Coordination compounds are an essential part of inorganic chemistry, playing a major role in biological systems, industrial processes, and modern materials science. These compounds consist of a central metal atom or ion bonded to surrounding molecules or ions known as ligands.
From oxygen transport in blood to catalysts used in chemical manufacturing, coordination compounds appear in many natural and technological systems. Understanding how these compounds form, behave, and interact helps chemists design new medicines, improve industrial reactions, and develop advanced materials.
What Are Coordination Compounds?
A coordination compound is a chemical structure where a central metal atom or metal ion is surrounded by ligands, which are molecules or ions capable of donating electron pairs to the metal.
These interactions form coordinate covalent bonds, meaning the electrons in the bond are donated entirely by the ligand.
A typical coordination compound contains two main components:
- Central metal ion – usually a transition metal
- Ligands – molecules or ions attached to the metal
Common transition metals involved include:
- Iron
- Copper
- Cobalt
- Nickel
- Platinum
These metals are particularly suitable because they can accept electron pairs and form multiple bonds with surrounding ligands.
Key Components of Coordination Compounds
Understanding coordination compounds requires recognizing their structural elements.
Central Metal Ion
The central metal ion acts as the coordination center. It accepts electron pairs from ligands and forms bonds that stabilize the overall complex.
Transition metals are commonly used because they have:
- Partially filled d-orbitals
- Multiple oxidation states
- Ability to form stable complexes
Ligands
Ligands are ions or molecules that donate a pair of electrons to the metal center.
Common ligands include:
- Water (H₂O)
- Ammonia (NH₃)
- Chloride ions (Cl⁻)
- Cyanide ions (CN⁻)
Ligands may be classified based on how many bonds they form with the metal.
Types of ligands include:
- Monodentate ligands – attach through one donor atom
- Bidentate ligands – attach through two donor atoms
- Polydentate ligands – attach through multiple donor atoms
Polydentate ligands often form chelate complexes, which are particularly stable.
Coordination Number
The coordination number refers to the number of ligand atoms directly bonded to the central metal ion.
Common coordination numbers include:
- 2
- 4
- 6
For example, a coordination number of six typically results in an octahedral structure.
Structure and Geometry of Coordination Complexes
Coordination compounds can adopt several geometric arrangements depending on the metal and ligands involved.
Linear Geometry
Occurs when the coordination number is 2.
Characteristics:
- Two ligands bonded to the metal
- 180° bond angle
Example metals that form linear complexes include silver and gold.
Tetrahedral Geometry
Occurs with coordination number 4.
Characteristics:
- Four ligands arranged around the metal
- Bond angles near 109.5°
This geometry is common in complexes involving zinc and nickel.
Octahedral Geometry
Occurs with coordination number 6.
Characteristics:
- Six ligands surrounding the metal
- Highly symmetrical structure
- One of the most common geometries in coordination chemistry
Octahedral complexes appear frequently in transition metal chemistry and biological molecules.
Naming Coordination Compounds
Coordination compounds follow a systematic naming method established by IUPAC.
The naming process generally follows these steps:
- Name the ligands first
- List them alphabetically
- Name the central metal
- Indicate the metal’s oxidation state in Roman numerals
Ligands also use modified names depending on their form. For example:
- Chloride becomes chloro
- Cyanide becomes cyano
- Water becomes aqua
- Ammonia becomes ammine
This standardized system allows chemists around the world to describe coordination compounds clearly and consistently.
Importance of Coordination Compounds in Biology

Many essential biological molecules are coordination complexes.
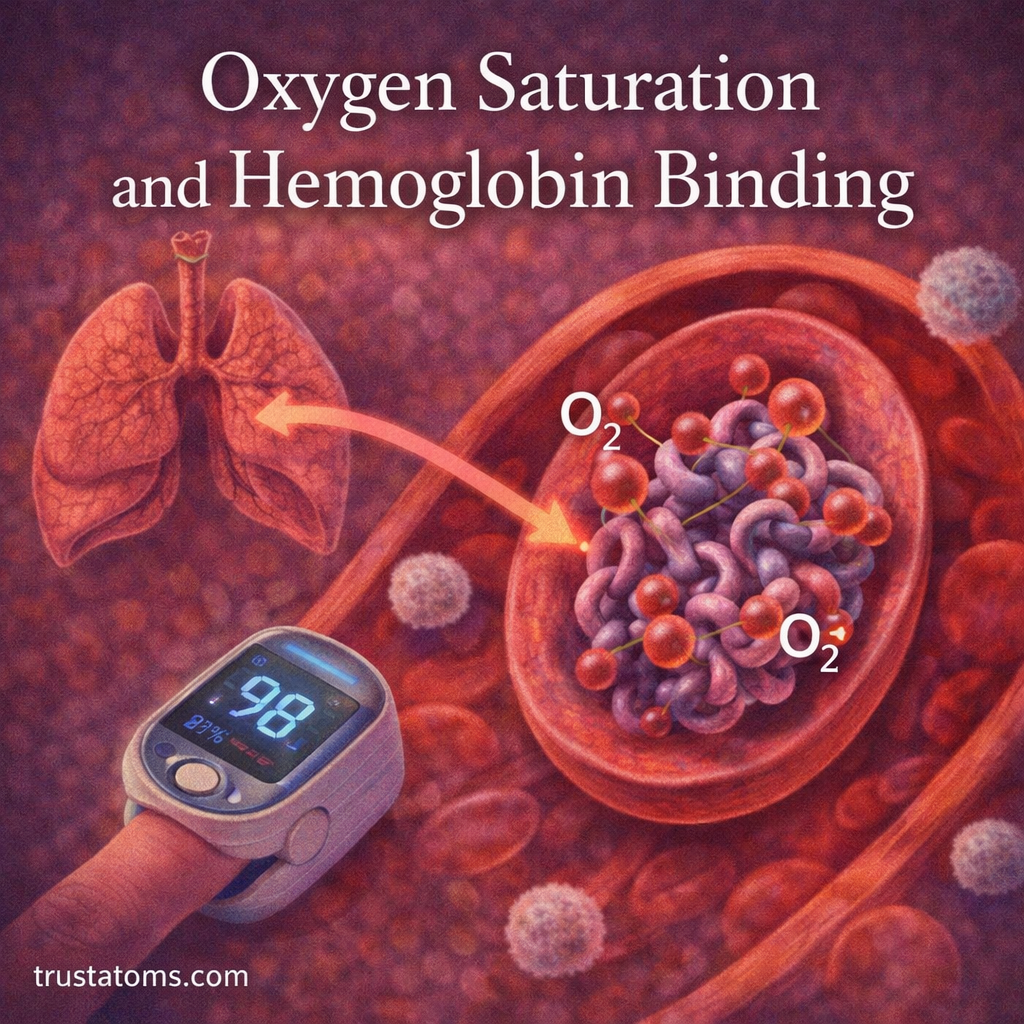
Hemoglobin
Hemoglobin contains an iron coordination complex that allows red blood cells to transport oxygen throughout the body.
The iron atom binds oxygen molecules reversibly, enabling oxygen delivery to tissues.
Chlorophyll
Chlorophyll, the molecule responsible for photosynthesis in plants, contains a magnesium coordination center.
This metal complex allows plants to capture sunlight and convert it into chemical energy.
Vitamin B12
Vitamin B12 includes a cobalt coordination complex that plays a vital role in metabolism and nerve function.
These examples demonstrate how coordination chemistry supports life at the molecular level.
Industrial Applications of Coordination Compounds
Coordination compounds are widely used in industrial chemistry and materials science.
Catalysts
Many industrial catalysts are coordination complexes.
They help accelerate reactions used to produce:
- Plastics
- Fuels
- Pharmaceuticals
- Agricultural chemicals
Transition metal complexes often provide the active sites for these catalytic reactions.
Electroplating
Metal coordination compounds are used in electroplating processes that coat materials with thin layers of metal for:
- Corrosion resistance
- Decorative finishes
- Electrical conductivity
Medical Treatments
Several important medicines are coordination compounds.
Examples include:
- Anti-cancer drugs
- Imaging agents used in diagnostics
- Antimicrobial treatments
These compounds interact with biological molecules in controlled ways to achieve therapeutic effects.
The Role of Ligand Field Theory
To understand the behavior of coordination compounds, chemists use models such as ligand field theory.
This theory explains how ligands influence the energy levels of electrons in the metal’s d-orbitals.
Key effects include:
- Color of coordination compounds
- Magnetic properties
- Stability of complexes
- Reactivity in chemical reactions
Different ligands create different energy environments, which determines how the complex behaves chemically and physically.
Why Coordination Chemistry Matters
Coordination chemistry is important across multiple scientific fields.
Its applications include:
- Understanding biological molecules
- Designing pharmaceutical drugs
- Creating advanced catalysts
- Developing electronic and magnetic materials
- Improving environmental remediation technologies
Because transition metals interact strongly with surrounding molecules, coordination compounds remain one of the most versatile and widely studied topics in inorganic chemistry.
Final Thoughts
Coordination compounds reveal how metal ions interact with surrounding molecules to form stable and functional structures. These complexes influence everything from oxygen transport in living organisms to large-scale chemical manufacturing.
By studying coordination chemistry, scientists gain insights into molecular structure, reactivity, and material design. As research advances, coordination compounds will continue to play a key role in medicine, environmental science, and modern technology.