Macromolecular chemistry focuses on the study of very large molecules called macromolecules, which are made by linking many smaller repeating units together. These large molecules—commonly known as polymers—are essential to modern life. They appear in plastics, rubbers, fibers, adhesives, coatings, and even biological systems like proteins and DNA.
One of the most important concepts in polymer science is polymer structure. The arrangement of repeating units and how polymer chains are organized determines a material’s flexibility, strength, heat resistance, and chemical stability.
Understanding polymer structures helps chemists design materials with specific properties for applications ranging from medical implants to aerospace engineering.
What Are Polymers?
A polymer is a large molecule made of repeating building blocks called monomers. These monomers chemically bond together to form long chains or networks.
Examples of common polymers include:
- Polyethylene (used in plastic bags and packaging)
- Polypropylene (used in containers and textiles)
- Polystyrene (used in insulation and foam products)
- Nylon (used in clothing and ropes)
- Natural polymers like cellulose, proteins, and DNA
The chemical structure and arrangement of these repeating units create different polymer architectures, which directly influence how the material behaves.
Basic Components of Polymer Structure
Polymer structure can be understood by examining several important structural levels.
Primary Structure
The primary structure refers to the chemical sequence of repeating units along the polymer chain.
This includes:
- The type of monomer used
- The order of monomers (for copolymers)
- The chemical bonds connecting the units
For example:
- A polymer made from a single repeating unit is called a homopolymer
- A polymer containing two or more different monomers is called a copolymer
The primary structure determines the basic chemical identity of the polymer.
Secondary Structure
Secondary structure involves local arrangements of polymer chains, including how they twist, fold, or interact with nearby chains.
In synthetic polymers, this may involve:
- Chain flexibility
- Rotation around bonds
- Local molecular packing
In biological macromolecules such as proteins, secondary structure becomes more complex, forming patterns like alpha-helices and beta-sheets.
Tertiary Structure
The tertiary structure describes the overall three-dimensional shape of a polymer chain in space.
Factors influencing tertiary structure include:
- Intermolecular forces
- Hydrogen bonding
- Van der Waals interactions
- Solvent interactions
This level of structure determines properties like elasticity, solubility, and mechanical strength.
Types of Polymer Chain Architectures
Beyond chemical composition, polymers are also classified by the shape and connectivity of their chains.
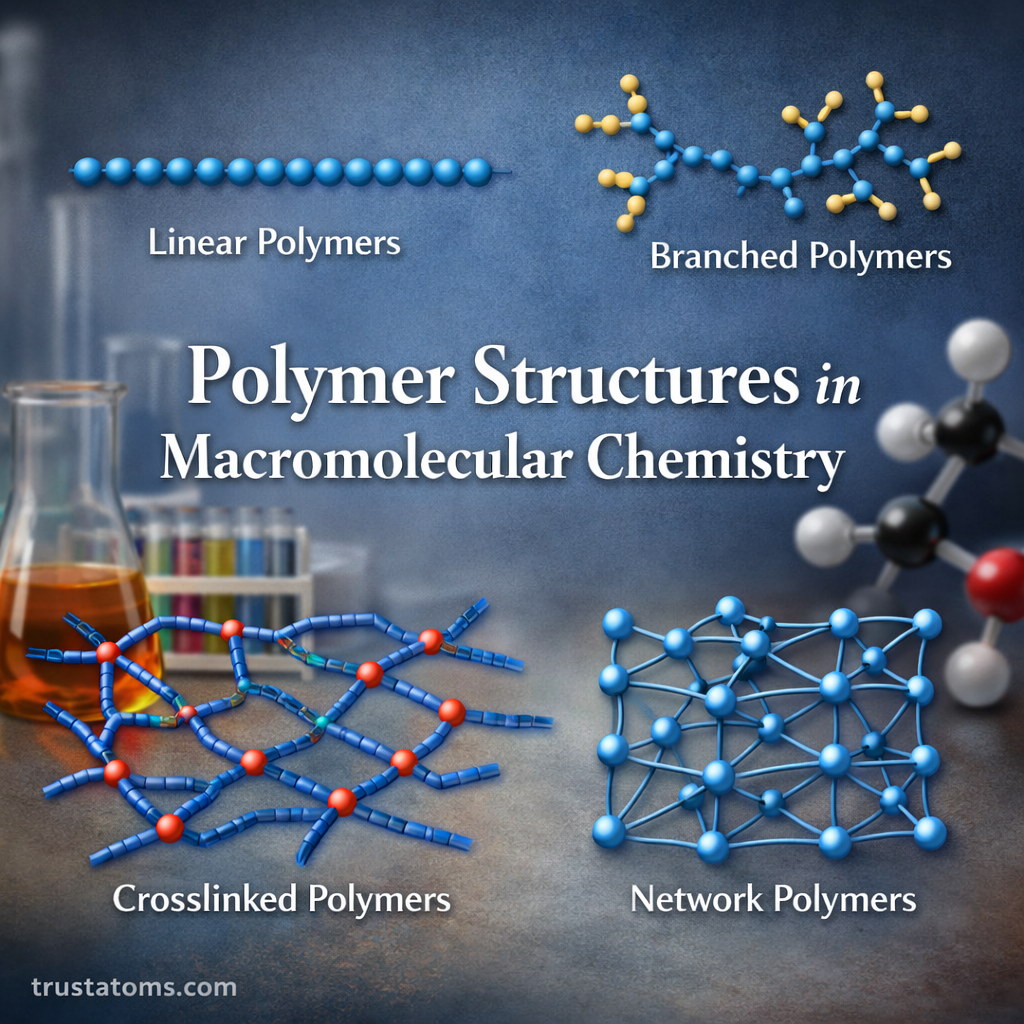
Linear Polymers
Linear polymers consist of long, straight chains where monomers are connected end-to-end.
Characteristics include:
- High packing efficiency
- Strong intermolecular interactions
- Good tensile strength
Examples include:
- High-density polyethylene (HDPE)
- Nylon
- Polyester
Linear polymers can align closely together, forming crystalline regions that improve strength.
Branched Polymers
Branched polymers contain side chains attached to the main backbone.
These branches prevent chains from packing tightly.
Key features:
- Lower density
- Increased flexibility
- Reduced crystallinity
An example is low-density polyethylene (LDPE), which is widely used in plastic films and packaging.
Cross-Linked Polymers
Cross-linked polymers form when individual polymer chains are chemically bonded together through linking points.
This structure creates a network that improves strength and stability.
Properties include:
- High rigidity
- Increased heat resistance
- Reduced solubility
Common examples:
- Epoxy resins
- Vulcanized rubber
- Thermosetting plastics
Cross-linking prevents chains from sliding past each other, making these materials durable.
Network Polymers
Network polymers are highly cross-linked materials forming three-dimensional structures.
These polymers are often rigid and resistant to deformation.
Examples include:
- Bakelite
- Melamine resins
- Phenolic plastics
Network polymers are widely used in electrical components, laminates, and adhesives due to their thermal stability.
Polymer Morphology and Structural Arrangement
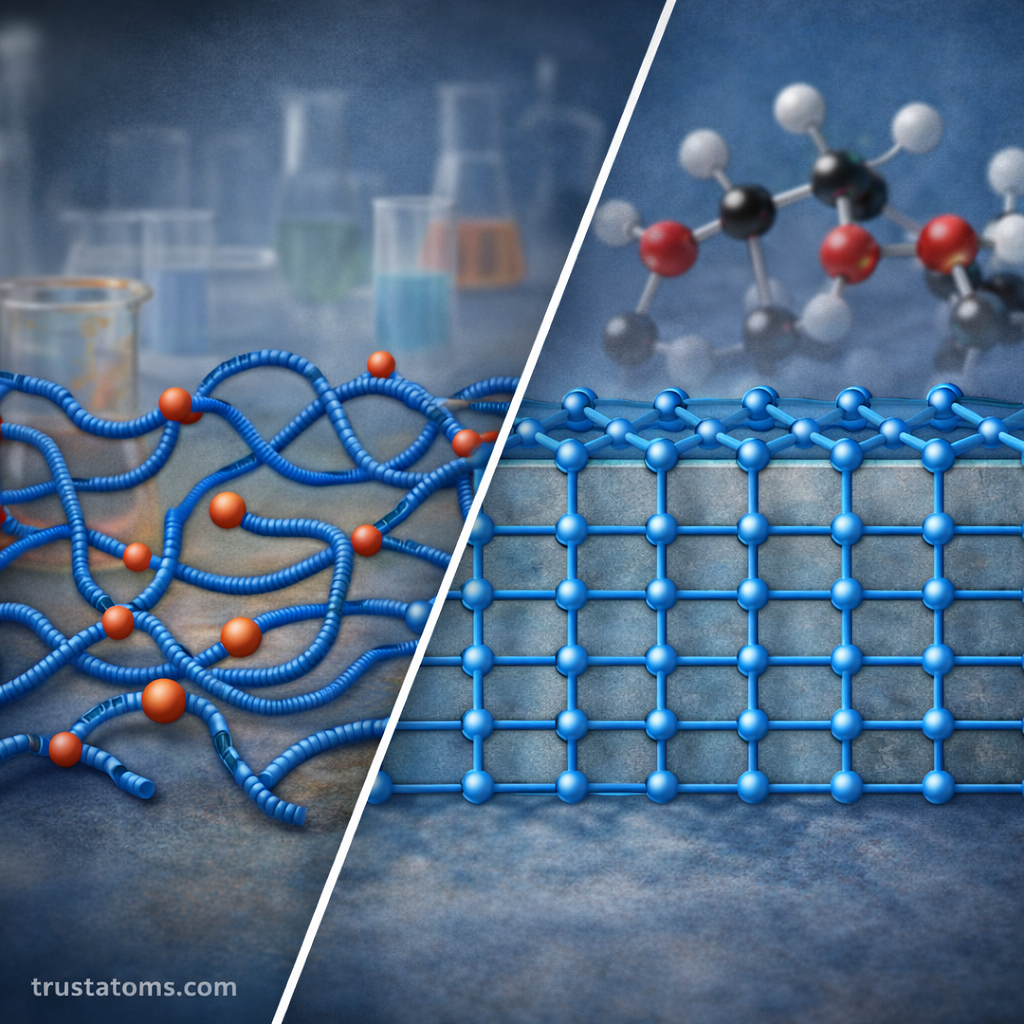
Polymer chains do not always arrange themselves randomly. Their spatial organization can influence material properties.
Two major structural arrangements include:
Amorphous Polymers
Amorphous polymers have randomly arranged chains without a clear repeating pattern.
Characteristics:
- Transparent or translucent appearance
- Flexible structure
- Lower melting temperatures
Examples include:
- Polystyrene
- Polycarbonate
- Acrylic (PMMA)
Crystalline and Semi-Crystalline Polymers
Some polymers form ordered regions where chains pack tightly together.
Most real polymers are semi-crystalline, meaning they contain both ordered and disordered regions.
Advantages of crystalline regions:
- Greater mechanical strength
- Higher melting points
- Improved chemical resistance
Examples include:
- Polyethylene
- Polypropylene
- Nylon
How Polymer Structure Influences Material Properties
The structure of a polymer directly affects its physical and chemical behavior.
Important relationships include:
Chain length
- Longer chains generally increase strength and toughness.
Branching
- More branching typically reduces density and crystallinity.
Cross-linking
- Increases rigidity, thermal stability, and resistance to deformation.
Chain mobility
- Flexible chains produce softer materials, while rigid chains produce stronger plastics.
Because of these relationships, polymer chemists can design materials with precise performance characteristics.
Applications of Polymer Structural Design
Understanding polymer structures allows scientists to create materials tailored for specific industries.
Examples include:
Packaging
Flexible polymers such as polyethylene provide lightweight and moisture-resistant packaging materials.
Medical Materials
Biocompatible polymers are used in:
- Surgical sutures
- Drug delivery systems
- Artificial joints
- Tissue engineering scaffolds
Textiles and Fibers
Synthetic fibers like polyester and nylon are engineered for strength, durability, and elasticity.
Electronics
Cross-linked polymers are used in circuit boards, insulation materials, and electronic coatings.
Aerospace and Automotive Engineering
Advanced polymer composites provide:
- Lightweight structural materials
- Heat-resistant coatings
- Impact-resistant components
The Future of Polymer Structure Research
Modern research in macromolecular chemistry focuses on designing advanced polymer architectures with highly specialized functions.
Emerging developments include:
- Self-healing polymers
- Biodegradable plastics
- Smart polymers that respond to temperature or pH
- Conductive polymers used in electronics
- Nanostructured polymer materials
These innovations are helping address environmental challenges while improving material performance.
Final Thoughts
Polymer structures play a central role in macromolecular chemistry and material science. From simple linear chains to complex cross-linked networks, the arrangement of polymer molecules determines how materials behave under real-world conditions.
By controlling polymer architecture, scientists can design materials that are stronger, lighter, more flexible, or more heat-resistant depending on the application.
As research continues, understanding polymer structure will remain essential for developing sustainable materials, advanced technologies, and next-generation industrial products.