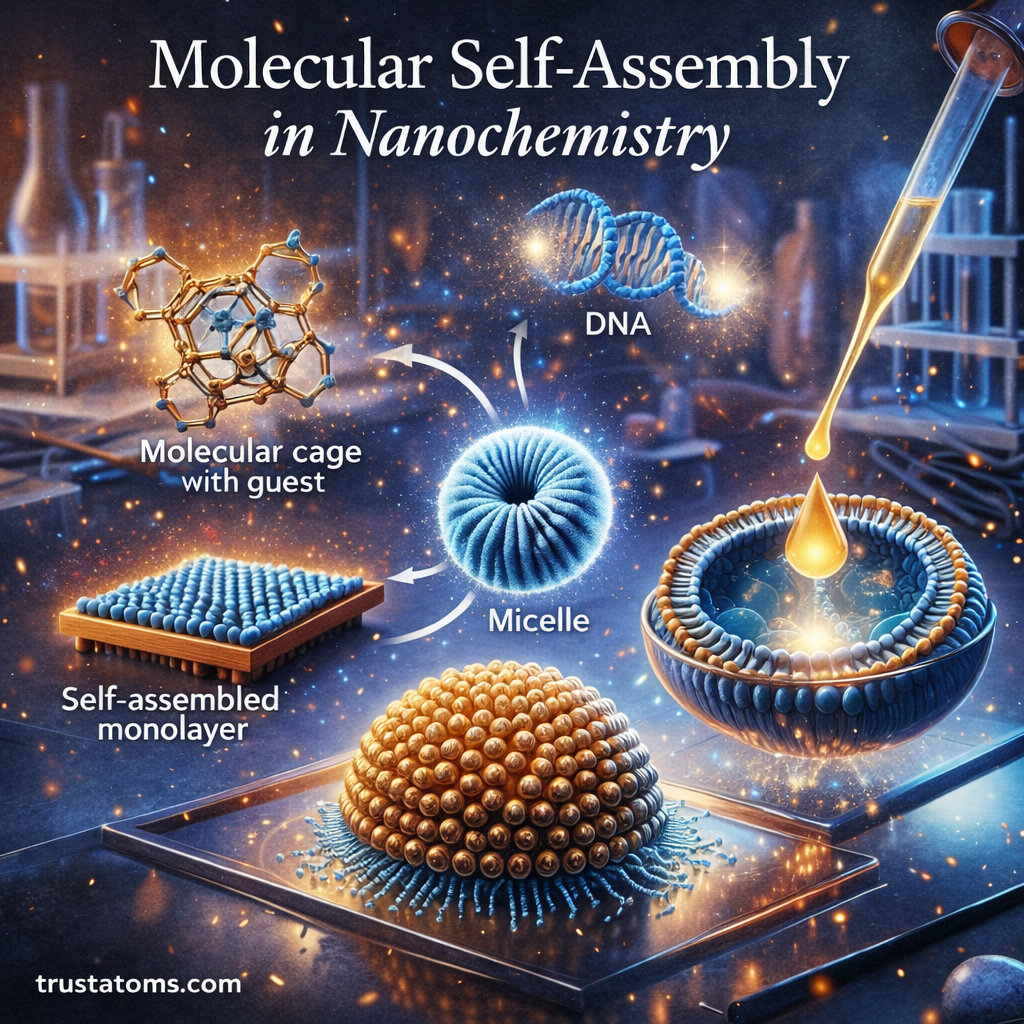
Molecular self-assembly is a fundamental concept in nanochemistry where molecules organize themselves into structured arrangements without external direction. Through natural chemical interactions, individual molecules can spontaneously form ordered patterns, layers, and complex nanostructures.
This phenomenon plays a critical role in the creation of advanced materials, nanotechnology devices, drug delivery systems, and biological structures. By understanding and controlling self-assembly processes, scientists can design materials at the molecular scale with precise functions and properties.
In many ways, molecular self-assembly allows chemistry to mimic nature’s ability to build complex structures efficiently and reliably.
What Is Molecular Self-Assembly?
Molecular self-assembly is the spontaneous organization of molecules into stable, ordered structures through non-covalent interactions.
These interactions include:
- Hydrogen bonding
- Electrostatic interactions
- Van der Waals forces
- Hydrophobic interactions
- π–π stacking between aromatic molecules
Unlike traditional chemical synthesis, where bonds are formed step by step, self-assembly occurs when molecules naturally arrange themselves into a preferred configuration based on their chemical properties.
This process often results in highly organized structures at the nanoscale.
Why Self-Assembly Matters in Nanochemistry
Nanochemistry focuses on materials and systems that operate at extremely small scales, typically between 1 and 100 nanometers.
Self-assembly is especially important at this scale because it enables:
- Precise molecular organization
- Efficient formation of complex nanostructures
- Reduced need for complex manufacturing processes
- Creation of functional materials with specialized properties
Rather than constructing nanoscale materials atom by atom, scientists can design molecules that automatically organize into the desired structure.
How Molecular Self-Assembly Works
Self-assembly occurs when molecules interact in ways that minimize the system’s overall energy.
The process generally follows several stages:
- Molecular recognition
Molecules identify compatible partners through complementary shapes, charges, or functional groups. - Initial aggregation
Small clusters begin to form as molecules interact through weak forces. - Structural organization
The clusters rearrange and grow into more ordered structures. - Formation of stable nanostructures
The system reaches a stable configuration with minimal energy.
Because these processes occur through reversible interactions, self-assembled systems can sometimes reorganize or repair themselves if disturbed.
Types of Molecular Self-Assembly

Different forms of self-assembly occur depending on the molecules involved and the environment in which they interact.
Static Self-Assembly
Static self-assembly occurs when a system reaches equilibrium and forms a stable structure.
Examples include:
- Crystal formation
- Molecular monolayers
- DNA double helices
Once formed, these structures remain stable unless external conditions change.
Dynamic Self-Assembly
Dynamic self-assembly occurs in systems that require continuous energy input.
These systems remain organized only while energy is supplied.
Examples include:
- Biological molecular motors
- Active nanomaterials
- Some chemical oscillating reactions
Dynamic self-assembly is common in living systems.
Common Self-Assembled Nanostructures
Molecular self-assembly can create a wide range of nanoscale structures.
Self-Assembled Monolayers (SAMs)
Self-assembled monolayers are thin molecular layers that form when molecules organize themselves on surfaces.
These layers are widely used in:
- Surface chemistry
- Biosensors
- Nanoelectronics
- Corrosion protection
SAMs allow scientists to modify surface properties with high precision.
Micelles
Micelles form when amphiphilic molecules organize themselves in solution.
These molecules contain:
- A hydrophilic (water-attracting) head
- A hydrophobic (water-repelling) tail
In water, the molecules arrange themselves so that the hydrophobic parts are hidden inside while the hydrophilic heads face outward.
Micelles are important in:
- Drug delivery
- Detergents
- Emulsion chemistry
Liposomes
Liposomes are spherical structures formed from lipid molecules.
They contain a bilayer membrane similar to biological cell membranes.
Liposomes are widely studied in pharmaceutical research because they can carry drugs and release them at targeted locations within the body.
Supramolecular Structures
Supramolecular chemistry studies large structures formed through non-covalent interactions.
Examples include:
- Molecular cages
- Host–guest complexes
- Molecular capsules
These structures are useful for molecular recognition and selective chemical reactions.
Applications of Molecular Self-Assembly
Self-assembly is used in many cutting-edge technologies.
Nanomaterials
Self-assembly allows scientists to build nanomaterials with specific mechanical, electrical, or optical properties.
Applications include:
- Nanoparticle arrays
- Photonic crystals
- Conductive nanomaterials
Drug Delivery Systems
Self-assembled nanostructures can transport drugs directly to specific cells or tissues.
Benefits include:
- Controlled drug release
- Reduced side effects
- Improved therapeutic efficiency
Liposomes and polymer nanoparticles are commonly used in these systems.
Electronics and Sensors
Self-assembled molecular layers are used in nanoelectronics and chemical sensing devices.
These materials help create:
- Molecular transistors
- Sensitive biosensors
- Flexible electronic materials
Biomimetic Materials
Self-assembly allows scientists to replicate structures found in biological systems.
Examples include:
- Artificial membranes
- Synthetic proteins
- Tissue engineering scaffolds
These materials can mimic the behavior of natural biological structures.
Factors That Influence Self-Assembly
Several environmental and chemical factors affect how molecules assemble.
Key factors include:
Molecular Shape
The geometry of molecules determines how they fit together during assembly.
Chemical Functional Groups
Specific functional groups can attract or repel each other, guiding molecular arrangement.
Temperature
Temperature affects molecular motion and the strength of intermolecular interactions.
Solvent Environment
Different solvents influence how molecules interact and organize.
Concentration
The number of molecules present in a system affects the likelihood of aggregation and structure formation.
Challenges in Controlling Self-Assembly
Although molecular self-assembly is powerful, controlling it precisely can be difficult.
Common challenges include:
- Maintaining structural stability
- Preventing unwanted aggregation
- Achieving consistent nanoscale patterns
- Scaling up processes for industrial production
Researchers continue developing methods to guide and control self-assembly more reliably.
The Future of Molecular Self-Assembly
Molecular self-assembly is expected to play a major role in the future of nanotechnology and advanced materials.
Researchers are exploring new areas such as:
- Programmable molecular systems
- Self-healing materials
- Smart nanomaterials
- Artificial molecular machines
These technologies may lead to breakthroughs in medicine, electronics, environmental science, and energy systems.
By learning how molecules organize themselves naturally, scientists can design new materials and technologies that operate with the same efficiency and precision found in nature.