Chemical reactions rarely occur in a single, simple step. Instead, most reactions proceed through a sequence of smaller transformations that gradually convert reactants into products. These sequences are known as reaction pathways.
Understanding reaction pathways helps chemists explain how reactions occur, how fast they proceed, and what intermediate structures form during the process. Reaction pathway analysis is essential in fields such as organic chemistry, biochemistry, materials science, and industrial chemical engineering.
By studying reaction pathways, scientists can predict reaction behavior, improve reaction efficiency, and design safer chemical processes.
What Are Reaction Pathways?
A reaction pathway describes the step-by-step sequence of molecular events that occur during a chemical reaction.
Rather than transforming directly from reactants to products, molecules often pass through several intermediate states.
A reaction pathway typically includes:
- Reactants (starting molecules)
- Transition states
- Reaction intermediates
- Final products
Each step in the pathway involves changes in molecular structure, bonding, and energy.
Understanding these stages allows chemists to map how molecules rearrange during chemical reactions.
Energy Changes Along Reaction Pathways
Chemical reactions involve changes in energy as molecules break and form chemical bonds.
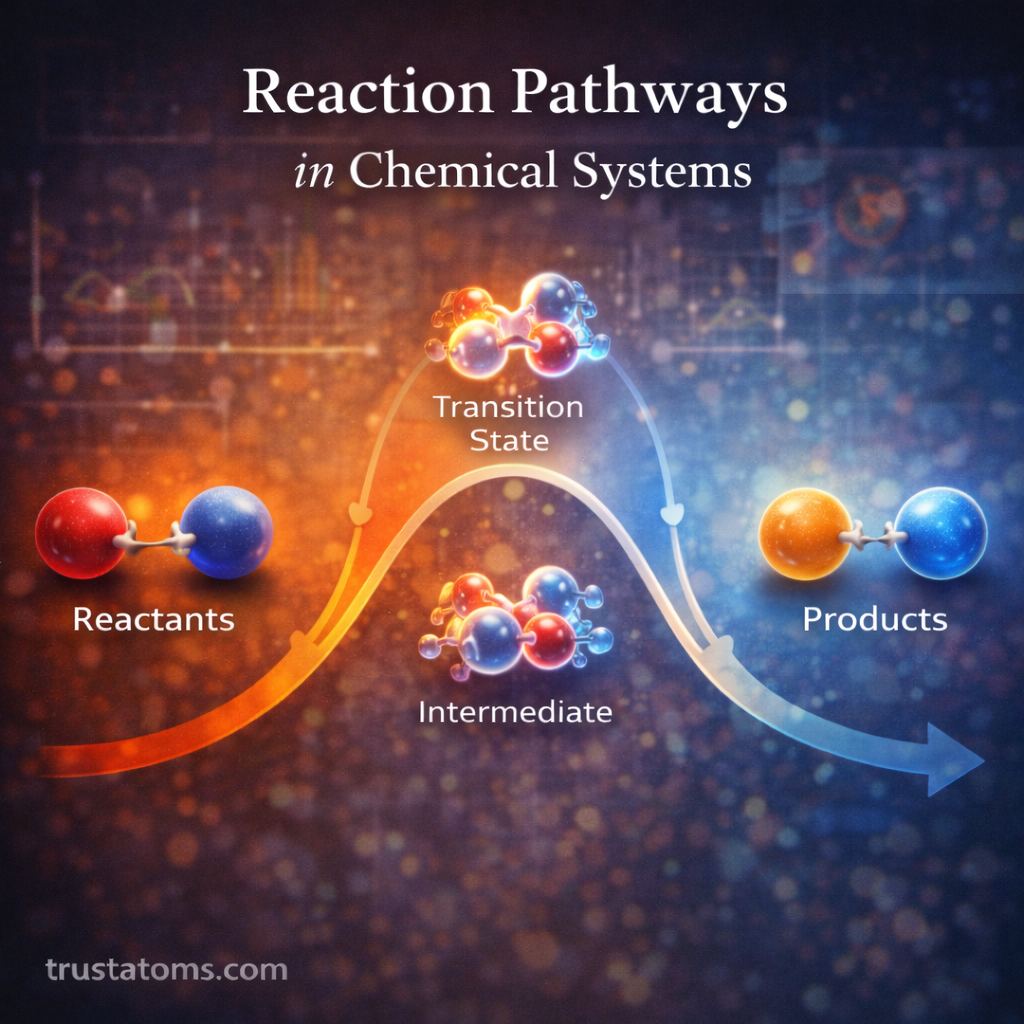
A common way to visualize reaction pathways is through a reaction energy diagram, which shows how energy changes during the reaction.
Important features of reaction energy diagrams include:
Reactants
The starting point of the reaction with a defined energy level.
Transition States
High-energy configurations that occur during bond rearrangement.
Transition states represent the highest energy point of each reaction step.
Intermediates
Temporary molecular structures formed between steps of a reaction.
These species are often unstable but can sometimes be detected experimentally.
Products
The final molecules formed once the reaction is complete.
The difference in energy between reactants and products determines whether a reaction releases or absorbs energy.
Activation Energy and Reaction Rates

For a reaction to proceed, molecules must overcome an energy barrier known as activation energy.
Activation energy represents the minimum energy required to reach the transition state.
Factors that influence activation energy include:
- Bond strength
- Molecular orientation
- Temperature
- Presence of catalysts
Lower activation energy generally leads to faster reaction rates, because more molecules can reach the transition state under normal conditions.
This concept explains why some reactions occur rapidly while others proceed very slowly.
Multi-Step Reaction Mechanisms
Many reactions occur through multiple elementary steps rather than a single direct transformation.
These sequences are known as reaction mechanisms.
Each step typically involves:
- Bond breaking
- Bond formation
- Electron movement
- Formation of intermediates
For example, a reaction might proceed through:
- Formation of an unstable intermediate
- Rearrangement of the intermediate
- Formation of the final product
The slowest step in a reaction mechanism is called the rate-determining step because it controls the overall reaction speed.
Reaction Intermediates
Reaction intermediates are temporary species that form during multi-step reactions.
Although short-lived, intermediates play an important role in determining reaction pathways.
Common types of intermediates include:
- Carbocations
- Carbanions
- Free radicals
- Organometallic intermediates
Some intermediates exist only briefly, while others may persist long enough to be observed with advanced analytical techniques.
Understanding intermediates helps chemists determine how reactions proceed and how to control them.
Catalysts and Reaction Pathways
Catalysts are substances that speed up chemical reactions without being permanently consumed.
They work by changing the reaction pathway, typically by providing an alternative route with lower activation energy.
Benefits of catalysts include:
- Faster reaction rates
- Reduced energy requirements
- Improved reaction efficiency
- Greater selectivity for desired products
Catalysts are essential in many industrial and biological processes.
Examples include:
- Enzymes in biological systems
- Metal catalysts in industrial chemistry
- Acid-base catalysts in organic reactions
By modifying reaction pathways, catalysts make many reactions practical that would otherwise occur too slowly.
Reaction Pathways in Biological Systems
In biological chemistry, reaction pathways are often part of complex metabolic networks.
These pathways involve sequences of enzyme-catalyzed reactions that transform molecules inside living cells.
Examples include:
- Cellular respiration
- Photosynthesis
- Amino acid synthesis
- DNA replication
Biological pathways are highly regulated to maintain efficient energy use and stable cellular conditions.
Enzymes play a crucial role by lowering activation energies and guiding reactions along specific pathways.
Computational Modeling of Reaction Pathways
Modern chemistry often uses computational methods to analyze reaction pathways.
These techniques allow scientists to simulate molecular interactions and predict reaction behavior.
Computational chemistry can help:
- Identify transition states
- Predict reaction intermediates
- Estimate activation energies
- Model complex reaction networks
These tools are especially valuable for studying reactions that are difficult to observe experimentally.
Why Reaction Pathways Matter
Understanding reaction pathways provides insights into how chemical systems behave and how reactions can be controlled.
Key benefits include:
- Predicting reaction outcomes
- Designing more efficient chemical processes
- Improving catalysts
- Understanding biological metabolism
- Developing new pharmaceuticals and materials
By mapping the sequence of steps that occur during a reaction, chemists gain a deeper understanding of molecular transformations.
Final Thoughts
Reaction pathways reveal the detailed sequence of events that occur during chemical reactions. Instead of simple transformations, most reactions involve multiple steps, intermediates, and energy barriers that shape how molecules rearrange.
By analyzing reaction pathways, chemists can predict reaction rates, design better catalysts, and optimize chemical processes in both laboratory and industrial settings. This deeper understanding of molecular transformations continues to play a central role in modern chemistry, from pharmaceutical development to energy research.