Intermolecular forces are the attractions that occur between molecules. While these forces are generally weaker than the chemical bonds that hold atoms together within a molecule, they play a crucial role in determining the physical properties of substances.
Intermolecular forces influence how molecules interact with one another in liquids, solids, and gases. They help explain properties such as boiling point, melting point, viscosity, solubility, and surface tension.
Understanding these forces allows chemists to predict how substances behave in different environments and why certain materials interact the way they do.
What Are Intermolecular Forces?
Intermolecular forces are attractive forces between separate molecules. Unlike chemical bonds, which hold atoms together within a molecule, intermolecular forces operate between entire molecules.
These forces arise because of interactions between electric charges, partial charges, or temporary charge fluctuations in molecules.
Key characteristics include:
- Weaker than covalent or ionic bonds
- Responsible for many physical properties of substances
- Important in liquids and solids where molecules are close together
Although relatively weak individually, these forces collectively have a significant impact on how matter behaves.
Why Intermolecular Forces Matter
Intermolecular forces influence many observable properties of substances.
Physical state of matter
The strength of intermolecular forces affects whether a substance exists as a gas, liquid, or solid at a given temperature.
For example:
- Weak forces → substances often exist as gases
- Stronger forces → substances form liquids or solids
Boiling and melting points
Stronger intermolecular forces require more energy to overcome.
This results in:
- Higher boiling points
- Higher melting points
Solubility
Intermolecular forces determine whether substances dissolve in one another.
A common rule is:
“Like dissolves like.”
Polar molecules tend to dissolve in polar solvents, while nonpolar molecules dissolve in nonpolar solvents.
Types of Intermolecular Forces
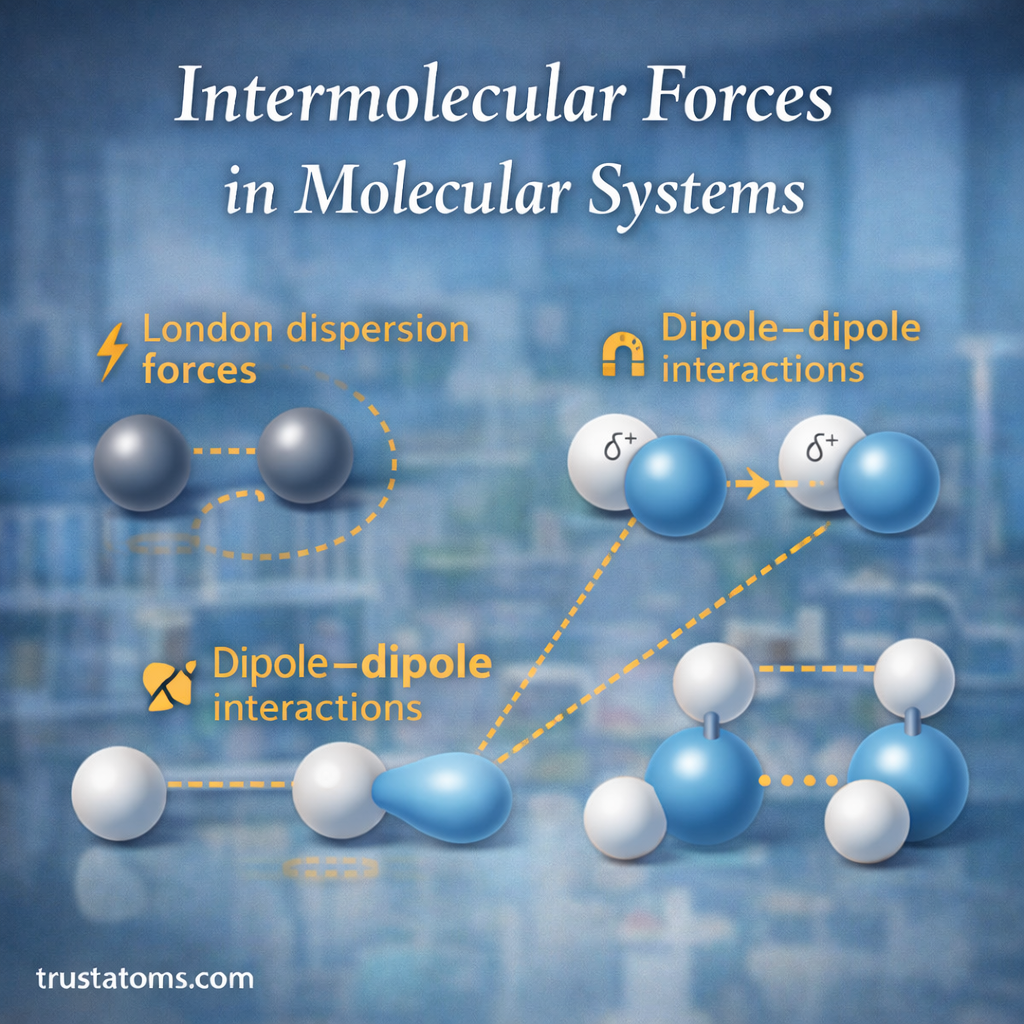
There are several main types of intermolecular forces that occur in molecular systems.
The three most commonly discussed include:
- London dispersion forces
- Dipole–dipole interactions
- Hydrogen bonding
Each type varies in strength and occurs under different molecular conditions.
London Dispersion Forces
London dispersion forces are the weakest and most universal intermolecular force. They occur in all molecules, including nonpolar ones.
These forces arise due to temporary fluctuations in electron distribution within atoms or molecules.
How dispersion forces form
Electrons constantly move around atoms. Occasionally, electrons may shift to one side of a molecule, creating a temporary uneven distribution of charge.
This creates:
- A temporary dipole in one molecule
- An induced dipole in a nearby molecule
These temporary attractions create dispersion forces.
Key characteristics
- Present in all molecules
- Stronger in larger atoms and molecules
- Increase with molecular size and electron count
Examples include noble gases and nonpolar molecules like methane.
Dipole–Dipole Interactions
Dipole–dipole forces occur between polar molecules.
Polar molecules have an uneven distribution of electrons, which creates:
- A partially positive end
- A partially negative end
These permanent dipoles attract one another.
How dipole–dipole forces work
The positive end of one molecule attracts the negative end of another molecule.
This creates intermolecular attraction that is stronger than dispersion forces in many cases.
Common examples
- Hydrogen chloride (HCl)
- Sulfur dioxide (SO₂)
These molecules have permanent dipoles that cause them to attract neighboring molecules.
Hydrogen Bonding
Hydrogen bonding is a strong type of dipole–dipole interaction that occurs when hydrogen atoms bond with highly electronegative atoms.
Hydrogen bonding typically occurs when hydrogen is bonded to:
- Oxygen
- Nitrogen
- Fluorine
These atoms pull electron density toward themselves, leaving hydrogen with a strong partial positive charge.
How hydrogen bonding forms
The partially positive hydrogen atom of one molecule is attracted to a lone pair of electrons on a nearby electronegative atom.
This interaction creates a particularly strong intermolecular force.
Examples of hydrogen bonding
- Water (H₂O)
- Ammonia (NH₃)
- Hydrogen fluoride (HF)
Hydrogen bonding is responsible for many unusual properties of water, including its relatively high boiling point.
Relative Strength of Intermolecular Forces
Different intermolecular forces vary in strength.
A general ranking from weakest to strongest is:
- London dispersion forces
- Dipole–dipole interactions
- Hydrogen bonding
Even though dispersion forces are the weakest individually, they can become significant in large molecules where many interactions occur simultaneously.
Factors That Affect Intermolecular Forces
Several factors influence the strength of intermolecular forces in a molecular system.
Molecular size
Larger molecules tend to have stronger dispersion forces because they contain more electrons and larger electron clouds.
Molecular shape
The shape of a molecule affects how closely molecules can approach one another.
Molecules with larger surface areas may experience stronger dispersion forces.
Polarity
Polar molecules experience dipole–dipole interactions in addition to dispersion forces.
This generally results in stronger intermolecular attractions compared to nonpolar molecules.
Intermolecular Forces and Everyday Chemistry
Intermolecular forces play an important role in many real-world phenomena.
Surface tension
Surface tension occurs when molecules at the surface of a liquid experience strong intermolecular attractions.
Water’s high surface tension results from hydrogen bonding.
Evaporation
Liquids evaporate when molecules gain enough energy to overcome intermolecular attractions.
Substances with weaker intermolecular forces evaporate more easily.
Biological systems
Intermolecular forces help stabilize biological structures such as:
- Protein folding
- DNA structure
- Enzyme interactions
These forces allow biological molecules to interact precisely with one another.
Intermolecular Forces vs Chemical Bonds
It is important to distinguish intermolecular forces from chemical bonds.
Chemical bonds
Chemical bonds occur within molecules and include:
- Covalent bonds
- Ionic bonds
These bonds involve the sharing or transfer of electrons and are relatively strong.
Intermolecular forces
Intermolecular forces occur between molecules and are significantly weaker.
However, they still play a critical role in determining the physical behavior of substances.
Final Thoughts
Intermolecular forces are essential for understanding how molecules interact in liquids, solids, and gases. Although these forces are weaker than chemical bonds, they strongly influence physical properties such as boiling point, solubility, and viscosity.
By studying London dispersion forces, dipole–dipole interactions, and hydrogen bonding, chemists can better predict how substances behave in both natural and industrial systems. These interactions are fundamental to chemistry and help explain many processes that occur in everyday life.








