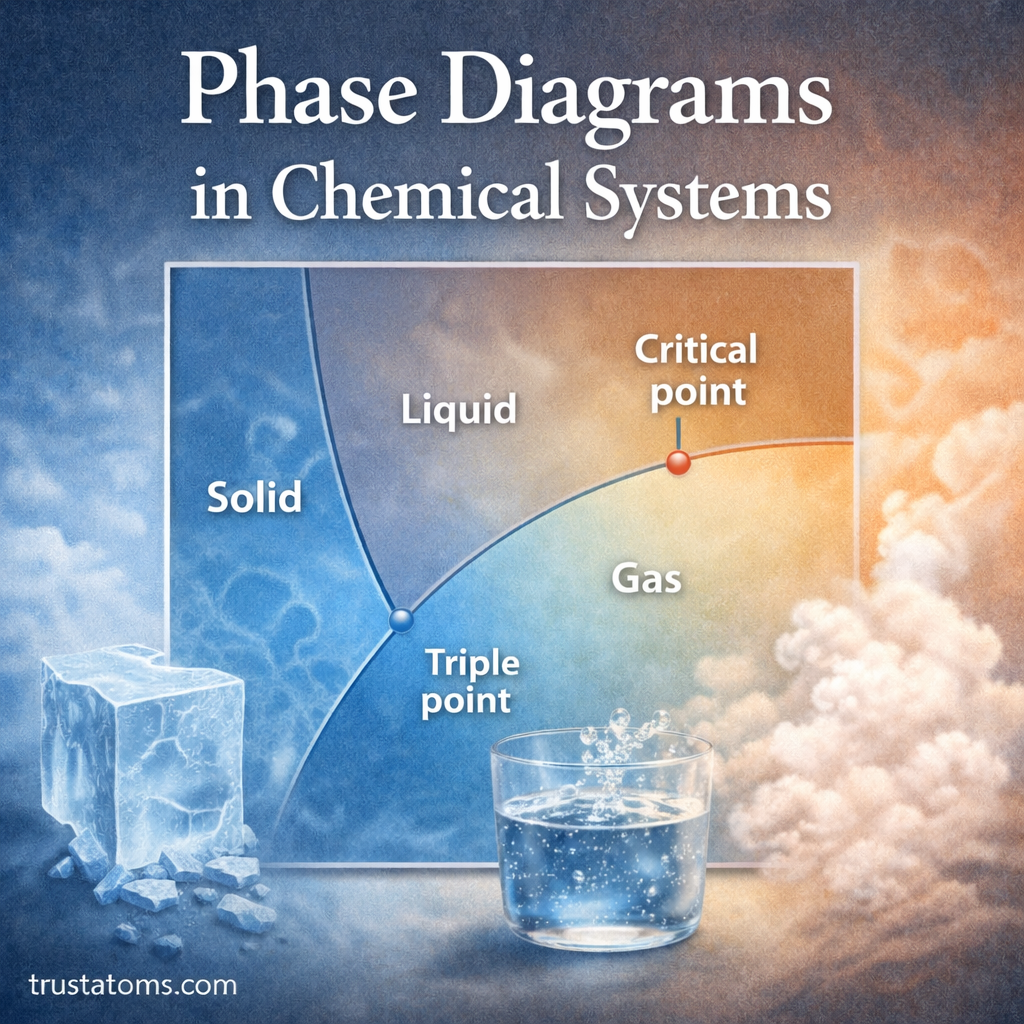
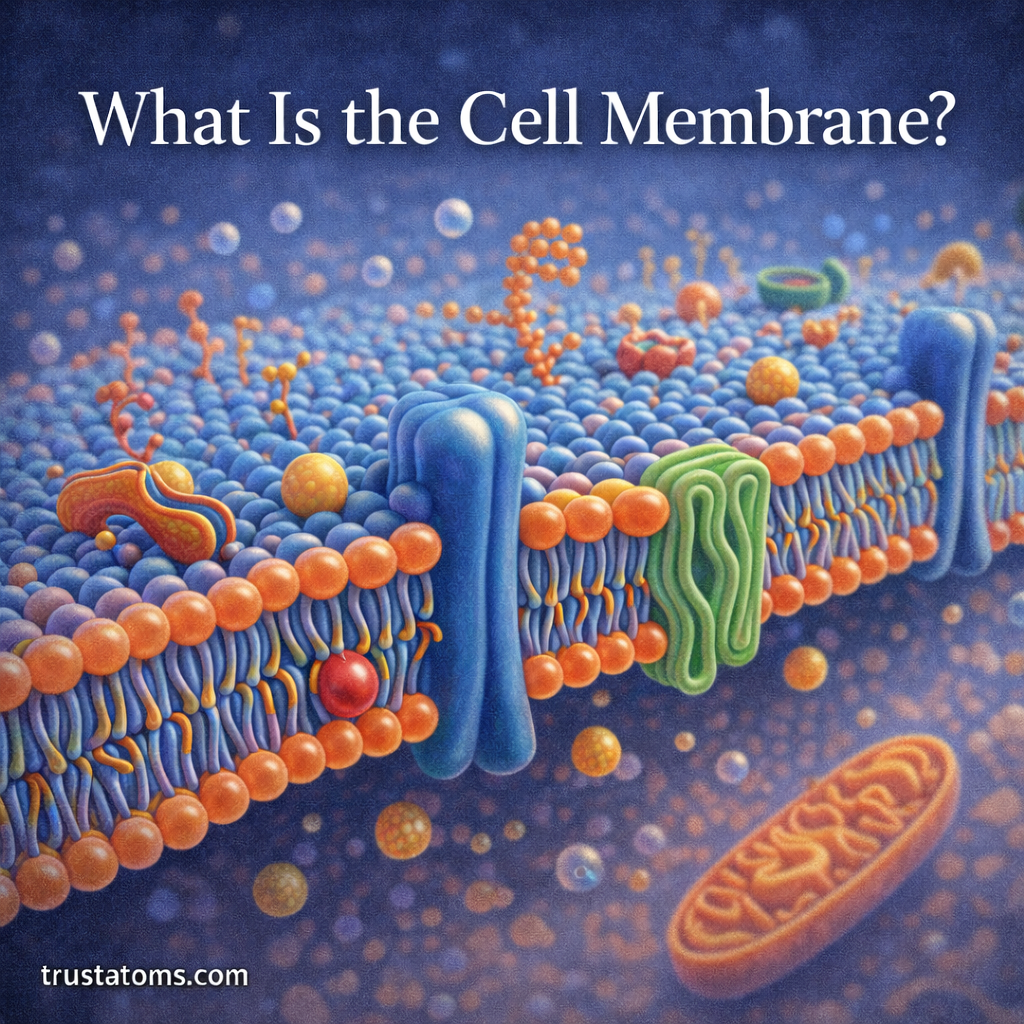
Chemical reactions occur when existing chemical bonds break and new ones form. The energy required to break these bonds plays a critical role in determining how reactions proceed. One of the most important concepts chemists use to understand this process is bond dissociation energy (BDE).
Bond dissociation energy measures the strength of a chemical bond and indicates how much energy is needed to separate two bonded atoms. This concept helps scientists predict reaction pathways, estimate reaction energies, and understand why some molecules are more stable than others.
From combustion reactions to biological metabolism, bond dissociation energy is a key factor that explains how chemical reactions occur and how much energy they release or absorb.
What Is Bond Dissociation Energy?
Bond dissociation energy is the amount of energy required to break a chemical bond between two atoms in a molecule.
When a bond breaks, the atoms separate and the shared electrons are divided between them. The energy needed for this process reflects how strongly the atoms were bonded together.
In general:
- Higher bond dissociation energy means a stronger bond.
- Lower bond dissociation energy means a weaker bond.
Strong bonds require more energy to break, while weaker bonds break more easily during chemical reactions.
Homolytic Bond Cleavage
Bond dissociation energy usually refers to a specific type of bond breaking called homolytic cleavage.
In homolytic cleavage:
- Each atom keeps one of the shared electrons.
- Two neutral fragments called free radicals are produced.
Free radicals are highly reactive species that play important roles in many chemical processes.
Example
If a bond between two atoms breaks evenly:
- The shared electron pair splits.
- Each atom receives one electron.
- Two reactive radical species are formed.
This process is common in high-energy environments such as combustion reactions and atmospheric chemistry.
Factors That Influence Bond Dissociation Energy
Not all chemical bonds require the same amount of energy to break. Several factors influence bond strength.
Bond Type
Single, double, and triple bonds have different strengths.
Typical trends include:
- Single bonds are generally weaker
- Double bonds are stronger
- Triple bonds are the strongest
More shared electrons between atoms create stronger attractions.
Atomic Size
Atoms that are larger tend to form weaker bonds.
This occurs because:
- Larger atoms have electrons farther from the nucleus
- The bond distance increases
- Attraction between atoms weakens
Bond Length
Bond length and bond strength are closely related.
Shorter bonds usually have:
- Stronger attractions
- Higher bond dissociation energies
Longer bonds generally require less energy to break.
Electronegativity
The difference in electronegativity between atoms can influence bond strength.
Atoms that attract electrons strongly may create bonds with greater stability, depending on the molecular structure.
Bond Dissociation Energy and Reaction Energetics
Bond dissociation energy plays a central role in determining whether a chemical reaction releases or absorbs energy.
Chemical reactions involve two main steps:
- Breaking bonds in reactant molecules
- Forming new bonds in product molecules
Breaking bonds requires energy, while forming bonds releases energy.
Energy Balance in Reactions
The overall energy change in a reaction depends on the difference between these two processes.
- If more energy is released than absorbed, the reaction releases heat.
- If more energy is required than released, the reaction absorbs heat.
This energy balance explains why some reactions occur easily while others require external energy.
Estimating Reaction Energy Using Bond Dissociation Energies
Chemists often estimate reaction energy changes using known bond dissociation energies.
The general approach involves:
- Identifying all bonds broken in the reactants
- Adding the energy required to break those bonds
- Identifying all bonds formed in the products
- Subtracting the energy released from forming those bonds
This method provides an approximate way to predict whether reactions are energy-releasing or energy-absorbing.
Although simplified, it is a useful tool for understanding reaction thermodynamics.
Bond Dissociation Energy in Organic Chemistry
Bond dissociation energy is especially important in organic chemistry, where reactions often involve complex molecules.
In many organic reactions:
- Weak bonds break first
- Radicals or reactive intermediates form
- New molecular structures are produced
Understanding bond strengths allows chemists to predict which bonds will break during reactions.
Examples in Organic Chemistry
Common reaction types influenced by bond dissociation energy include:
- Combustion reactions
- Polymerization reactions
- Free radical reactions
- Photochemical reactions
In these reactions, energy from heat or light often initiates bond breaking.
Applications in Energy and Combustion

Bond dissociation energy helps explain why fuels release energy when burned.
In combustion reactions:
- Bonds in fuel molecules break
- Oxygen bonds rearrange
- New bonds form in products like carbon dioxide and water
Because the new bonds formed are often stronger than the original bonds, the reaction releases large amounts of energy.
This energy release powers engines, power plants, and many industrial systems.
Importance in Atmospheric and Environmental Chemistry
Bond dissociation energy also plays an important role in atmospheric chemistry.
Sunlight can supply enough energy to break certain molecular bonds in the atmosphere.
This leads to:
- Formation of reactive radicals
- Ozone formation and breakdown
- Chemical cycles affecting air quality
Understanding bond energies helps scientists study environmental processes and predict chemical changes in the atmosphere.
Why Bond Dissociation Energy Matters
Bond dissociation energy provides a fundamental measure of bond strength in molecules. By understanding how much energy is required to break different bonds, chemists gain insight into reaction pathways and molecular stability.
This concept helps scientists:
- Predict which reactions are likely to occur
- Estimate energy changes during reactions
- Understand reaction mechanisms
- Design efficient chemical processes
Bond dissociation energy connects molecular structure with chemical behavior, making it a cornerstone of chemical thermodynamics and reaction chemistry.
Final Thoughts
Bond dissociation energy is a powerful concept that helps explain how chemical reactions work. Every reaction depends on the balance between breaking old bonds and forming new ones, and bond energies provide a way to measure these changes.
By studying bond dissociation energy, chemists can understand why certain reactions release energy, why some molecules are more stable than others, and how complex chemical transformations occur. This knowledge is essential in fields ranging from organic chemistry to environmental science and energy research.