain many common substances and materials.
Water (H₂O)
Oxygen attracts electrons more strongly than hydrogen, making water a polar molecule. This polarity allows water to dissolve many substances and gives it unique properties like surface tension.
Sodium Chloride (Table Salt)
Sodium has very low electronegativity while chlorine has high electronegativity. Chlorine takes an electron from sodium, creating an ionic bond.
Carbon Compounds
Carbon has a moderate electronegativity, allowing it to bond with many elements. This flexibility is one reason carbon forms the backbone of organic chemistry and life.
Common Misunderstandings About Electronegativity
Several misconceptions can make electronegativity confusing at first.
Electronegativity vs. Electron Affinity
These are different concepts:
- Electronegativity: attraction for electrons in a bond
- Electron affinity: energy change when an atom gains an electron
Electronegativity vs. Ionization Energy
Ionization energy measures how much energy is required to remove an electron, while electronegativity measures how strongly atoms pull shared electrons in bonds.
Electronegativity Only Applies in Bonds
An isolated atom does not display electronegativity. The concept only applies when atoms interact in molecules.
Why Electronegativity Is Fundamental in Chemistry
Electronegativity is a key concept because it helps scientists:
- Predict bond types
- Understand molecular structure
- Explain chemical reactivity
- Determine polarity and intermolecular forces
From simple salts to complex biological molecules, electronegativity plays a central role in how matter behaves at the atomic level.
Understanding this concept provides a foundation for studying chemical bonding, molecular geometry, and chemical reactions throughout chemistry.