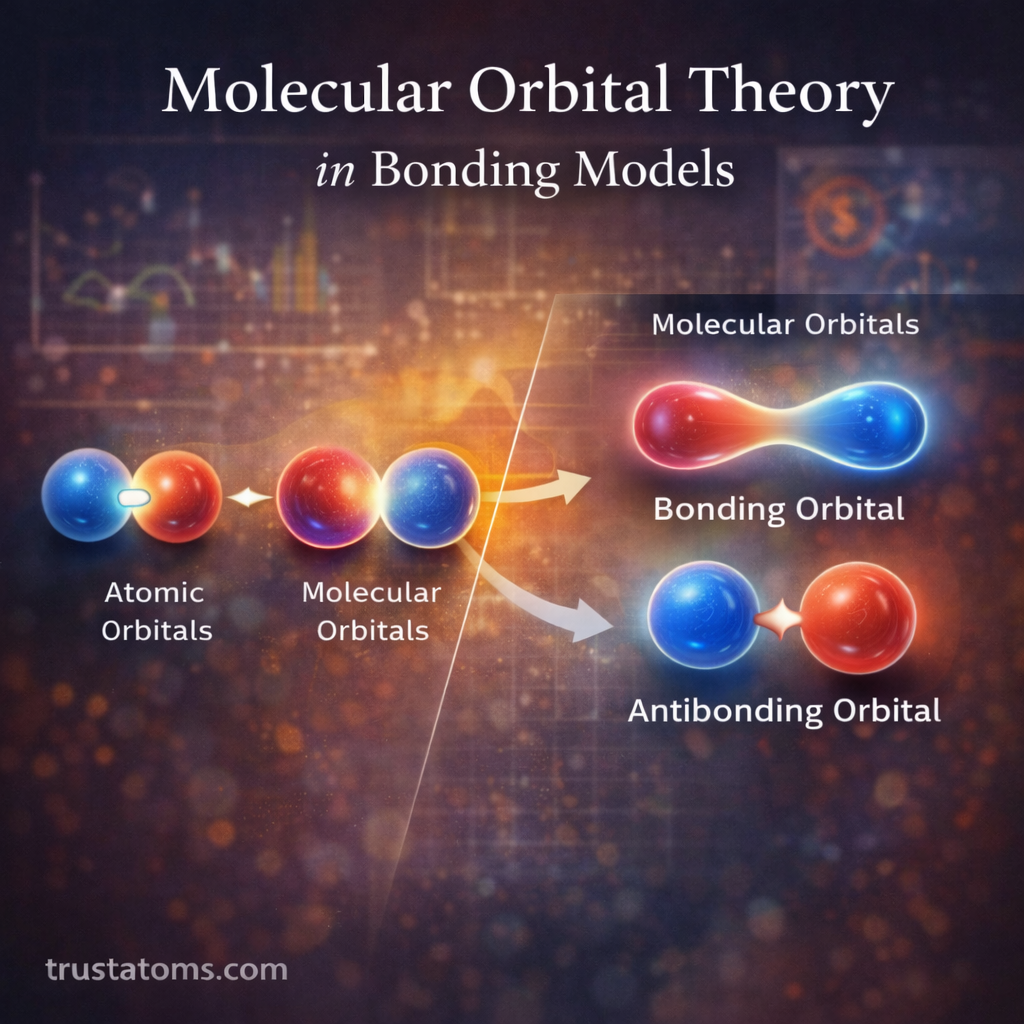
Chemical bond energy is the amount of energy required to break a chemical bond between atoms in a molecule. It represents how strong a bond is and how much energy is needed to separate the bonded atoms.
In chemistry, bond energy is an important concept because it helps scientists understand how molecules form, break apart, and participate in chemical reactions. Reactions often involve breaking existing bonds and forming new ones, and the energy involved in these processes determines whether a reaction releases energy or absorbs it.
Understanding chemical bond energy provides insight into the stability of molecules and the behavior of chemical reactions.
The Basic Concept of Bond Energy
Chemical bonds hold atoms together in molecules. The energy that keeps these atoms connected is called bond energy.
Bond energy is typically measured as the amount of energy required to break a bond in one mole of a substance.
In simple terms:
- Stronger bonds require more energy to break
- Weaker bonds require less energy to break
This means that molecules with high bond energies are generally more stable.
How Bond Energy Works in Chemical Reactions

Chemical reactions involve both breaking old bonds and forming new ones.
The overall energy change in a reaction depends on the balance between these two processes.
The general sequence is:
- Energy is absorbed to break existing bonds in reactants.
- Atoms rearrange and form new bonds.
- Energy is released when the new bonds form.
If the reaction releases more energy than it absorbs, it is considered an exothermic reaction.
If the reaction absorbs more energy than it releases, it is an endothermic reaction.
Factors That Affect Bond Energy
Several factors influence how strong a chemical bond is and how much energy is required to break it.
Type of Bond
Different types of chemical bonds have different strengths.
Examples include:
- Single bonds – generally weaker
- Double bonds – stronger than single bonds
- Triple bonds – typically the strongest
The more electrons shared between atoms, the stronger the bond usually becomes.
Atomic Size
Atoms with smaller sizes tend to form stronger bonds because their nuclei hold shared electrons more tightly.
When atoms are larger, the shared electrons are farther from the nucleus, which can weaken the bond.
Bond Length
Bond length refers to the distance between the nuclei of bonded atoms.
- Shorter bonds usually have higher bond energy.
- Longer bonds usually have lower bond energy.
This relationship helps explain why some molecules are more stable than others.
Examples of Bond Energy Differences
Different chemical bonds require different amounts of energy to break.
For example:
- Hydrogen–hydrogen bonds require moderate energy.
- Oxygen–oxygen double bonds require more energy.
- Nitrogen–nitrogen triple bonds are extremely strong and require a large amount of energy to break.
These differences help determine how substances behave during chemical reactions.
Why Bond Energy Matters in Chemistry
Bond energy plays a key role in understanding chemical reactions and molecular stability.
Chemists use bond energy to:
- Predict whether reactions will release or absorb energy
- Estimate reaction energy changes
- Understand molecular stability
- Design chemical processes in laboratories and industries
For example, fuels release energy during combustion because the new bonds formed in the reaction release more energy than the bonds that were broken.
Bond Energy and Molecular Stability
Molecules with high bond energy tend to be more stable because their atoms are strongly connected.
Stable molecules are generally less likely to react unless enough energy is provided to break their bonds.
On the other hand, molecules with lower bond energy may react more easily because their bonds require less energy to break.
This concept helps chemists predict which substances will react quickly and which will remain relatively unchanged.
Bond Energy in Everyday Applications
Chemical bond energy is involved in many processes encountered in everyday life.
Examples include:
Fuel Combustion
Fuels such as gasoline or natural gas release energy when their chemical bonds break and new bonds form during combustion.
Metabolism in the Human Body
Biological molecules release energy when bonds break during metabolic reactions that power cells.
Industrial Chemistry
Chemical manufacturing processes rely on understanding bond energy to control reaction conditions and improve efficiency.
Final Thoughts
Chemical bond energy measures how much energy is required to break the bonds that hold atoms together in molecules. It is a key concept for understanding how chemical reactions occur and why some molecules are more stable than others.
By studying bond energy, chemists can predict reaction behavior, design efficient chemical processes, and better understand the structure and stability of molecules.