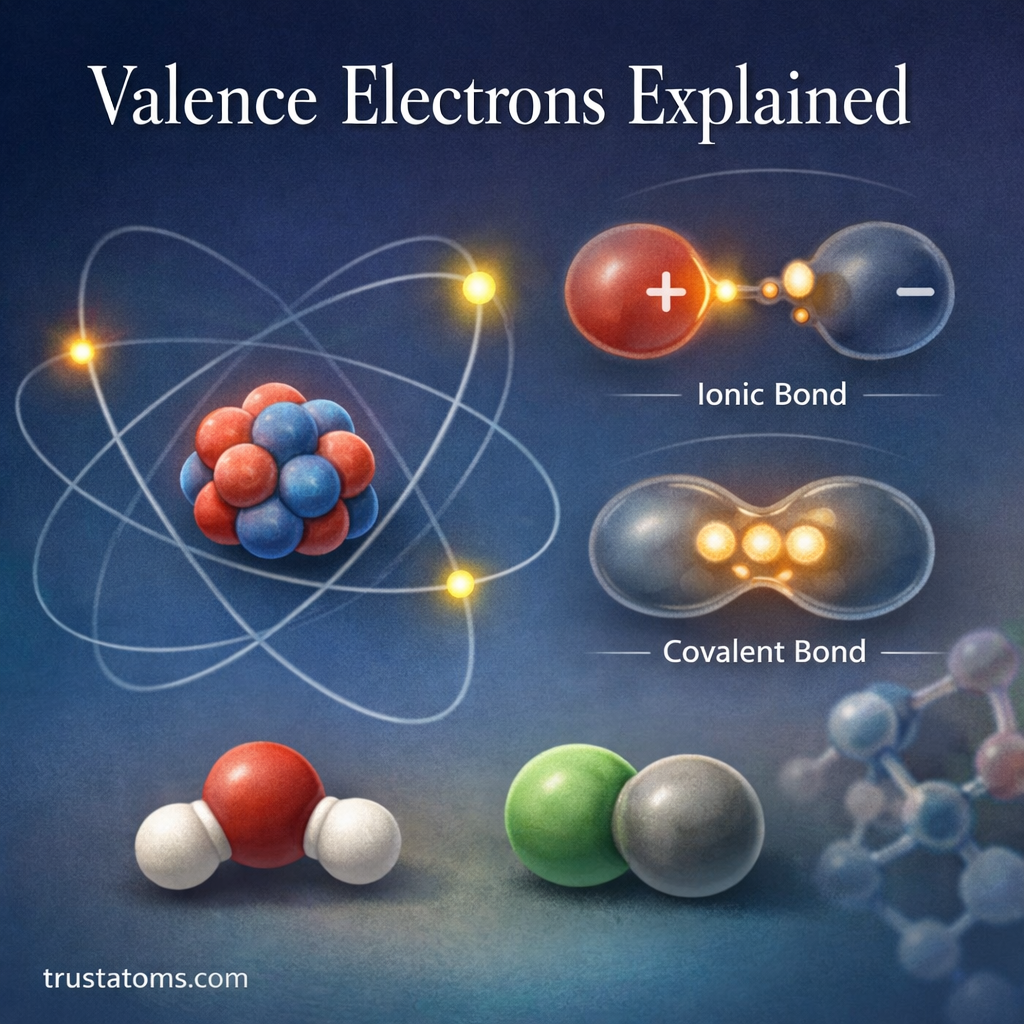
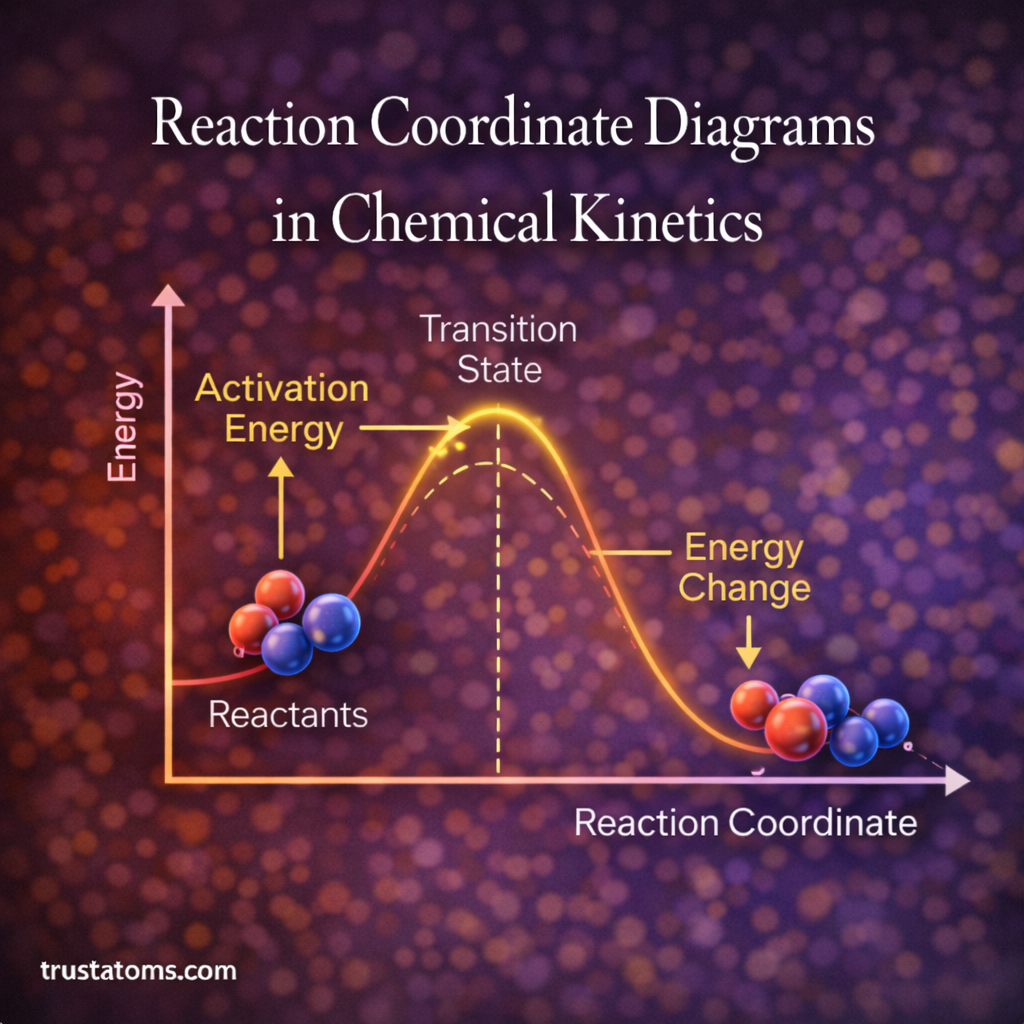
Isotope effects play an important role in understanding how chemical reactions occur. In chemistry, isotopes are atoms of the same element that contain the same number of protons but different numbers of neutrons, giving them different atomic masses.
Although isotopes behave similarly in many chemical reactions, their mass differences can influence reaction rates and mechanisms. By observing how reactions change when atoms are replaced with heavier or lighter isotopes, chemists can gain valuable insights into the steps that occur during a reaction.
These differences are known as isotope effects, and they have become powerful tools in modern chemistry for studying reaction mechanisms, enzyme behavior, and molecular dynamics.
What Are Isotopes?
Atoms of the same element always have the same number of protons, but they can vary in the number of neutrons in their nuclei.
These variations create isotopes.
Examples include:
- Hydrogen, deuterium, and tritium
- Carbon-12 and carbon-13
- Oxygen-16 and oxygen-18
Despite having similar chemical properties, isotopes differ in atomic mass, which affects how atoms vibrate within chemical bonds.
These differences can influence the speed or pathway of certain chemical reactions.
What Is the Isotope Effect?
The isotope effect refers to the change in reaction rate or reaction mechanism that occurs when one atom in a molecule is replaced by one of its isotopes.
Because heavier isotopes move more slowly and form slightly stronger bonds, reactions involving them often proceed at different rates.
In many cases:
- Bonds involving lighter isotopes break more easily
- Reactions involving heavier isotopes occur more slowly
By comparing reaction rates between isotopic variants of a molecule, chemists can identify which bonds are involved in key reaction steps.
Types of Isotope Effects

Isotope effects are generally classified into two major categories depending on how the isotopic substitution influences the reaction.
Primary Isotope Effects
A primary isotope effect occurs when the bond to the isotopic atom is directly broken or formed during the reaction.
This type of isotope effect has the strongest impact on reaction rate.
For example:
- Replacing hydrogen with deuterium in a bond that breaks during the reaction can significantly slow the reaction.
Primary isotope effects help chemists determine:
- Which bond is broken in the rate-determining step
- Whether hydrogen transfer is involved in the mechanism
These observations provide direct clues about how the reaction proceeds.
Secondary Isotope Effects
A secondary isotope effect occurs when the isotopic substitution is not directly involved in bond breaking or formation but still influences the reaction.
These effects are typically smaller than primary isotope effects.
Secondary isotope effects can reveal subtle details such as:
- Changes in molecular geometry
- Hybridization changes during reactions
- Electronic structure shifts
Even though the isotopic atom is not directly involved in the reaction step, its presence can still alter the behavior of nearby bonds.
Kinetic Isotope Effects
The most commonly studied isotope effects in chemistry are kinetic isotope effects (KIEs).
Kinetic isotope effects occur when isotopic substitution changes the rate of a chemical reaction.
Chemists measure these effects by comparing reaction rates of isotopically substituted molecules.
For example:
- A reaction is performed with a molecule containing hydrogen.
- The same reaction is repeated with deuterium replacing hydrogen.
- The reaction rates are compared.
If the reaction slows significantly with the heavier isotope, this indicates that the hydrogen bond is likely involved in the rate-limiting step.
Kinetic isotope effects are widely used to study:
- Organic reaction mechanisms
- Catalytic processes
- Enzyme reactions
Equilibrium Isotope Effects
In addition to influencing reaction rates, isotopes can also affect chemical equilibrium.
This phenomenon is called the equilibrium isotope effect.
Equilibrium isotope effects arise because heavier isotopes often form slightly stronger bonds.
As a result:
- Molecules containing heavier isotopes may be slightly more stable
- Chemical equilibria may shift depending on isotopic composition
These effects are especially important in fields such as:
- Environmental chemistry
- Geochemistry
- Climate science
Scientists often analyze isotope distributions to understand natural processes occurring over long timescales.
Why Isotope Effects Occur
The isotope effect arises primarily from differences in atomic mass, which affect how atoms vibrate within chemical bonds.
Key factors include:
- Heavier isotopes vibrate more slowly
- Bonds involving heavier isotopes are slightly stronger
- More energy is required to break these bonds
Because many chemical reactions involve bond breaking or bond rearrangement, these subtle differences can influence reaction pathways.
This effect is particularly noticeable when hydrogen is replaced by deuterium, since hydrogen isotopes show relatively large mass differences.
Applications of Isotope Effects
Isotope effects are widely used as analytical tools in chemistry and related scientific fields.
Studying Reaction Mechanisms
Chemists often substitute atoms with isotopes to determine how reactions occur.
Isotope experiments can reveal:
- Which bonds break during the reaction
- The order of reaction steps
- Whether hydrogen transfer occurs
These insights help refine reaction models.
Enzyme Mechanism Studies
Biochemists use isotope effects to investigate enzyme-catalyzed reactions.
Enzyme studies may involve:
- Substituting hydrogen with deuterium
- Measuring changes in reaction rates
- Determining rate-limiting steps in metabolic pathways
This information helps researchers understand how enzymes function at the molecular level.
Environmental and Geochemical Analysis
Isotopes are also valuable tracers in environmental science.
Scientists use isotope ratios to study:
- Atmospheric chemical reactions
- Water cycles
- Geological processes
- Climate history
Small isotope effects in natural systems can reveal large-scale environmental patterns.
Pharmaceutical Research
In drug development, isotopic substitution can influence metabolic stability.
For example:
- Replacing hydrogen with deuterium can slow certain metabolic reactions
- This technique can improve drug stability and effectiveness
Some modern pharmaceuticals use deuterated compounds to enhance performance.
Experimental Methods for Measuring Isotope Effects
Scientists measure isotope effects using several analytical techniques.
Common methods include:
- Mass spectrometry
- Nuclear magnetic resonance (NMR) spectroscopy
- Isotopic labeling experiments
- Reaction rate measurements
These methods allow researchers to compare isotopic variants and analyze subtle changes in reaction behavior.
Final Thoughts
Isotope effects provide powerful insights into the mechanisms of chemical reactions. By comparing how reactions proceed with different isotopes of the same element, chemists can uncover which bonds break, how molecules rearrange, and which steps control the overall reaction rate.
From studying enzyme activity to analyzing environmental processes, isotope effects help scientists understand the hidden details of chemical transformations. As analytical techniques continue to improve, isotope studies will remain an essential tool for exploring the complex world of chemical reactions.