Reactive intermediates are short-lived chemical species that appear during the transformation of molecules in organic reactions. Although they usually exist only for fractions of a second, they play a crucial role in determining how reactions proceed and what products are formed.
Understanding reactive intermediates helps chemists explain reaction mechanisms, design new synthetic pathways, and develop pharmaceuticals, materials, and industrial chemicals. These transient species reveal what happens between the starting materials and the final products during complex chemical transformations.
Because intermediates are often highly unstable and reactive, they are difficult to observe directly. However, modern experimental techniques and computational chemistry have made it possible to study them in increasing detail.
What Are Reactive Intermediates?
Reactive intermediates are temporary molecular species formed during the steps of a chemical reaction. They are neither the original reactants nor the final products but exist between these stages.
These species are usually characterized by:
- High reactivity
- Short lifetimes
- Unstable electronic structures
- High energy compared to stable molecules
Reactive intermediates form when bonds break or rearrange during chemical reactions. They quickly transform into more stable structures, which is why they are often difficult to isolate.
Why Reactive Intermediates Matter
Reactive intermediates are essential for understanding reaction mechanisms, which describe the sequence of steps that occur during a chemical reaction.
By identifying intermediates, chemists can:
- Predict reaction outcomes
- Control reaction conditions
- Improve efficiency of chemical synthesis
- Design better catalysts
- Avoid unwanted side reactions
In fields such as pharmaceutical chemistry and materials science, understanding intermediates allows researchers to create more precise and efficient chemical processes.
How Reactive Intermediates Form
Reactive intermediates typically form when chemical bonds break or when electrons shift during a reaction.
A typical reaction pathway might include several stages:
- Bond breaking – Existing bonds within a molecule are broken.
- Formation of an unstable species – A high-energy intermediate forms.
- Rearrangement or reaction – The intermediate reacts further with nearby molecules or atoms.
- Formation of stable products – New bonds form to produce the final product.
Each step in the process may involve a different intermediate species.
Major Types of Reactive Intermediates
Organic chemistry includes several well-known classes of reactive intermediates. Each type has a distinctive electronic structure and behavior.
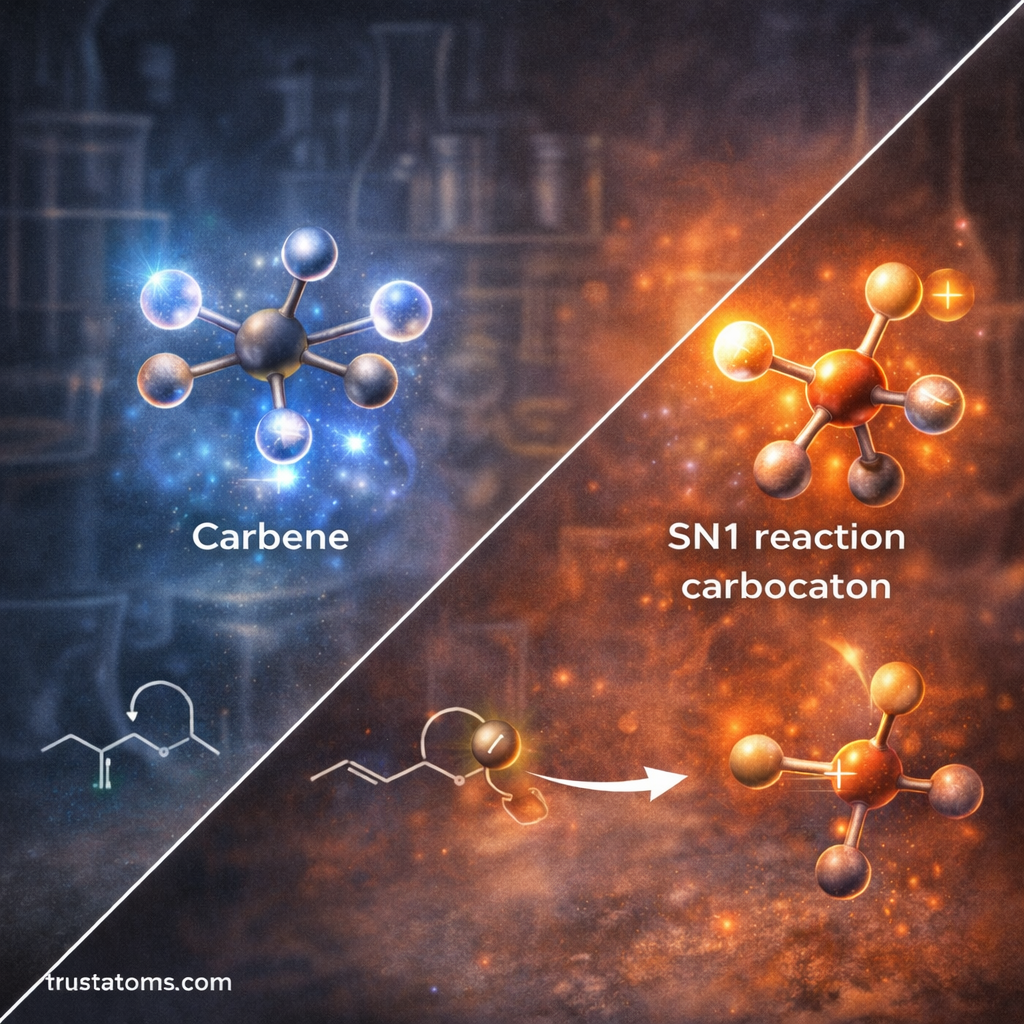
Carbocations
Carbocations are positively charged carbon atoms that contain only six electrons in their outer shell instead of the usual eight.
Characteristics of carbocations include:
- Strong electrophilic behavior
- Planar molecular geometry
- High reactivity toward electron-rich species
Carbocations commonly appear in reactions such as:
- Alkene addition reactions
- Rearrangement reactions
- SN1 substitution reactions
The stability of carbocations depends on the surrounding carbon groups and their ability to distribute positive charge.
Carbanions
Carbanions are negatively charged carbon atoms with an extra pair of electrons.
Key features include:
- Strong nucleophilic behavior
- High electron density
- Ability to attack positively charged or electron-deficient centers
Carbanions play important roles in reactions such as:
- Aldol reactions
- Enolate chemistry
- Carbon–carbon bond formation
Their stability often depends on nearby electron-withdrawing groups.
Free Radicals
Free radicals are atoms or molecules containing an unpaired electron.
Because electrons prefer to exist in pairs, radicals are extremely reactive and quickly react with other molecules.
Common radical reactions include:
- Combustion processes
- Polymerization reactions
- Halogenation of hydrocarbons
Radical intermediates are central to many industrial chemical processes.
Carbenes
Carbenes are neutral carbon species with only six valence electrons and two non-bonded electrons.
They are highly reactive and can insert themselves into chemical bonds.
Typical carbene reactions include:
- Cyclopropane formation
- Insertion into C–H bonds
- Rearrangement reactions
Carbenes can exist in two electronic states:
- Singlet carbenes
- Triplet carbenes
Each state exhibits different chemical behavior.
Nitrenes
Nitrenes are nitrogen-based reactive intermediates similar to carbenes but containing a nitrogen atom instead of carbon.
These species are often generated during photochemical or thermal reactions.
Nitrenes participate in reactions such as:
- Ring expansions
- Insertions into bonds
- Formation of nitrogen-containing compounds
Because they are extremely reactive, they are usually detected indirectly through reaction products.
Methods Used to Detect Reactive Intermediates
Because reactive intermediates are so short-lived, special techniques are required to study them.
Common methods include:
Spectroscopic Techniques
Advanced spectroscopy can sometimes detect intermediates directly.
Examples include:
- Ultraviolet-visible spectroscopy
- Infrared spectroscopy
- Nuclear magnetic resonance (NMR)
These methods help identify the structure and behavior of transient species.
Fast Reaction Methods
Techniques such as flash photolysis allow chemists to generate and observe intermediates within extremely short timeframes.
These experiments help reveal reaction pathways and intermediate lifetimes.
Trapping Experiments
In trapping experiments, a reagent is added to capture the intermediate before it disappears.
The trapped product provides evidence that the intermediate existed.
Computational Chemistry
Computer simulations can model reaction pathways and predict intermediate structures.
These theoretical methods help chemists visualize species that are difficult to observe experimentally.
Examples of Reactions Involving Reactive Intermediates
Many familiar organic reactions involve reactive intermediates.
Examples include:
- SN1 substitution reactions, which proceed through carbocation intermediates
- Radical polymerization, where free radicals initiate chain reactions
- Aldol reactions, which involve carbanion or enolate intermediates
- Photochemical reactions, which often generate carbenes or nitrenes
Studying these intermediates helps chemists control reaction conditions and improve yields.
Importance in Modern Chemistry
Reactive intermediates are fundamental to modern organic chemistry and chemical engineering.
Their study contributes to advancements in areas such as:
- Pharmaceutical development
- Synthetic organic chemistry
- Polymer science
- Green chemistry
- Catalysis research
By understanding how these short-lived species behave, chemists can design reactions that are faster, more selective, and more environmentally friendly.
The Future of Reactive Intermediate Research
Advances in spectroscopy, ultrafast laser technology, and computational modeling continue to improve our ability to study reactive intermediates.
Researchers are now able to observe reaction processes that occur in trillionths of a second, offering new insight into how molecules transform.
As scientific tools continue to evolve, the study of reactive intermediates will remain a central part of understanding and controlling chemical reactions at the molecular level.