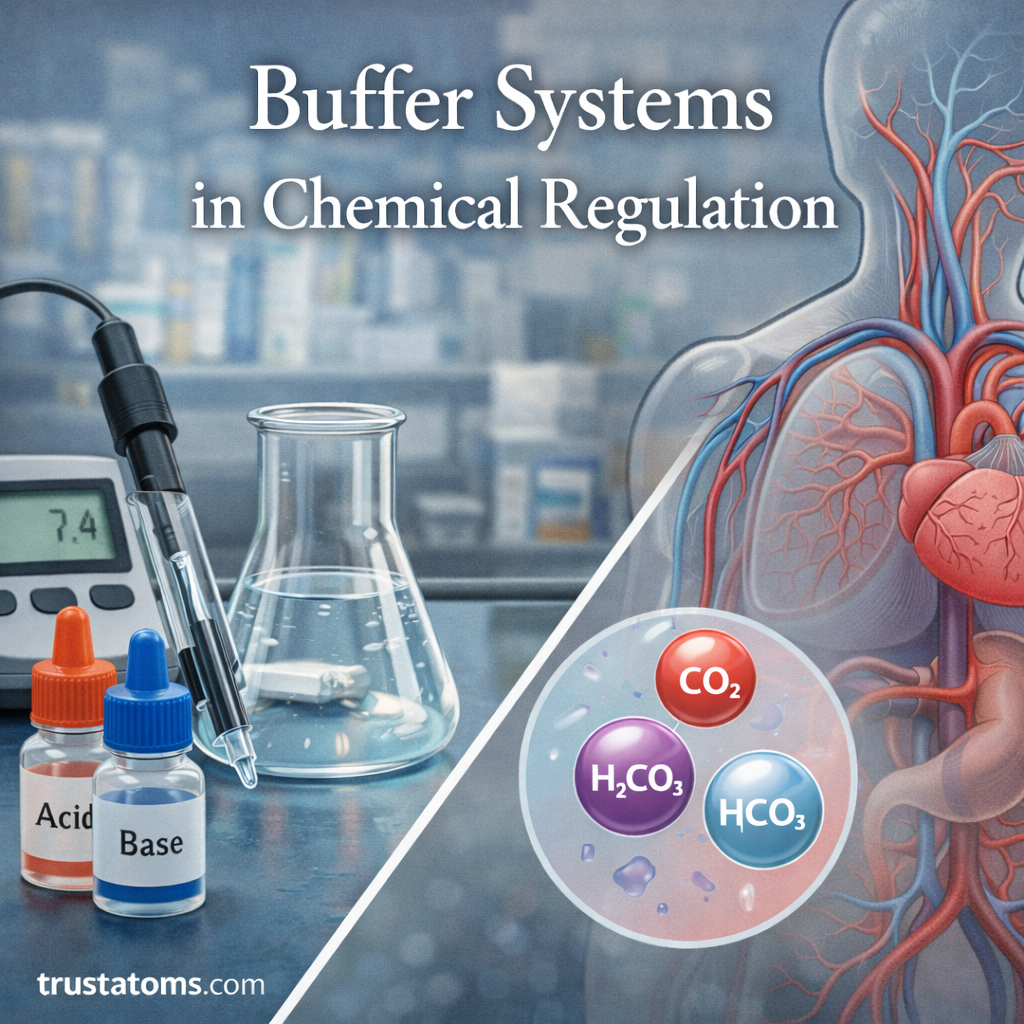
Buffer systems play a crucial role in maintaining chemical stability in many natural and industrial environments. In chemistry, buffers help resist sudden changes in pH when acids or bases are added to a solution.
This ability to regulate pH is essential in biological systems, environmental chemistry, pharmaceuticals, and laboratory experiments. Without buffer systems, many chemical reactions would become unstable or unpredictable due to rapid shifts in acidity or alkalinity.
Understanding how buffer systems work helps explain how living organisms regulate internal chemistry and how scientists maintain controlled conditions in chemical processes.
What Is a Buffer System?
A buffer system is a solution that resists significant changes in pH when small amounts of acid or base are added.
Buffers work through a combination of two components:
- A weak acid
- Its conjugate base
or
- A weak base
- Its conjugate acid
These paired substances work together to neutralize added acids or bases, helping maintain a relatively constant pH level.
Because weak acids and bases only partially dissociate in water, they can both release and absorb hydrogen ions when needed. This dynamic balance allows the solution to stabilize its pH.
Why pH Regulation Is Important
Many chemical reactions are highly sensitive to pH levels. Even small changes in acidity can alter reaction rates, molecular stability, or biological function.
Buffer systems are important because they:
- Maintain stable chemical environments
- Prevent sudden shifts in acidity
- Support biological and biochemical reactions
- Protect molecules from degradation
- Allow laboratory experiments to remain consistent
In biological systems especially, maintaining a narrow pH range is essential for proper function.
How Buffer Systems Work
Buffers regulate pH by neutralizing added acids or bases.
When Acid Is Added
If additional hydrogen ions enter the solution:
- The conjugate base component of the buffer reacts with the hydrogen ions.
- This reaction prevents a large increase in acidity.
When Base Is Added
If hydroxide ions are introduced:
- The weak acid component of the buffer donates hydrogen ions.
- This neutralizes the added base and prevents a large pH increase.
Because both components are present in equilibrium, the system can adjust to changes in either direction.
Components of a Typical Buffer
A functioning buffer requires two key ingredients that exist in chemical equilibrium.
Weak Acid
The weak acid provides hydrogen ions when necessary. Because it only partially dissociates, it can both release and retain hydrogen ions depending on the conditions.
Conjugate Base
The conjugate base can accept hydrogen ions when excess acid is introduced. This ability stabilizes the system when acidity increases.
Together, these two components form a chemical balance that maintains relatively stable pH levels.
The Buffer Capacity Concept
Buffer capacity refers to how effectively a buffer system can resist changes in pH.
Several factors influence buffer capacity:
- The concentration of buffer components
- The ratio of acid to conjugate base
- The amount of acid or base added
A buffer with higher concentrations of its components generally has greater capacity to absorb pH changes.
However, every buffer has a limit. If too much acid or base is added, the buffering components become overwhelmed and the pH will begin to change rapidly.
Common Buffer Systems in Chemistry
Several buffer systems are widely used in laboratories, industry, and biological research.
Acetic Acid–Acetate Buffer
This buffer consists of a weak organic acid paired with its conjugate base. It is commonly used in laboratory experiments that require a mildly acidic environment.
Phosphate Buffer System
Phosphate buffers are widely used in biological research because they operate effectively within pH ranges similar to many living systems.
Ammonia–Ammonium Buffer
This system involves a weak base and its conjugate acid. It is often used in analytical chemistry and industrial chemical processes.
Each buffer system works best within a specific pH range determined by the chemical properties of its components.
Buffer Systems in Biological Regulation
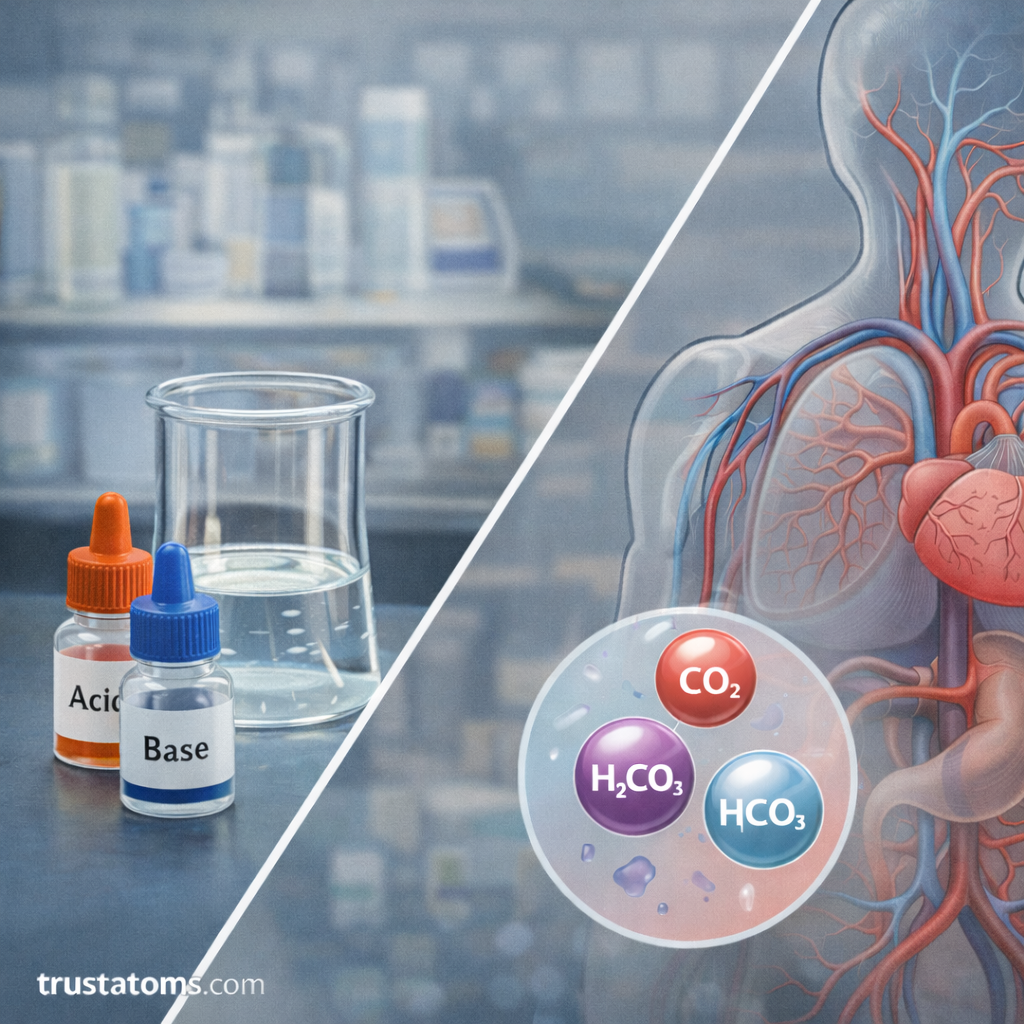
Living organisms rely heavily on buffer systems to maintain stable internal environments.
Biochemical reactions inside cells require tightly controlled pH conditions. Even small shifts in acidity can disrupt enzymes and metabolic processes.
One of the most important biological buffers is the bicarbonate buffer system, which helps regulate the pH of blood.
This system works through interactions between:
- Carbon dioxide
- Carbonic acid
- Bicarbonate ions
Together, these components maintain the delicate acid–base balance required for normal physiological function.
Industrial and Environmental Applications
Buffer systems are not limited to biological chemistry. They also play an important role in environmental science and industry.
Water Treatment
Buffers help stabilize pH levels in water treatment facilities to protect aquatic ecosystems and maintain water quality.
Pharmaceutical Manufacturing
Drug formulations often rely on buffers to maintain chemical stability and ensure medications remain effective during storage.
Food and Beverage Production
Many foods and beverages contain buffer systems that help maintain flavor stability, texture, and shelf life.
Chemical Manufacturing
Industrial processes frequently require precise pH control to ensure reactions proceed correctly and safely.
Factors That Influence Buffer Effectiveness
Several conditions determine how well a buffer system performs.
Important factors include:
- The pH range of the buffer
- The relative concentrations of acid and base components
- The temperature of the solution
- The total volume of the system
Selecting the correct buffer for a specific chemical process requires understanding these factors and matching them to the desired pH conditions.
Advantages of Using Buffer Systems
Buffer systems provide several advantages in chemistry and biology.
Key benefits include:
- Stabilizing chemical reactions
- Protecting sensitive biological molecules
- Supporting enzyme activity
- Maintaining consistent experimental conditions
- Improving reliability in industrial processes
Because many chemical reactions depend on controlled pH environments, buffers are essential tools in both research and applied chemistry.
Final Thoughts
Buffer systems are fundamental to chemical regulation. By combining weak acids and their conjugate bases, these systems resist changes in pH and maintain stable environments for chemical reactions.
From laboratory experiments to biological processes and industrial production, buffers help ensure that chemical systems remain balanced and predictable.
Understanding how buffer systems function provides valuable insight into how chemists control reactions, stabilize solutions, and support the delicate chemical balance required in many natural and technological systems.