Chemical reactions often appear simple on the surface—reactants transform into products. However, most reactions actually occur through a sequence of smaller steps called reaction mechanisms. Understanding how fast these steps occur and how they interact is the focus of chemical kinetics.
Chemical kinetics in reaction mechanisms helps scientists explain why reactions occur at certain speeds, how intermediate compounds form, and what factors influence the overall reaction pathway.
What Is Chemical Kinetics?
Chemical kinetics is the branch of chemistry that studies reaction rates and the processes that influence how quickly reactions occur.
Instead of focusing only on what substances react, kinetics explores:
- How fast a reaction proceeds
- What steps occur during the transformation
- What molecular interactions drive the reaction
- How conditions influence the speed of the reaction
Kinetics provides insight into the dynamic behavior of molecules during chemical change.
Understanding Reaction Mechanisms
A reaction mechanism describes the detailed sequence of elementary steps that lead from reactants to products.
Most chemical reactions do not happen in a single step. Instead, they involve several intermediate stages.
These mechanisms typically include:
- Elementary steps – individual molecular events
- Reaction intermediates – temporary species formed during the reaction
- Transition states – high-energy arrangements of atoms during bond changes
By studying kinetics, chemists can determine which steps control the overall reaction rate.
Elementary Steps in Reaction Mechanisms
Elementary steps represent the simplest events in a reaction mechanism. Each step describes a direct molecular interaction.
Common types include:
Unimolecular Steps
A single molecule rearranges or breaks apart.
Example process:
- A molecule decomposes into smaller products.
Bimolecular Steps
Two molecules collide and react.
These steps involve:
- Bond formation
- Bond breaking
- Rearrangement of atoms
Termolecular Steps
Three molecules interact simultaneously.
However, these are rare because simultaneous three-body collisions are statistically unlikely.
Reaction Intermediates
During many reactions, temporary species form before the final products appear. These are called reaction intermediates.
Intermediates are:
- Produced in one step of a mechanism
- Consumed in a later step
- Not present in the overall balanced equation
Examples of intermediates include:
- Free radicals
- Carbocations
- Carbanions
- Activated molecular complexes
Although intermediates exist briefly, they often determine which pathway the reaction follows.
The Role of the Rate-Determining Step
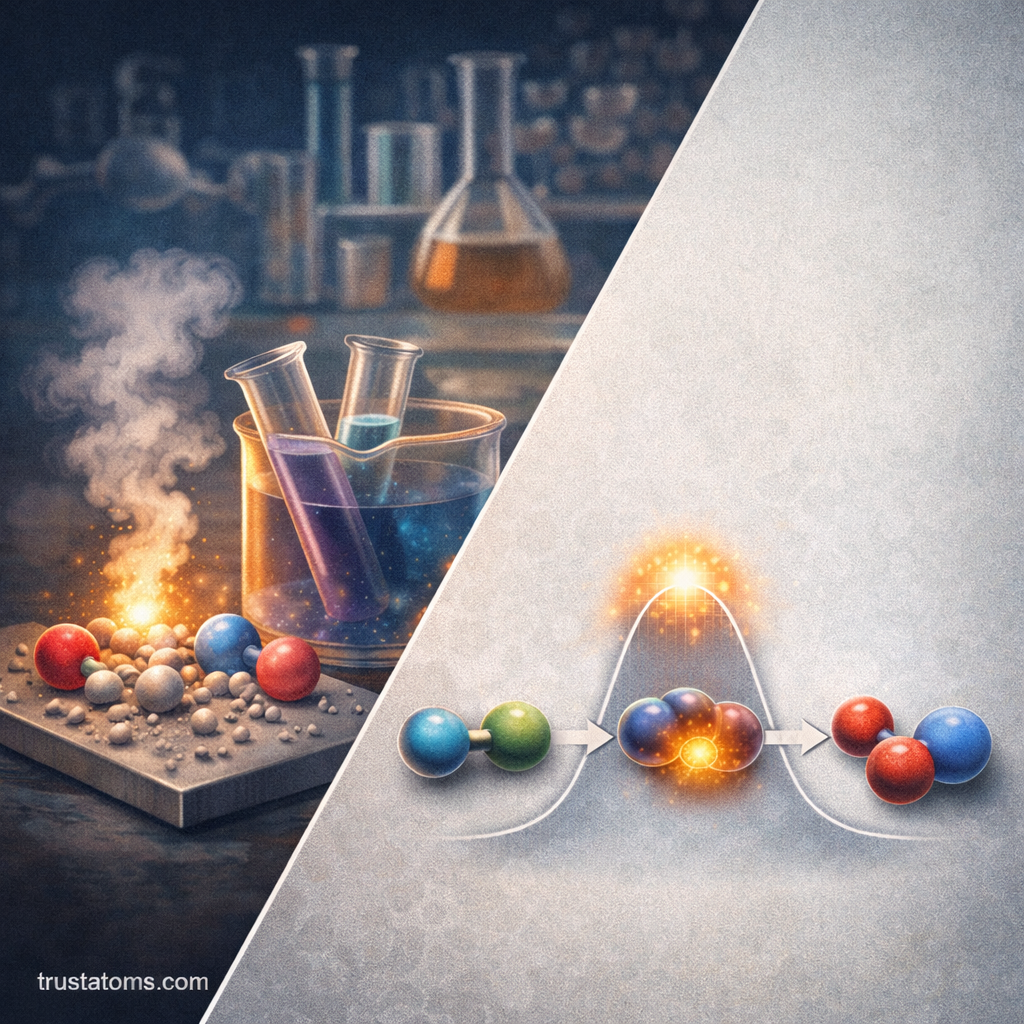
In multi-step reactions, one step is usually slower than the others. This is known as the rate-determining step.
The rate-determining step:
- Acts as a bottleneck for the reaction
- Controls the overall reaction speed
- Determines the observable rate law
If a slow step occurs early in the mechanism, it can significantly delay the formation of products.
Understanding this step is crucial when designing catalysts or optimizing industrial reactions.
Factors That Influence Reaction Rates
Several conditions influence the speed of reaction mechanisms.
Temperature
Higher temperatures increase molecular motion, leading to:
- More frequent molecular collisions
- Greater energy during collisions
- Higher probability of successful reactions
Concentration
Increasing reactant concentration leads to:
- More molecules in a given volume
- Higher collision frequency
- Faster reaction rates
Catalysts
Catalysts accelerate reactions by providing an alternative pathway with lower energy requirements.
Catalysts may:
- Stabilize reaction intermediates
- Lower energy barriers
- Improve reaction efficiency
Surface Area
For reactions involving solids, greater surface area allows more molecules to interact at once.
This is why powdered materials often react faster than solid chunks.
Collision Theory and Reaction Mechanisms
Chemical kinetics often uses collision theory to explain reaction rates.
According to this theory, molecules must collide in order to react. However, not every collision produces a reaction.
Successful collisions require:
- Sufficient energy to overcome activation barriers
- Proper molecular orientation
- Close contact between reacting atoms
Reaction mechanisms describe how these successful collisions lead to intermediate formation and final products.
Activation Energy and Reaction Pathways
Every reaction requires a minimum amount of energy known as activation energy.
Activation energy represents the energy needed to:
- Break existing bonds
- Form new chemical bonds
- Reach the transition state
In multi-step mechanisms, each elementary step has its own activation energy. The highest energy barrier typically corresponds to the rate-determining step.
Lower activation energy generally leads to faster reactions.
Experimental Methods for Studying Reaction Mechanisms
Chemists use several experimental techniques to understand reaction kinetics.
Common methods include:
- Monitoring concentration changes over time
- Measuring reaction rate constants
- Detecting reaction intermediates
- Using spectroscopy to observe molecular behavior
By analyzing these observations, scientists can infer the most likely reaction pathway.
Real-World Applications of Chemical Kinetics
Chemical kinetics plays an important role in many scientific and industrial fields.
Pharmaceutical Development
Drug synthesis relies on understanding reaction mechanisms to improve:
- Efficiency
- Yield
- Safety
Environmental Chemistry
Kinetic studies help scientists understand processes such as:
- Atmospheric reactions
- Pollution breakdown
- Ozone chemistry
Industrial Manufacturing
Industries optimize reactions to increase productivity in processes like:
- Polymer production
- Fuel refinement
- Chemical manufacturing
Understanding reaction kinetics allows engineers to design faster, safer, and more efficient chemical processes.
Why Reaction Mechanisms Matter in Chemistry
Chemical kinetics provides a deeper understanding of how reactions actually occur at the molecular level.
Studying reaction mechanisms helps chemists:
- Predict reaction behavior
- Improve industrial processes
- Develop new materials and medicines
- Control reaction outcomes
Rather than viewing reactions as simple transformations, kinetics reveals the step-by-step journey molecules take during chemical change.
Final Thoughts
Chemical kinetics in reaction mechanisms bridges the gap between chemical equations and real molecular behavior. By analyzing reaction rates, intermediate species, and energy barriers, chemists can uncover the pathways that guide chemical transformations.
This understanding allows scientists to control reactions more effectively, design better catalysts, and develop innovative technologies across science and industry.