Infrared spectroscopy (IR spectroscopy) is one of the most widely used analytical techniques in chemistry for identifying chemical compounds. By examining how molecules absorb infrared light, scientists can determine the types of chemical bonds present in a substance and often identify the compound itself.
Because each molecule absorbs infrared radiation in a unique way, infrared spectroscopy provides a kind of molecular fingerprint. This makes the technique extremely valuable in research laboratories, environmental testing, pharmaceuticals, and forensic science.
This guide explains how infrared spectroscopy works, why it is useful for chemical identification, and where it is commonly applied in modern science.
What Is Infrared Spectroscopy?
Infrared spectroscopy is a technique that measures how molecules interact with infrared radiation, a portion of the electromagnetic spectrum located between visible light and microwaves.
When infrared light passes through or reflects from a chemical sample, certain wavelengths are absorbed. These absorbed wavelengths correspond to vibrations within the molecule’s chemical bonds.
By analyzing which wavelengths are absorbed, scientists can determine:
- The types of chemical bonds present
- Functional groups within a molecule
- The identity of unknown compounds
The resulting output is called an infrared spectrum, which displays absorption patterns specific to a particular substance.
The Role of Infrared Light in Molecular Vibrations
Atoms within molecules are constantly moving. They vibrate by stretching, bending, or twisting relative to each other.
Infrared radiation interacts with these vibrations when the energy of the radiation matches the natural vibration frequency of a chemical bond.
Common types of molecular vibrations include:
- Stretching vibrations (atoms move closer and farther apart)
- Bending vibrations (bond angles change)
- Twisting or rocking motions within molecules
When a bond absorbs infrared energy, its vibration increases. Spectroscopic instruments detect this absorption and convert it into measurable data.
Understanding an Infrared Spectrum
An infrared spectrum is typically displayed as a graph showing how much infrared radiation a molecule absorbs at different wavelengths.
The spectrum usually includes:
- A horizontal axis representing wavelength or frequency
- A vertical axis representing absorbance or transmission
Different regions of the spectrum correspond to specific molecular vibrations.
Scientists analyze these regions to identify chemical structures.
The Functional Group Region
One of the most important areas of an infrared spectrum is the functional group region.
This region typically contains absorption signals produced by common chemical bonds, including:
- Oxygen–hydrogen bonds
- Carbon–hydrogen bonds
- Carbon–oxygen bonds
- Nitrogen–hydrogen bonds
Because many functional groups absorb infrared radiation at predictable wavelengths, scientists can quickly determine which groups are present in a molecule.
The Fingerprint Region
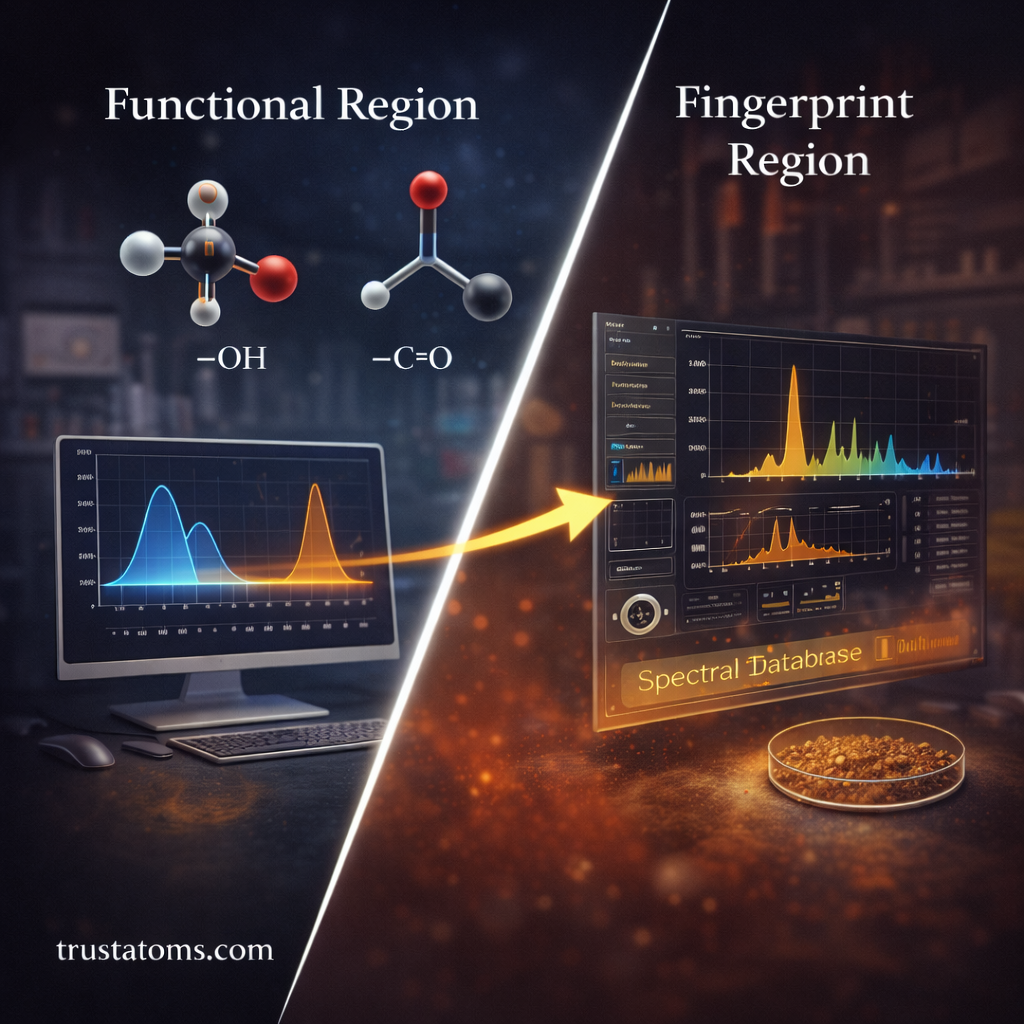
Another key part of the infrared spectrum is known as the fingerprint region.
This region contains complex absorption patterns created by interactions between multiple vibrations within the molecule.
Although these patterns may be difficult to interpret individually, they are extremely useful for identifying specific substances.
Scientists often compare the fingerprint region of an unknown sample with known reference spectra stored in spectral databases.
If the patterns match, the compound can often be identified with high confidence.
How Infrared Spectroscopy Instruments Work
Modern infrared spectroscopy instruments use precise optical systems to measure radiation absorption.
A typical infrared spectrometer includes several key components:
- Infrared radiation source – generates infrared light
- Sample holder – contains the chemical sample
- Monochromator or interferometer – separates wavelengths of light
- Detector – measures absorbed radiation
- Computer system – analyzes and displays the spectrum
One of the most common modern instruments is the Fourier Transform Infrared (FTIR) spectrometer, which collects spectral data rapidly and with high sensitivity.
Steps in Identifying Chemicals with Infrared Spectroscopy
Infrared spectroscopy is often used to identify unknown substances through a systematic process.
Step 1: Prepare the Sample
Samples may be prepared in different forms, including:
- Solid powders
- Liquid films
- Gas samples
- Thin films on specialized plates
Step 2: Collect the Infrared Spectrum
The sample is exposed to infrared radiation, and the instrument measures which wavelengths are absorbed.
Step 3: Analyze the Absorption Peaks
Scientists examine the spectrum to identify characteristic absorption peaks associated with specific chemical bonds.
Step 4: Compare With Reference Spectra
The spectrum is compared to spectral databases to determine the most likely chemical match.
Applications of Infrared Spectroscopy
Infrared spectroscopy is used across many industries and scientific fields.
Pharmaceutical Research
Pharmaceutical laboratories use IR spectroscopy to verify the identity and purity of drug compounds.
Applications include:
- Confirming molecular structures
- Detecting impurities
- Monitoring chemical reactions during drug synthesis
Environmental Monitoring
IR spectroscopy helps scientists detect pollutants in air, water, and soil.
Examples include:
- Identifying industrial emissions
- Monitoring atmospheric gases
- Detecting chemical contaminants in water samples
Forensic Investigations
Forensic scientists frequently use infrared spectroscopy to analyze unknown substances.
Common uses include:
- Identifying drugs and controlled substances
- Analyzing fibers and paints
- Detecting chemical residues at crime scenes
Food and Agriculture
Infrared spectroscopy can also help analyze food composition.
Scientists use it to measure:
- Fat content
- Protein levels
- Moisture levels
- Food authenticity
Advantages of Infrared Spectroscopy
Infrared spectroscopy offers several benefits that make it a valuable tool for chemical identification.
These advantages include:
- Rapid analysis of samples
- Non-destructive testing
- Minimal sample preparation
- Ability to analyze solids, liquids, and gases
- High reliability for identifying functional groups
Because of these strengths, infrared spectroscopy is widely used in both academic research and industrial laboratories.
Limitations of Infrared Spectroscopy
Despite its usefulness, infrared spectroscopy does have some limitations.
Challenges may include:
- Overlapping absorption peaks
- Difficulty analyzing very complex mixtures
- Limited sensitivity for extremely dilute samples
- Requirement for careful sample preparation
In many cases, scientists combine infrared spectroscopy with other analytical techniques to obtain more complete molecular information.
The Future of Infrared Spectroscopy
Technological improvements continue to expand the capabilities of infrared spectroscopy.
Modern advances include:
- Portable IR spectrometers for field use
- Automated spectral identification software
- Miniaturized sensors for environmental monitoring
- Integration with artificial intelligence for faster analysis
These innovations are making infrared spectroscopy more accessible and powerful than ever before.
Final Thoughts
Infrared spectroscopy is a fundamental method for identifying chemical substances and studying molecular structures. By analyzing how molecules absorb infrared radiation, scientists can determine the presence of functional groups, compare spectral fingerprints, and identify unknown compounds with remarkable accuracy.
From pharmaceuticals and environmental monitoring to forensic investigations and food science, infrared spectroscopy remains one of the most important analytical techniques in modern chemistry.