Organocatalysis has become an important tool in modern chemical synthesis. Unlike traditional catalysis that relies on metals or enzymes, organocatalysis uses small organic molecules to accelerate chemical reactions. These catalysts often contain elements such as carbon, hydrogen, nitrogen, oxygen, sulfur, or phosphorus.
Because organocatalysts are typically stable, inexpensive, and environmentally friendly, they have gained significant attention in organic chemistry. Today, organocatalysis is widely used in pharmaceutical synthesis, materials science, and green chemistry applications.
What Is Organocatalysis?
Organocatalysis refers to chemical reactions that are accelerated by organic molecules acting as catalysts. These molecules participate in the reaction process without being consumed, allowing them to facilitate multiple reaction cycles.
Organocatalysts typically work by:
- Activating reactants
- Stabilizing reaction intermediates
- Lowering activation energy
- Controlling reaction selectivity
Because they are made from organic molecules rather than metals, organocatalysts avoid many problems associated with metal contamination in chemical synthesis.
Why Organocatalysis Is Important in Modern Chemistry
Modern chemical synthesis often requires reactions that are efficient, selective, and environmentally responsible. Organocatalysis offers several advantages compared to traditional catalytic methods.
Key benefits include:
- Metal-free reactions, reducing contamination risks
- Lower toxicity compared to many metal catalysts
- Operational simplicity, often requiring mild reaction conditions
- High selectivity, particularly for asymmetric synthesis
- Compatibility with green chemistry principles
These advantages make organocatalysis especially valuable in pharmaceutical and fine chemical production.
Types of Organocatalysts
Organocatalysts can be grouped into several categories depending on how they interact with reactants.
Aminocatalysts
Aminocatalysts contain nitrogen atoms and often function through the formation of reactive intermediates.
Common aminocatalysts include:
- Proline derivatives
- Secondary amines
- Imidazolidinones
These catalysts are widely used in reactions such as aldol reactions and Michael additions.
Hydrogen-Bonding Catalysts
Some organocatalysts operate by forming hydrogen bonds with reactants. These interactions help activate molecules and stabilize transition states.
Examples include:
- Thioureas
- Ureas
- Squaramides
Hydrogen-bonding catalysts are commonly used in asymmetric synthesis to control molecular orientation during reactions.
Brønsted Acid and Base Catalysts
Certain organocatalysts act as proton donors or acceptors.
These catalysts facilitate reactions by:
- Activating electrophiles through protonation
- Generating nucleophiles through deprotonation
- Stabilizing reaction intermediates
This category includes chiral phosphoric acids and organic bases.
Asymmetric Organocatalysis
One of the most important contributions of organocatalysis is its role in asymmetric synthesis, where reactions produce molecules with specific three-dimensional arrangements.
Many biologically active molecules exist in only one chiral form. Producing the correct stereochemistry is therefore critical in pharmaceutical development.
Asymmetric organocatalysis enables:
- Control of molecular chirality
- High enantioselectivity
- Efficient production of complex molecules
This area of chemistry gained global recognition when the 2021 Nobel Prize in Chemistry was awarded for the development of asymmetric organocatalysis.
Common Reactions in Organocatalysis
Organocatalysts are used to accelerate many important organic reactions.
Some widely studied reactions include:
Aldol Reactions
Aldol reactions form carbon–carbon bonds between aldehydes or ketones. Organocatalysts such as proline derivatives are commonly used to control stereochemistry in these reactions.
Michael Additions
Michael addition reactions involve the addition of nucleophiles to activated double bonds. Organocatalysts help control both reaction speed and selectivity.
Diels–Alder Reactions
Certain organocatalysts enhance cycloaddition reactions by activating reactants through hydrogen bonding or iminium ion formation.
Mannich Reactions
These reactions create β-amino carbonyl compounds, which are useful building blocks in pharmaceutical synthesis.
Mechanisms of Organocatalysis
Organocatalysts can accelerate reactions through several different mechanisms.
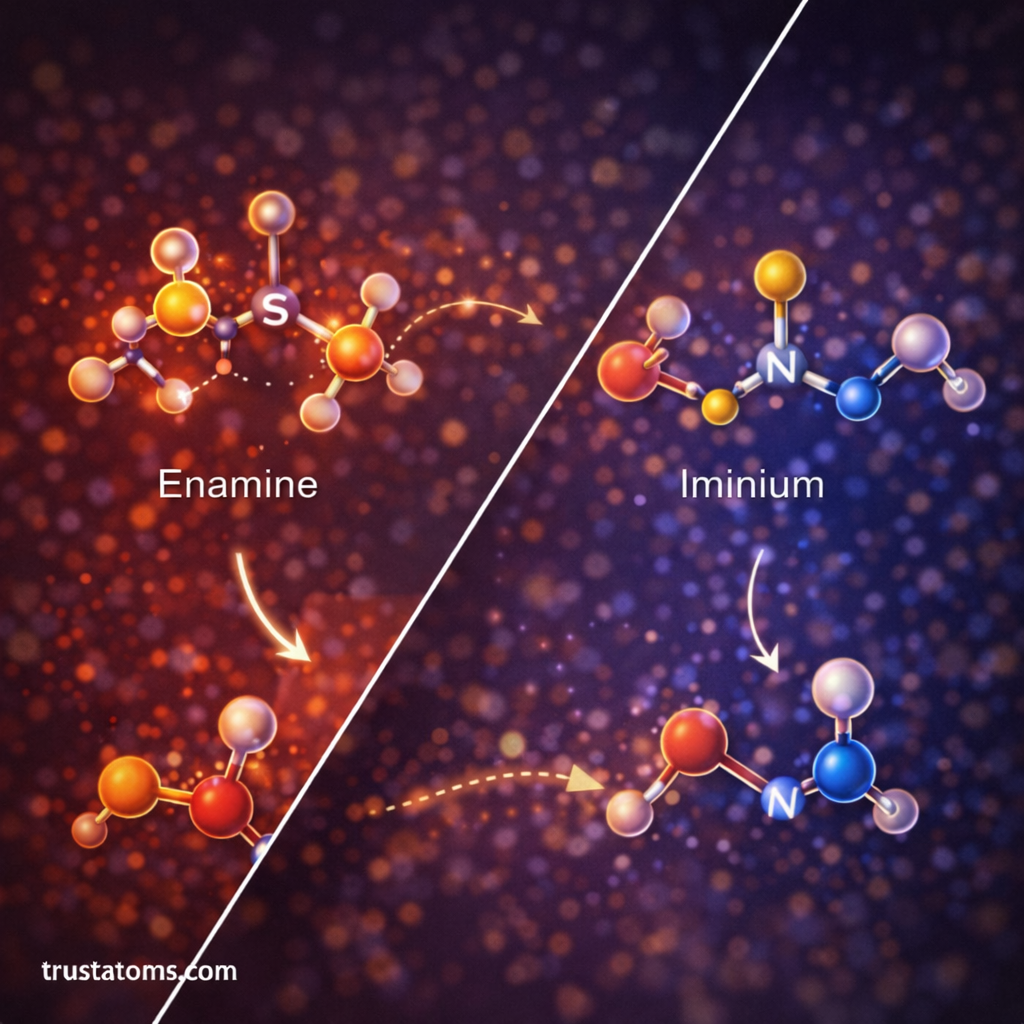
Enamine Catalysis
In this mechanism:
- The organocatalyst reacts with a carbonyl compound.
- An enamine intermediate forms.
- The enamine reacts with an electrophile.
- The catalyst is regenerated.
This pathway is common in proline-catalyzed aldol reactions.
Iminium Catalysis
In iminium catalysis:
- The catalyst forms an iminium ion with a carbonyl compound.
- The iminium ion activates the molecule toward nucleophilic attack.
- The catalyst is released at the end of the reaction.
This mechanism is frequently used in asymmetric reactions.
Hydrogen-Bond Activation
Some organocatalysts stabilize transition states through hydrogen bonding. This interaction helps align molecules in the proper orientation for reaction.
Applications of Organocatalysis
Organocatalysis has numerous applications across modern chemistry and industry.
Pharmaceutical Synthesis
Many pharmaceutical compounds require precise stereochemistry. Organocatalysis enables efficient production of chiral drug molecules.
Green Chemistry
Because organocatalysts often avoid heavy metals and toxic reagents, they support environmentally friendly chemical processes.
Natural Product Synthesis
Complex natural molecules often require highly selective synthetic pathways. Organocatalysts provide precise control during multi-step syntheses.
Polymer Chemistry
Organocatalysis is increasingly used in polymerization reactions and advanced materials development.
Advantages Compared to Metal Catalysis
Traditional metal catalysts remain widely used, but organocatalysis offers several distinct advantages.
Comparison highlights include:
| Feature | Organocatalysis | Metal Catalysis |
|---|---|---|
| Catalyst type | Organic molecules | Metal complexes |
| Toxicity | Generally lower | Often higher |
| Environmental impact | Lower | Higher |
| Metal contamination risk | None | Possible |
| Reaction conditions | Often mild | Sometimes harsher |
These differences make organocatalysis particularly appealing for sensitive chemical processes.
Limitations of Organocatalysis
Despite its benefits, organocatalysis also faces certain limitations.
Challenges include:
- Slower reaction rates in some systems
- Higher catalyst loading compared to metal catalysts
- Limited substrate scope for certain reactions
- Difficulty scaling some processes for industrial production
Researchers continue working to overcome these challenges by designing more efficient organocatalysts.
The Future of Organocatalysis
Organocatalysis continues to expand as chemists develop new catalyst designs and reaction pathways.
Emerging research areas include:
- Dual catalysis combining organocatalysts with metal catalysts
- Photoredox organocatalysis using light-driven reactions
- Machine-learning-guided catalyst design
- Sustainable chemical manufacturing
These developments are helping shape the next generation of synthetic chemistry.
Conclusion
Organocatalysis has transformed modern synthetic chemistry by providing efficient, metal-free catalytic systems based on small organic molecules. Through mechanisms such as enamine catalysis, iminium activation, and hydrogen bonding, organocatalysts enable a wide range of reactions with high selectivity and environmental benefits.
From pharmaceutical synthesis to green chemistry innovations, organocatalysis continues to play a crucial role in developing sustainable and efficient chemical processes. As research advances, this field will remain a cornerstone of modern chemical synthesis.