Electrochemical systems play a critical role in modern technology, powering devices such as batteries, fuel cells, corrosion protection systems, and electrolysis equipment. At the center of these processes are electrodes, where chemical reactions involving electron transfer occur.
Electrode kinetics focuses on the rate at which electrochemical reactions occur at the electrode surface. By studying these reaction rates, scientists and engineers can improve energy storage devices, optimize industrial electrochemical processes, and better understand corrosion and material degradation.
Understanding electrode kinetics is essential for designing efficient electrochemical systems and predicting how they behave under different conditions.
What Are Electrochemical Reactions?
Electrochemical reactions involve the transfer of electrons between chemical species and an electrode. These reactions occur at the interface between the electrode surface and the surrounding electrolyte.
Two primary types of electrochemical reactions occur in these systems:
- Oxidation – loss of electrons
- Reduction – gain of electrons
Together, these paired reactions form a redox reaction.
In an electrochemical cell:
- One electrode promotes oxidation
- The other electrode promotes reduction
The flow of electrons through an external circuit generates electrical energy or drives chemical transformations.
What Is Electrode Kinetics?
Electrode kinetics describes how quickly electrochemical reactions occur at an electrode surface. The rate of these reactions determines how efficiently a system can generate or consume electrical energy.
Key factors influencing electrode kinetics include:
- Electron transfer rate
- Concentration of reactants
- Temperature
- Surface properties of the electrode
- Applied electrical potential
Even when a reaction is thermodynamically favorable, slow electrode kinetics can significantly reduce the efficiency of an electrochemical system.
The Electrode–Electrolyte Interface
Electrochemical reactions occur at the interface between the electrode and the electrolyte.
This region is highly complex and plays a major role in determining reaction behavior.
At the interface:
- Reactant molecules approach the electrode surface
- Electrons transfer between the electrode and the reactant
- Chemical species are converted into products
- Products diffuse away from the surface
The microscopic structure of this interface influences how easily electrons move between the electrode and molecules in the electrolyte.
Overpotential and Reaction Rate
One of the most important concepts in electrode kinetics is overpotential.
Overpotential refers to the additional electrical potential required to drive an electrochemical reaction at a practical rate.
In theory, reactions should occur at a specific equilibrium potential. However, in real systems, extra energy is often needed to overcome kinetic limitations.
There are several types of overpotential:
Activation Overpotential
This arises from the energy barrier associated with electron transfer during the reaction.
Concentration Overpotential
Occurs when the supply of reactants near the electrode surface becomes limited due to slow diffusion.
Resistance Overpotential
Caused by electrical resistance in the electrolyte, electrode material, or external circuit.
Reducing these overpotentials is essential for improving electrochemical efficiency.
The Butler–Volmer Equation
One of the most widely used models in electrode kinetics is the Butler–Volmer equation. This equation describes the relationship between current and electrode potential.
In practical terms, it explains how reaction rates change as the applied voltage increases or decreases.
The Butler–Volmer model helps scientists analyze:
- Reaction speed at different voltages
- Symmetry between oxidation and reduction reactions
- Exchange current density
By studying these relationships, researchers can better understand the performance limits of electrochemical devices.
Exchange Current Density
Exchange current density is a fundamental parameter in electrode kinetics. It represents the rate of electron transfer when the system is at equilibrium.
Even when no net current flows, microscopic oxidation and reduction reactions are continuously occurring.
A high exchange current density indicates:
- Faster electron transfer
- More efficient electrode reactions
A low exchange current density suggests slower kinetics and higher energy losses.
Different electrode materials exhibit different exchange current densities depending on their chemical properties and surface structure.
Factors That Affect Electrode Kinetics
Several variables influence the rate of electrochemical reactions.
Electrode Material
Different materials facilitate electron transfer at different speeds. Metals such as platinum often exhibit excellent catalytic properties.
Surface Area
Larger electrode surface areas allow more reaction sites, increasing the overall reaction rate.
Temperature
Higher temperatures generally increase reaction rates by providing molecules with more kinetic energy.
Electrolyte Composition
The type and concentration of ions in the electrolyte influence reaction efficiency and diffusion rates.
Surface Structure
Microscopic roughness or nanostructuring of electrode surfaces can significantly improve reaction performance.
Mass Transport in Electrochemical Systems
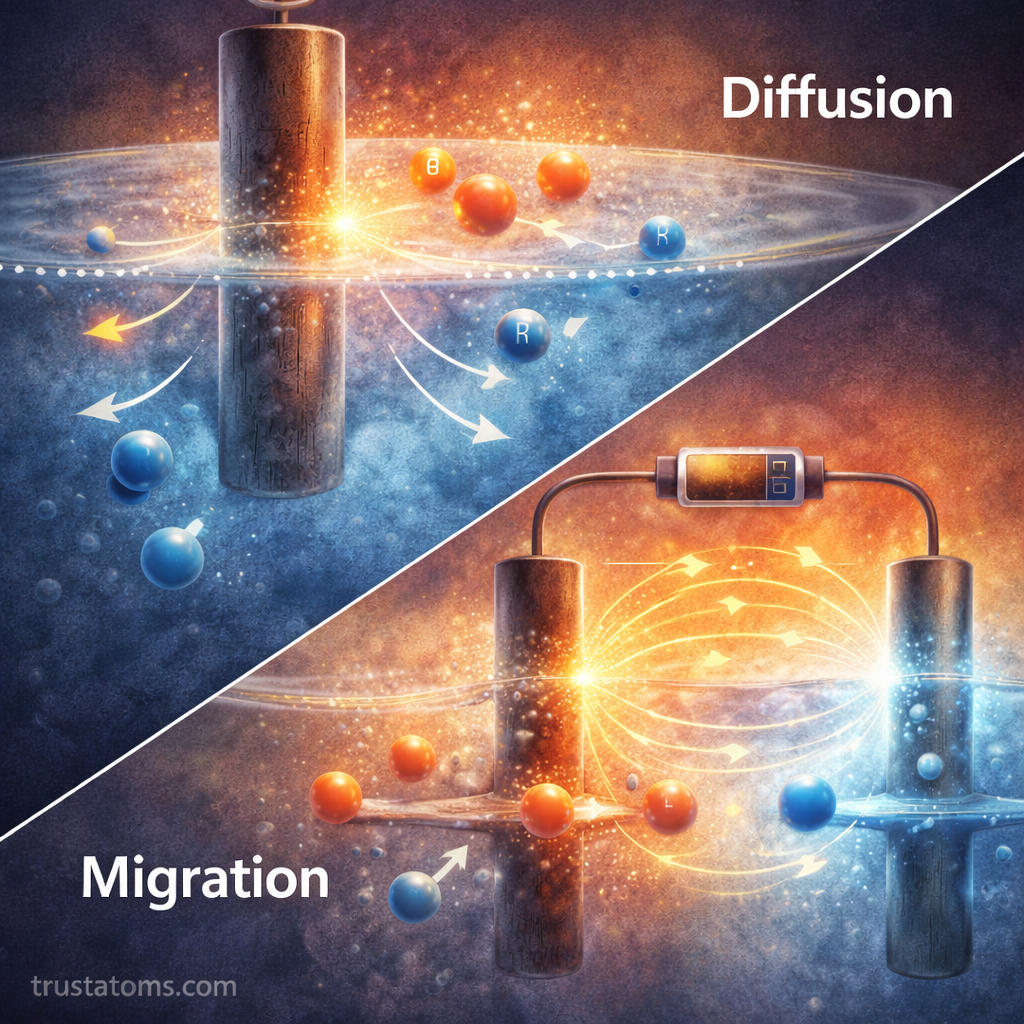
In addition to electron transfer, electrochemical reactions depend on mass transport.
Mass transport refers to the movement of chemical species toward or away from the electrode surface.
Three main mechanisms are involved:
- Diffusion – movement due to concentration gradients
- Migration – movement due to electric fields
- Convection – movement caused by fluid motion
If reactants cannot reach the electrode quickly enough, reaction rates become limited regardless of how fast electron transfer occurs.
Applications of Electrode Kinetics
Electrode kinetics plays a crucial role in many modern technologies.
Batteries
Lithium-ion batteries rely on controlled electrode reactions to store and release energy efficiently.
Fuel Cells
Fuel cells convert chemical energy directly into electricity through electrochemical reactions at electrodes.
Electrolysis
Electrolysis systems use electricity to drive chemical reactions, such as hydrogen production from water.
Corrosion Science
Electrode kinetics helps scientists understand and prevent corrosion in metals exposed to electrolytes.
Sensors and Analytical Devices
Electrochemical sensors detect substances by measuring small electrical signals generated during electrode reactions.
Improving Electrochemical System Performance
Researchers continually explore ways to improve electrode kinetics and overall electrochemical efficiency.
Common strategies include:
- Developing advanced catalytic materials
- Increasing electrode surface area
- Engineering nanoscale electrode structures
- Optimizing electrolyte composition
- Improving system temperature control
These improvements help reduce energy losses and increase the performance of electrochemical technologies.
Why Electrode Kinetics Matters
Electrode kinetics provides a deeper understanding of how electrochemical reactions occur and how quickly they proceed. By analyzing reaction rates at electrode surfaces, scientists can design more efficient batteries, fuel cells, and industrial electrochemical processes.
As demand grows for renewable energy storage and sustainable technologies, the study of electrode kinetics continues to play an increasingly important role in modern chemistry and engineering.